DBT 301 POWDER METALLURGY CHAPTER 3 POWDER FABRICATION

DBT 301 POWDER METALLURGY CHAPTER 3: POWDER FABRICATION Semester 1 2016/2017

3. 0 Outline 1) Introduction 2) Mechanical techniques 3) Electrolytic techniques 4) Chemical techniques 5) Atomization techniques 6) Forming specific metal powders

1. Introduction A knowledge in how a powder is fabricated is needed to estimate its characteristic. Almost all materials can be made into powder. The method of powder fabrication to be selected depends on the material properties. Powder formation involves the delivery of energy to the material to create a new surface area. 4 important characteristics: 1. 2. 3. 4. The efficiency of the process. The energy content. Types of feedstock. Sources of contamination.

2. Mechanical Fabrication 4 fundamental mechanical processes: � Impaction � Attritioning � Shearing � Compression

2. Mechanical Fabrication 1) Impaction � Involves rapid instantaneous delivery of a blow to a material, cause crack & size reduction. 2) Attritioning � Reduction in particle size by rubbing motion. 3) Shearing � By cutting, a cleavage type of fracture is formed. � Powder formed is coarse, not often used unless material is extremely hard. 4) Compression � Can break material into coarse powder if it is brittle.

2. Mechanical Fabrication Attrition, impact, shear & compression usually combined to form metal powder. Powder produced is often irregular in shape.

2. Mechanical Fabrication 3 methods; � Machining � Milling � Mechanical alloying

2. Mechanical Fabrication (I) Machining � An easy technique, useful for small scale production. � But lack of control over powder characteristics, inefficient & slow, too irregular or coarse. � Is not a first choice to fabricate powder. � Useful in consuming scrap from other process. � Require further milling. � Done on lathe machine by applying force to get fine metal chips � Produce large amount of scrap further reduced in size by grinding


2. Mechanical Fabrication (II) Milling � Impaction using hard balls, a classic technique to fabricate powder (brittle materials). � Impact & shear force between two materials – a hard & a soft – causing the soft material to be ground into fine particles � Using jar mill consist of balls, with materials. � When jar rotates, ball collide with materials, crushing it into powder. � Large particle = less impact stress � Smaller particle = impact stress increase � Mill time depend on power, change in particle size,

2. Mechanical Fabrication � Milling is not useful for ductile metals that cannot be fractured easily. � Rotation of jar must fast enough to carry the balls to the top of the jar before falling back on the bed of material. � Optimal milling = balls diameter approximately 30 x diameter of material, balls volume ½ jar volume, material fill ½ of jar volume. � Fluid or inert atm. used to reduce oxide & help in grinding.

2. Mechanical Fabrication

2. Mechanical Fabrication � This create metal powder of irregular, poor flow & packing. � Have problems of contamination from jar & balls.





2. Mechanical Fabrication (III) Mechanical Alloying � Since 1960’s, employ of attritioning motion between agitated balls to create alloy composite particle. � Start with mixing balls & powders in stirred mill, then repeat impact, cold welding & fracture. � Contamination problem: use balls, stir rod & tank same material as powder.

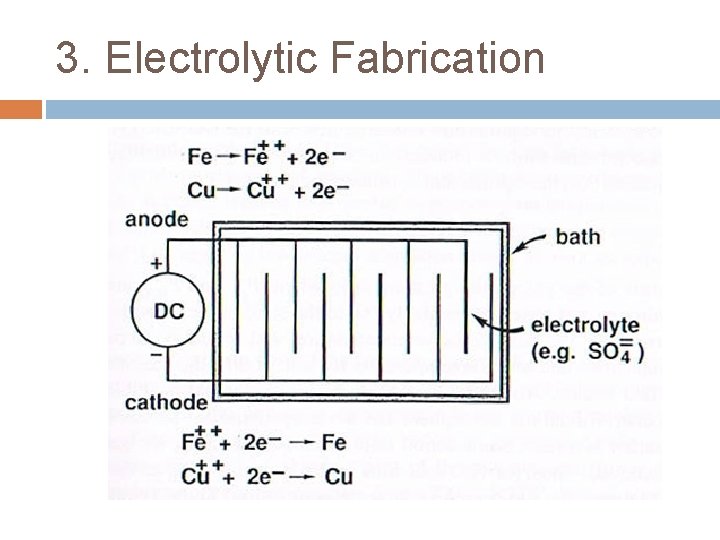
3. Electrolytic Fabrication Powder can be precipitated at cathode of electrolytic cell under certain operating conditions. Pd, Cu, Fe, Zn, Mn, Ag. Dissolution of anode under applied voltage in an electrolytic cell. Anode & Cathode reacts – transport through eletrolyte – purify deposit formed at cathode. Porous cathode is removed – washer – dried – grind into fine powder – annealed.
3. Electrolytic Fabrication

