DCBs for Coronary ISR Trial Designs Donald E

DCBs for Coronary ISR Trial Designs Donald E. Cutlip, MD Beth Israel Deaconess Medical Center Baim Institute for Clinical Research Professor of Medicine, Harvard Medical School

Donald Cutlip, MD Salary support: Baim Institute Scientific advisory board: Celonova Corvia

Unmet Need and Scientific Rationale • Effective and safe treatment for first and recurrent in-stent restenosis • Current standard of repeat DES effective but associated with risks: – – • Thrombosis risk Prolonged DAPT Reduce vessel flexibility Reduce vasomotion POBA has poor effectiveness and is not current standard of care
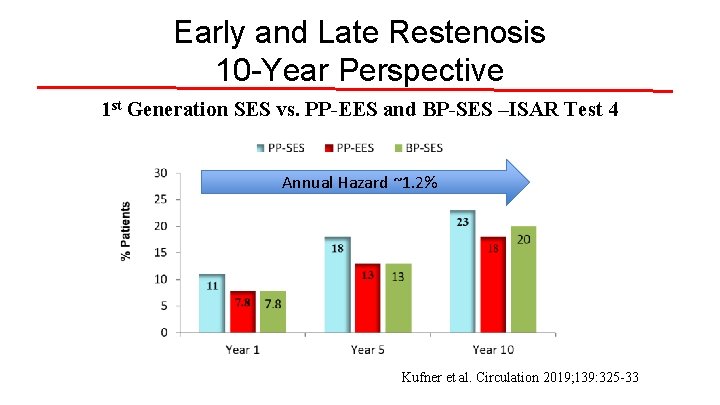
Early and Late Restenosis 10 -Year Perspective 1 st Generation SES vs. PP-EES and BP-SES –ISAR Test 4 Annual Hazard ~1. 2% Kufner et al. Circulation 2019; 139: 325 -33

DCB for ISR in Coronary Trials Study Question: Do Drug-coated (eluting) balloons (DCB) provide reasonable assurance of safety and effectiveness? 1. 2. Effectiveness – Are DCB significantly more effective than POBA? – Are DCB clinically and statistically non-inferior to DES? Safety – Are there concerning trends in major coronary safety endpoints? (Death, MI, vessel thrombosis) – Drug toxicity or particulate embolization adverse events? – Safety benefits (reduce DAPT, bleeding, vessel healing)

Clinical Trial Designs for ISR Clinical Trial Population • BMS or DES ISR • 0 -1 prior restenosis > RCT • 2 or more prior restenosis excluded from RCT; consider possible nonrandomized parallel registry

Options for Randomized Trial Designs • Compare DCB to a POBA control – Superiority design for clinical effectiveness – Risk of slow enrollment (NCDR data = ~20% POBA) – Risk of excessive “bail-out” stenting in POBA group • Reduce potential for demonstrating superiority – Risk of “straw man” control – must verify procedural success • Compare DCB to contemporary DES (EES) – Non-inferiority for clinical effectiveness endpoint – ~20% of ISR treated without DES (limits enrollment)

Options for Randomized Trial Designs • Compare DCB to a SOC control – ≥ 80% repeat DES; ≤ 20% POBA – Non-inferiority based on meta-analytic rate assuming 80% DES – Non-inferiority margin based on conserving at least 50% of delta for DES versus POBA

Endpoints • Primary effectiveness – Target lesion failure (cardiac death, TV MI, TLR) at 12 months • Secondary endpoints – – TLF components Safety (stent thrombosis, bleeding) Angiographic subset (FU MLD, late loss) OCT subset (NIH, uncovered struts, malapposition)

Clinical Trial Design Outlines DCB vs. DES DCB vs. POBA • Primary endpoint TLF • Non-inferiority margin ~10% • Null = no difference (50% of lowest estimated effect for DES vs. POBA) • Estimated POBA TLF rate =. 31 (based on lower 95% CI of meta analysis) • Alpha 0. 05; Power 90%; • Estimate DCB = 30% reduction • Sample size ~ 300 (2: 1) • Estimated event rates ~10% (based on upper DES rate) • Alpha 1 -sided 0. 025; Power 90% • Sample size ~ 360

Clinical Trial Designs Other Options • Non-randomized single arm study vs. performance goal Unlikely to be acceptable to FDA • Surrogate endpoint with earlier timepoint, smaller sample (angiographic, OCT) Might allow earlier submission; Still require clinical EP for approval • Hybrid control group Similar sample size as DES control; May improve enrollment; Close to real-world practice. (80% DES, 20% POBA per NCDR observational data)

Conclusions • There is a clear unmet need for coronary ISR treatment. • Anticipate at least 10% of cases due to ISR over time. • DCBs may offer advantages over POBA or repeat DES. • Clinical trials must demonstrate superiority over POBA and approximate the effect of repeat DES for a meaningful clinical endpoint. • The important secondary advantage of avoiding an additional stent layer should allow for favorable noninferiority margin versus DES.
- Slides: 12