DCB for Peripheral Application Show Me the Data

DCB for Peripheral Application: Show Me the Data I – DCB for SFA Lesions Marianne Brodmann Division of Angiology, Medical University Graz, Austria

Marianne Brodmann, MD, Prof I have following financial relationships Type of Relationship Advisory Board, Consultant, Honoraria, Speaker’s Bureau BARD, Medtronic, Daiichi, Bayer, Intact, Spectranetics, Avinger, CSI, Shockwave
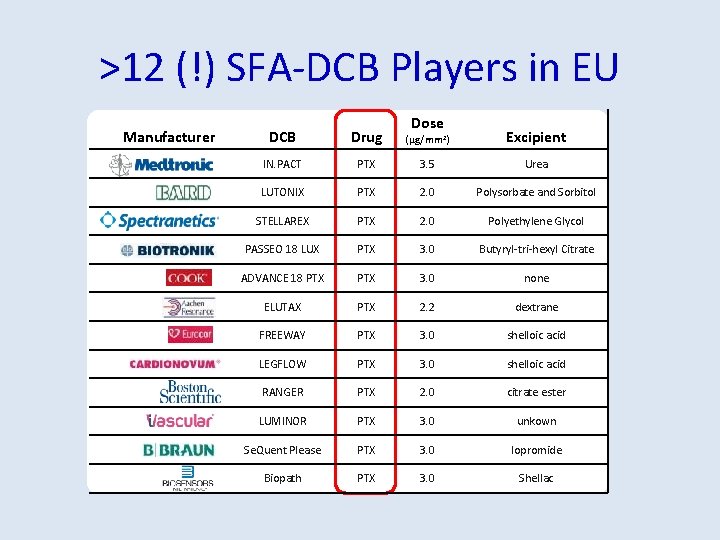
>12 (!) SFA-DCB Players in EU Manufacturer Dose DCB Drug (μg/mm 2) Excipient IN. PACT PTX 3. 5 Urea LUTONIX PTX 2. 0 Polysorbate and Sorbitol STELLAREX PTX 2. 0 Polyethylene Glycol PASSEO 18 LUX PTX 3. 0 Butyryl-tri-hexyl Citrate ADVANCE 18 PTX 3. 0 none ELUTAX PTX 2. 2 dextrane FREEWAY PTX 3. 0 shelloic acid LEGFLOW PTX 3. 0 shelloic acid RANGER PTX 2. 0 citrate ester LUMINOR PTX 3. 0 unkown Se. Quent Please PTX 3. 0 Iopromide Biopath PTX 3. 0 Shellac

DCB for SFA Lesions DCB SFA Technologies: Same Drug, Similar Mode of Action Different Technologies ü Different Dose (2. 0 3. 5 μg/mm 2) ü Different Drug Formulation (cristal vs. amorphous vs. hybrid) ü Different Excipients (or no-excipient) ü Different Surface Energies ü Different Coating Methods ü Different usage method per IFU (i. e. with or w/out protective sheath) ü ……………. .