3. Electrolytic Fabrication Main benefit is high purity product. Powder formed shape is often dendritic or sponge. Contamination will disturb formation & deposition of powders at cathode. Product must be clean and milled after fabrication (add cost & process). Environmental problems (chemical).

3. Electrolytic Fabrication

4. Chemical Fabrication 4 methods; Decomposition of solid by Gas Thermal decomposition Precipitation from Liquid Precipitation from Gas

4. Chemical Fabrication (I) Decomposition of solid by Gas � A purified oxide milled into fine powders. � Oxide reduction by thermochemical reactions involving reducing gases such as CO or H. � Low temperature for producing small particle sizes ensure minimal diffusional bonding. � Usually sponge shape as it involved large volume change in low temp oxide reduction. � Powder is grinded to fracture interparticle diffusion bonds & to achieve particle size. � Annealing process to improve compressibility of

4. Chemical Fabrication (II) Thermal decomposition � Powder were fabricated by the combination of vapor decomposition & condensation. � Formation of gaseous molecule requires simultaneous pressurization & heating. � The molecule is then cooled to a liquid & to purify it, fractional distillation is applied. � Reheating the liquid with catalyst leading to vapor decomposition which gives a metal powder.

4. Chemical Fabrication (III) Precipitation from Liquid � Dissolved metal salt such as nitrate, chloride or sulfate can be treated to produce metallic precipitate or metal containing precipitate. � Salt is dissolved in water and precipitated by a second compound. � Resulting solid Ag precipitate is milled to a powder.

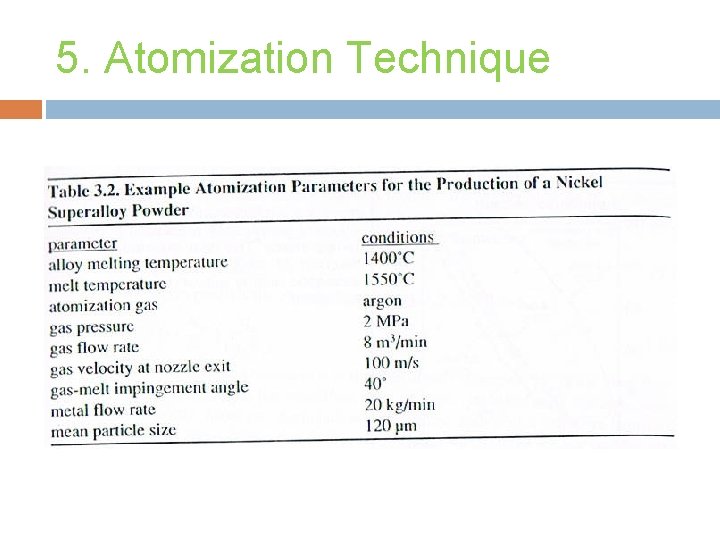
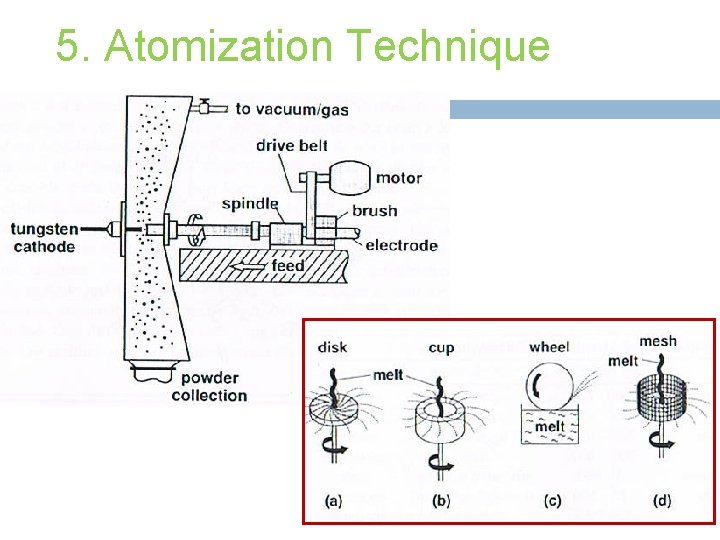
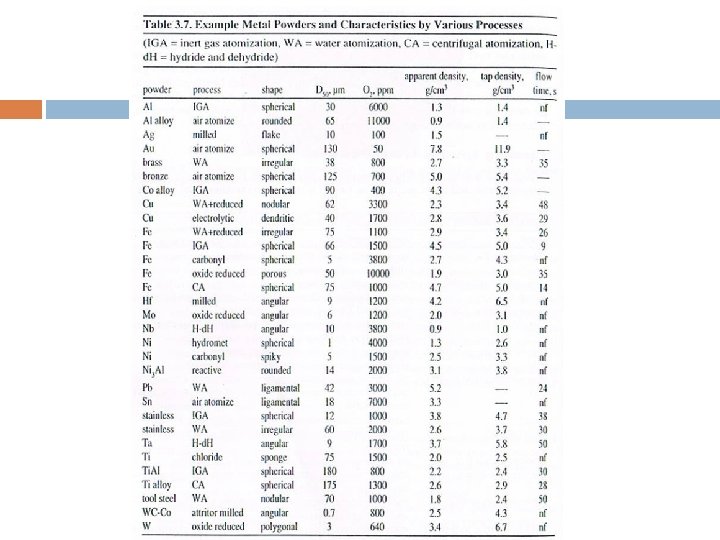
5. Atomization Technique Powder is formed from molten metal using a spray of droplet. Applicable to several alloys & easy process control. Fusion based technology that provides control over melt purification and alloy chemistry.

5. Atomization Technique Gas Atomization. Water Atomization. Centrifugal Atomization. Others.

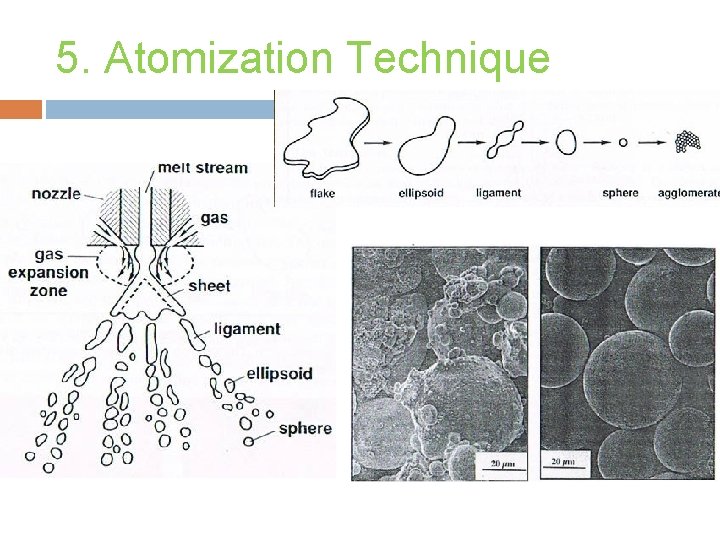
5. Atomization Technique (I) Gas Atomization � Use air, nitrogen, helium or argon as fluid to break up molten metal stream. � The liquid metal stream is disintegrated by rapid gas expansion out of a nozzle. � Can be performed totally under inert conditions. � Spherical particle shape, wide size distribution. � Process has too many operating variables. � Advantages: product homogeneity, good packing properties.