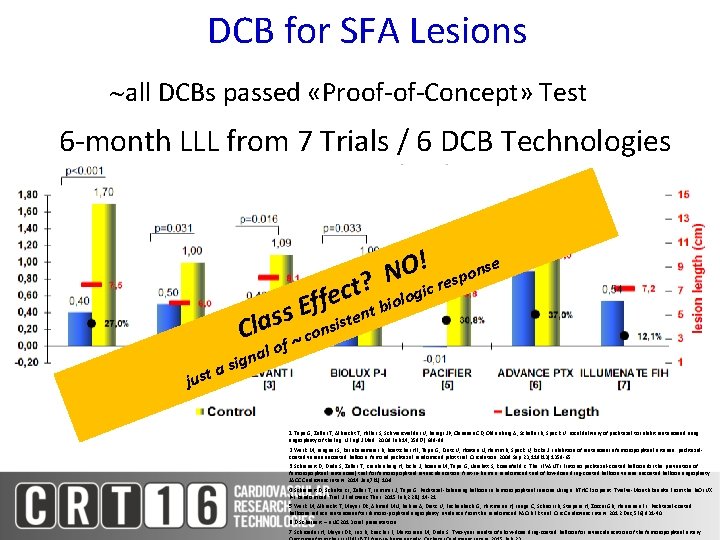
DCB for SFA Lesions all DCBs passed «Proof-of-Concept» Test 6 -month LLL from 7 Trials / 6 DCB Technologies O! esponse N ? cr t i c g o fe nt biol f E s nsiste s a l C f co just nal g i s a o 1. Tepe G, Zeller T, Albrecht T, Heller S, Schwarzwälder U, Beregi JP, Claussen CD, Oldenburg A, Scheller B, Speck U. Local delivery of paclitaxel to inhibit restenosis during angioplasty of the leg. N Engl J Med. 2008 Feb 14; 358(7): 689 -99 2. Werk M, Langner S, Reinkensmeier B, Boettcher HF, Tepe G, Dietz U, Hosten N, Hamm B, Speck U, Ricke J. Inhibition of restenosis in femoropopliteal arteries: paclitaxelcoated versus uncoated balloon: femoral paclitaxel randomized pilot trial. Circulation. 2008 Sep 23; 118(13): 1358 -65 3. Scheinert D, Duda S, Zeller T, Krankenberg H, Ricke J, Bosiers M, Tepe G, Naisbitt S, Rosenfield K. The LEVANT I (Lutonix paclitaxel-coated balloon for the prevention of femoropopliteal restenosis) trial for femoropopliteal revascularization: first-in-human randomized trial of low-dose drug-coated balloon versus uncoated balloon angioplasty. JACC Cardiovasc Interv. 2014 Jan; 7(1): 10 -9 4. Scheinert D, Schulte KL, Zeller T, Lammer J, Tepe G. Paclitaxel-Releasing Balloon in Femoropopliteal Lesions Using a BTHC Excipient: Twelve-Month Results From the BIOLUX P-I Randomized Trial. J Endovasc Ther. 2015 Feb; 22(1): 14 -21 5. Werk M, Albrecht T, Meyer DR, Ahmed MN, Behne A, Dietz U, Eschenbach G, Hartmann H, Lange C, Schnorr B, Stiepani H, Zoccai GB, Hänninen EL. Paclitaxel-coated balloons reduce restenosis after femoro-popliteal angioplasty: evidence from the randomized PACIFIER trial. Circ Cardiovasc Interv. 2012 Dec; 5(6): 831 -40 6. D. Scheinert – LINC 2013 oral presentation 7. Schroeder H, Meyer DR, Lux B, Ruecker F, Martorana M, Duda S. Two-year results of a low-dose drug-coated balloon for revascularization of the femoropopliteal artery:

DCB for SFA Lesions 3 DCB with sound, comprehensive Clinical Programs Manufacturer Dose DCB Drug (μg/mm 2) Excipient IN. PACT PTX 3. 5 Urea LUTONIX PTX 2. 0 Polysorbate and Sorbitol STELLAREX PTX 2. 0 Polyethylene Glycol PASSEO 18 LUX PTX 3. 0 Butyryl-tri-hexyl Citrate ADVANCE 18 PTX 3. 0 none ELUTAX PTX 2. 2 dextrane FREEWAY PTX 3. 0 shelloic acid LEGFLOW PTX 3. 0 shelloic acid RANGER PTX 2. 0 citrate ester LUMINOR PTX 3. 0 unkown Se. Quent Please PTX 3. 0 Iopromide Biopath PTX 3. 0 Shellac

DCB for SFA Lesions 3 DCB with sound, comprehensive Clinical Programs Manufacturer Dose DCB Drug (μg/mm 2) Excipient IN. PACT PTX 3. 5 Urea LUTONIX PTX 2. 0 Polysorbate and Sorbitol STELLAREX PTX 2. 0 Polyethylene Glycol 3 DCB with 2 -year Core lab adjudicated Primary Patency ü same definition (PSVR ≤ 2. 4) ü same KM reporting method ü same Duplex Core laboratory
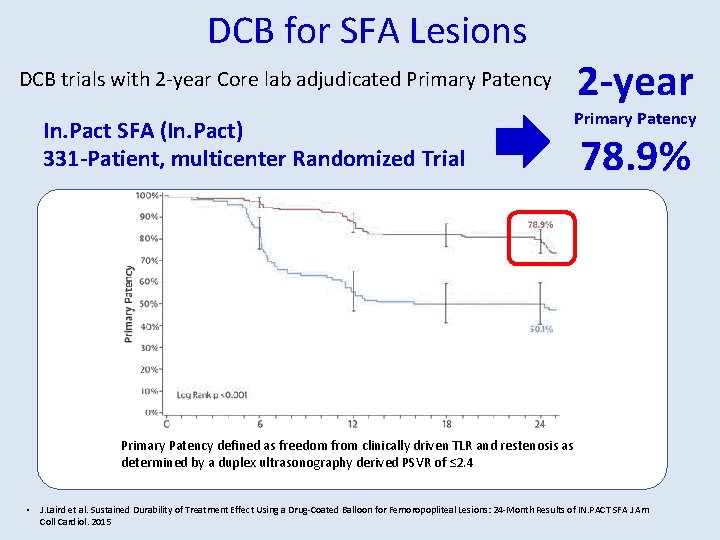
DCB for SFA Lesions DCB trials with 2 -year Core lab adjudicated Primary Patency In. Pact SFA (In. Pact) 331 -Patient, multicenter Randomized Trial 2 -year Primary Patency 78. 9% Primary Patency defined as freedom from clinically driven TLR and restenosis as determined by a duplex ultrasonography derived PSVR of ≤ 2. 4 • J. Laird et al. Sustained Durability of Treatment Effect Using a Drug-Coated Balloon for Femoropopliteal Lesions: 24 -Month Results of IN. PACT SFA J Am Coll Cardiol. 2015