5. Atomization Technique
5. Atomization Technique
5. Atomization Technique

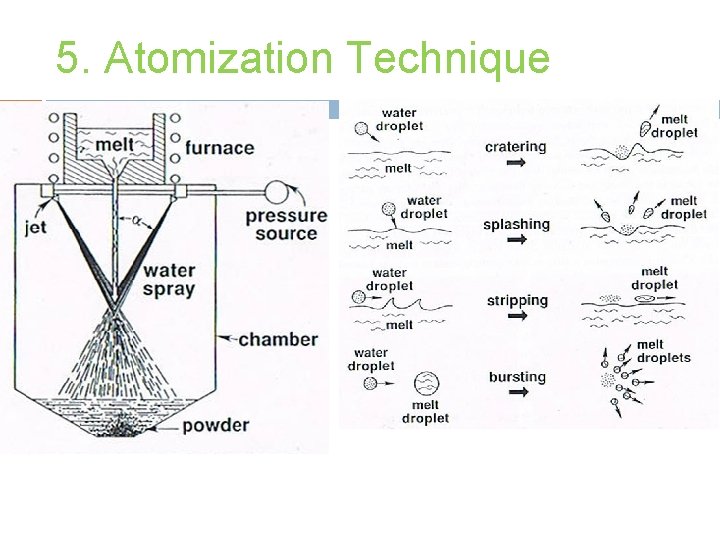
5. Atomization Technique (II) Water Atomization � Most common technique to produce elemental and alloy powders from metals which melt below 1600°C. � Process is similar to gas atomization except for rapid quenching and differing fluid properties. � Particle shape irregular & rough with some oxidation. � Synthetic oils/other nonreactive liquids can be used instead of waters to counter above problems.
5. Atomization Technique

5. Atomization Technique (III) Centrifugal Atomization � Developed this method due to control particle size & to fabricate powders from reactive metals. � The benefits of rotating electrode process are; - powder cleanliness, - spherical shape (high packing density & easy flow), - uniform particle size, and - low contamination. � Disadvantages: low production rates, high equipment & processing costs.
5. Atomization Technique

5. Atomization Technique

5. Atomization Technique Others � Vibrating wires, high speed rollers, spinning crucibles, & melt explosion atomizers.

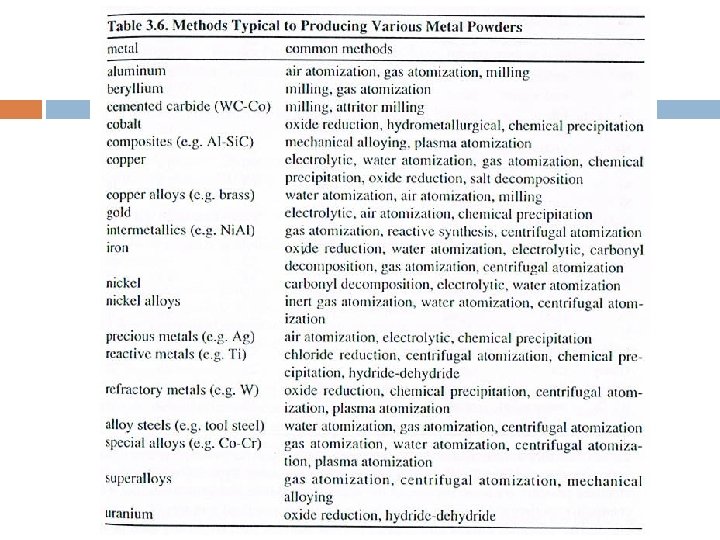
6. Forming Specific Metal Powders. Water atomization: iron, copper, tin & SS. Gas/Centrifugal Atomization: high purity materials for aerospace; superalloys, aluminum alloys, titanium alloys & high strength steels.

~END~
- Slides: 43