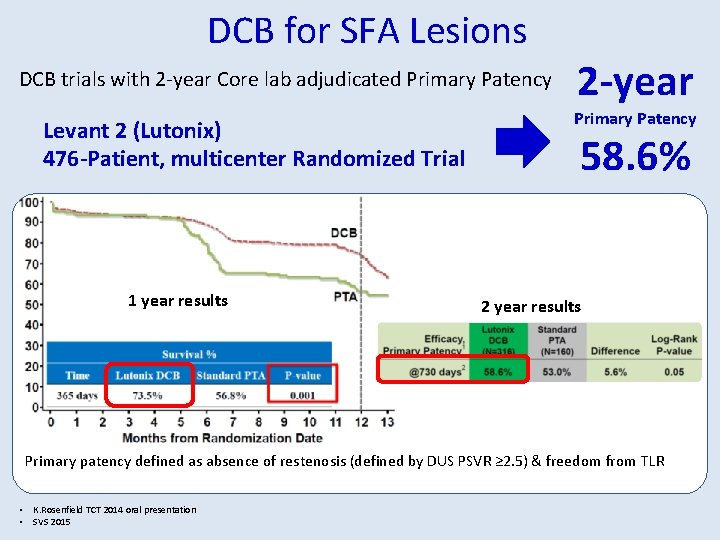
DCB for SFA Lesions DCB trials with 2 -year Core lab adjudicated Primary Patency Levant 2 (Lutonix) 476 -Patient, multicenter Randomized Trial 1 year results 2 -year Primary Patency 58. 6% 2 year results Primary patency defined as absence of restenosis (defined by DUS PSVR ≥ 2. 5) & freedom from TLR • K. Rosenfield TCT 2014 oral presentation • SVS 2015
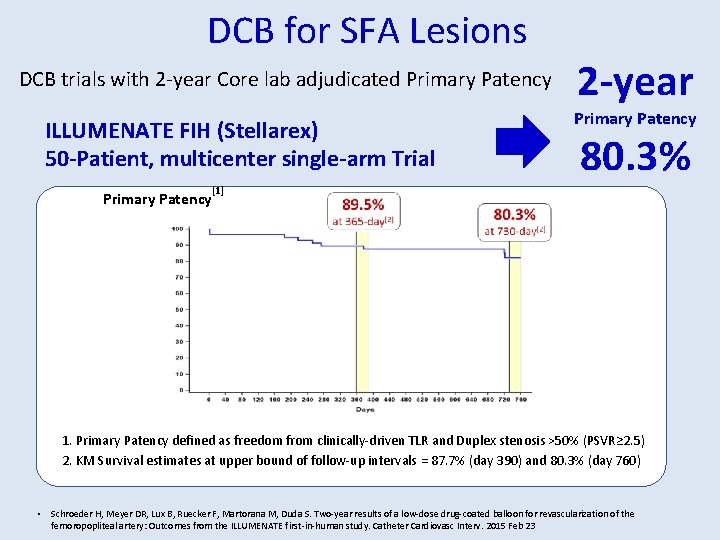
DCB for SFA Lesions DCB trials with 2 -year Core lab adjudicated Primary Patency ILLUMENATE FIH (Stellarex) 50 -Patient, multicenter single-arm Trial Primary Patency 2 -year Primary Patency 80. 3% [1] 1. Primary Patency defined as freedom from clinically-driven TLR and Duplex stenosis >50% (PSVR≥ 2. 5) 2. KM Survival estimates at upper bound of follow-up intervals = 87. 7% (day 390) and 80. 3% (day 760) • Schroeder H, Meyer DR, Lux B, Ruecker F, Martorana M, Duda S. Two-year results of a low-dose drug-coated balloon for revascularization of the femoropopliteal artery: Outcomes from the ILLUMENATE first-in-human study. Catheter Cardiovasc Interv. 2015 Feb 23

DCB for SFA Lesions Role of Large scale Registries • Designed and powered to: – unveil DCB performance within the broad PAD spectrum – detect potential rare events • Critical to confirm each DCB’s Efficacy and Safety • Important to inform the decision whether a DCB class effect exists or not

DCB for SFA Lesions IN. PACT Global Study: Real-world, prospective, multicenter, single arm independently-adjudicated femoropopliteal study • >1, 500 patients enrolled All-comers (RCC 2 -4) √ √ √ √ Bilateral disease Multiple lesions SFA and Popliteal TASC A, B, C, D de novo ISR Long Lesions CTOs 1. Syntactx Clinical Events Committee, New York, NY, US 2. Vas. Core DUS Core Lab, Boston, MA, US 3. Synva. Cor Angiographic Core Lab, Springfield, IL, US • 64 sites in EU, Mid-East, Latin America, Asia • Independent adjudication by Clinical Events Committee 1 • Prospective subset analysis with core lab 2, 3 reported results (de novo ISR, long lesions ≥ 15 cm, CTOs ≥ 5 cm) • Safety and effectiveness data on 150 mm DCB

DCB for SFA Lesions IN. PACT Global Study Patient Cohorts >1, 500 patients enrolled Clinical Cohort Imaging Subsets >1, 400 pts de novo ISR* ≥ 150 pts Long Lesion (≥ 15 cm) ≥ 150 pts ≥ 100 pts DCB 150 mm CTO (≥ 5 cm) ≥ 150 pts
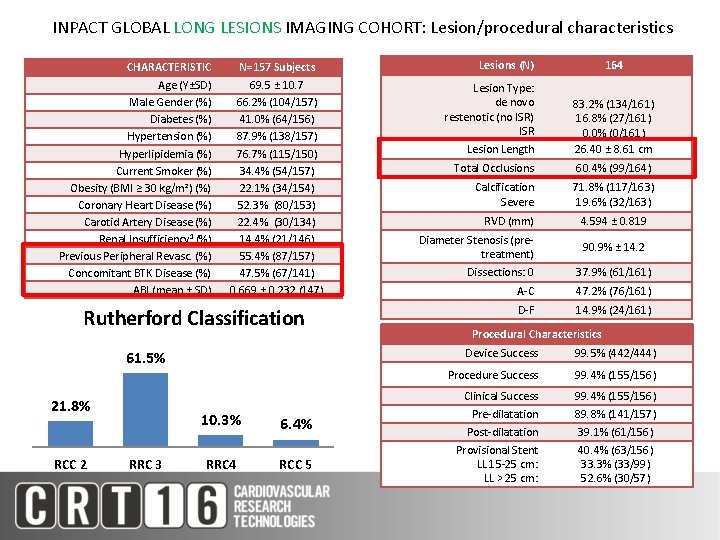
INPACT GLOBAL LONG LESIONS IMAGING COHORT: Lesion/procedural characteristics CHARACTERISTIC Age (Y±SD) Male Gender (%) Diabetes (%) Hypertension (%) Hyperlipidemia (%) Current Smoker (%) Obesity (BMI ≥ 30 kg/m²) (%) Coronary Heart Disease (%) Carotid Artery Disease (%) Renal Insufficiency 1 (%) Previous Peripheral Revasc. (%) Concomitant BTK Disease (%) ABI (mean ± SD) N=157 Subjects 69. 5 ± 10. 7 66. 2% (104/157) 41. 0% (64/156) 87. 9% (138/157) 76. 7% (115/150) 34. 4% (54/157) 22. 1% (34/154) 52. 3% (80/153) 22. 4% (30/134) 14. 4% (21/146) 55. 4% (87/157) 47. 5% (67/141) 0. 669 ± 0. 232 (147) Rutherford Classification 61. 5% 21. 8% RCC 2 RRC 3 10. 3% 6. 4% RRC 4 RCC 5 Lesions (N) Lesion Type: de novo restenotic (no ISR) ISR 164 Lesion Length 83. 2% (134/161) 16. 8% (27/161) 0. 0% (0/161) 26. 40 ± 8. 61 cm Total Occlusions 60. 4% (99/164) Calcification Severe 71. 8% (117/163) 19. 6% (32/163) RVD (mm) 4. 594 ± 0. 819 Diameter Stenosis (pretreatment) 90. 9% ± 14. 2 Dissections: 0 37. 9% (61/161) A-C 47. 2% (76/161) D-F 14. 9% (24/161) Procedural Characteristics Device Success 99. 5% (442/444) Procedure Success 99. 4% (155/156) Clinical Success Pre-dilatation Post-dilatation Provisional Stent LL 15 -25 cm: LL > 25 cm: 99. 4% (155/156) 89. 8% (141/157) 39. 1% (61/156) 40. 4% (63/156) 33. 3% (33/99) 52. 6% (30/57)
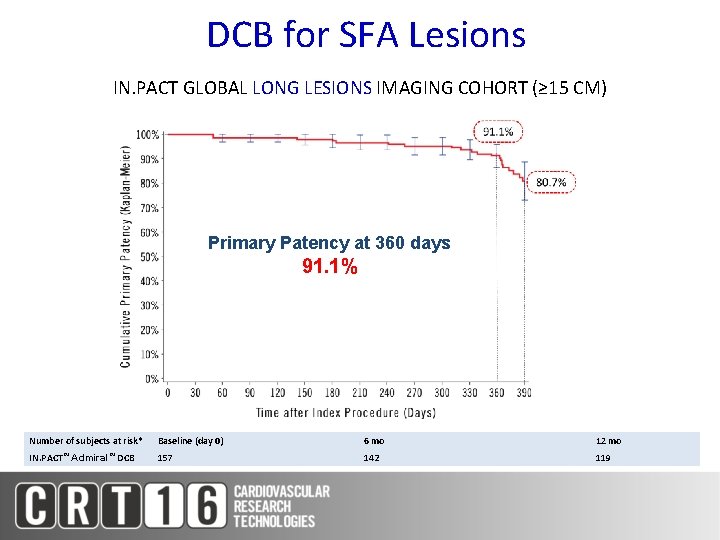
DCB for SFA Lesions IN. PACT GLOBAL LONG LESIONS IMAGING COHORT (≥ 15 CM) Primary Patency at 360 days 91. 1% Number of subjects at risk* Baseline (day 0) 6 mo 12 mo IN. PACT™ Admiral™ 157 142 119 DCB
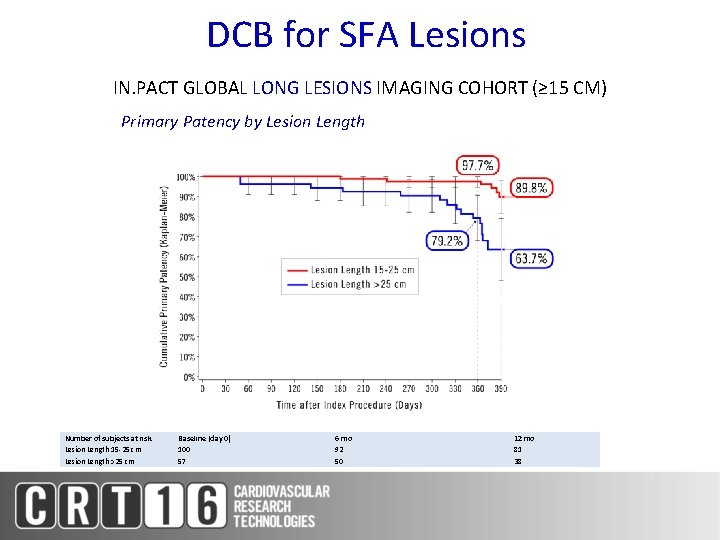
DCB for SFA Lesions IN. PACT GLOBAL LONG LESIONS IMAGING COHORT (≥ 15 CM) Primary Patency by Lesion Length Number of subjects at risk Lesion Length 15 -25 cm Lesion Length >25 cm Baseline (day 0) 100 57 6 mo 92 50 12 mo 81 38
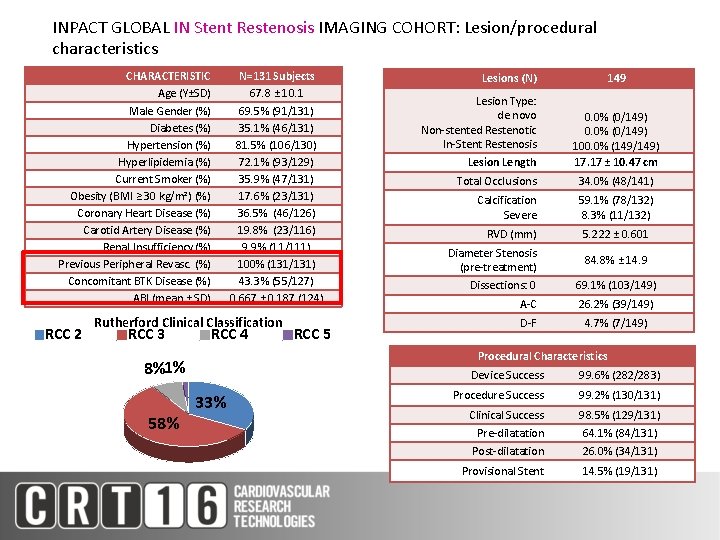
INPACT GLOBAL IN Stent Restenosis IMAGING COHORT: Lesion/procedural characteristics CHARACTERISTIC Age (Y±SD) Male Gender (%) Diabetes (%) Hypertension (%) Hyperlipidemia (%) Current Smoker (%) Obesity (BMI ≥ 30 kg/m²) (%) Coronary Heart Disease (%) Carotid Artery Disease (%) Renal Insufficiency (%) Previous Peripheral Revasc. (%) Concomitant BTK Disease (%) ABI (mean ± SD) RCC 2 N=131 Subjects 67. 8 ± 10. 1 69. 5% (91/131) 35. 1% (46/131) 81. 5% (106/130) 72. 1% (93/129) 35. 9% (47/131) 17. 6% (23/131) 36. 5% (46/126) 19. 8% (23/116) 9. 9% (11/111) 100% (131/131) 43. 3% (55/127) 0. 667 ± 0. 187 (124) Rutherford Clinical Classification RCC 3 RCC 4 Lesion Type: de novo Non-stented Restenotic In-Stent Restenosis Lesion Length 149 0. 0% (0/149) 100. 0% (149/149) 17. 17 ± 10. 47 cm Total Occlusions 34. 0% (48/141) Calcification Severe 59. 1% (78/132) 8. 3% (11/132) RVD (mm) 5. 222 ± 0. 601 Diameter Stenosis (pre-treatment) 84. 8% ± 14. 9 Dissections: 0 69. 1% (103/149) A-C 26. 2% (39/149) D-F 4. 7% (7/149) Procedural Characteristics 8%1% 58% RCC 5 Lesions (N) 33% Device Success 99. 6% (282/283) Procedure Success 99. 2% (130/131) Clinical Success Pre-dilatation Post-dilatation 98. 5% (129/131) 64. 1% (84/131) 26. 0% (34/131) Provisional Stent 14. 5% (19/131)
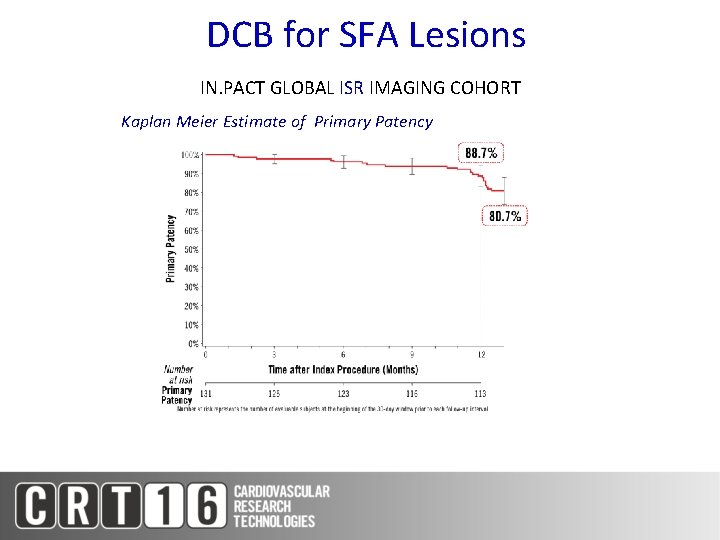
DCB for SFA Lesions IN. PACT GLOBAL ISR IMAGING COHORT Kaplan Meier Estimate of Primary Patency
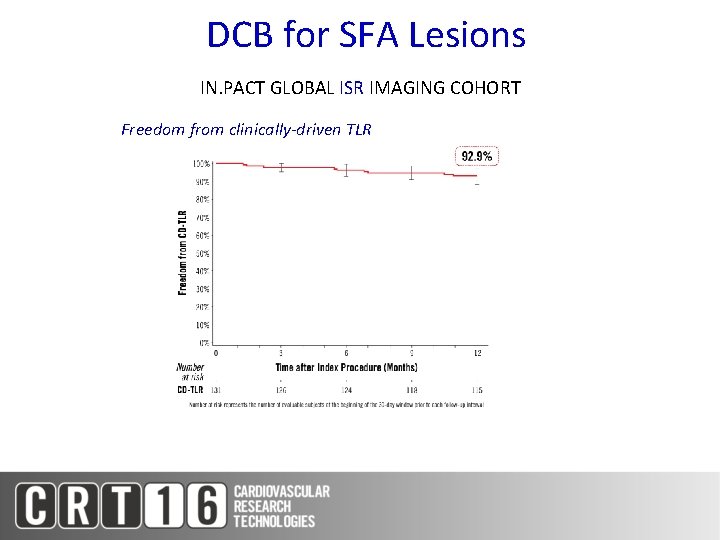
DCB for SFA Lesions IN. PACT GLOBAL ISR IMAGING COHORT Freedom from clinically-driven TLR

DCB for SFA Lesions Results across IN. PACT Clinical Studies IN. PACT SFA (DCB Arm) (N=220) IN. PACT Global Long Lesion Imaging Cohort (N=157) IN. PACT Global De Novo ISR Imaging Cohort (N=131) 8. 9 26. 4 17. 2 Primary Patency (KM 12 Month) 87. 5% 91. 1% 88. 7% CD-TLR 2. 4% 6. 0% 7. 3% CD-TVR 4. 3% 6. 0% 8. 9% Thrombosis 1. 4% 3. 7% 0. 8% Lesion Length (cm)

DCB for SFA Lesions Conclusions ü Clinical Results appraisal is two-step: 1) Quality of Evidence, 2) Quality of Outcomes ü 3 DCBs deserve attention based on the quality of their trials and trial programs ü 2 DCBs deserve adoption based on the quality of such trial’s results ü No Class effect: each DCB stands on the quality of its own evidence and associated outcomes ü Future work horse tool for SFA/Prox PA disease

Thank you for your attention
- Slides: 22