DBM PRODUCT PERFORMANCE CHRISTOPHER SABATINO PH D DIRECTOR

DBM PRODUCT PERFORMANCE CHRISTOPHER SABATINO PH. D. DIRECTOR, BIOLOGICS MARKETING MEDTRONIC SPINAL

THE MEDTRONIC MISSION Unchanged since 1960… To contribute to human welfare by application of biomedical engineering in the research, design, manufacture, and sale of instruments or appliances that alleviate pain, restore health, and extend life. PMD 017283 – 1. 0

HOW TO CHOOSE? Patient’s osteogenic capacity and product indications should drive decisions to achieve best outcomes Contributing Factors: Volume of Defect Site Complexity Vascularity As Osteogenic Capacity Decreases… …Reliance on the Graft Material Increases. PMD 017283 – 1. 0 Comorbidities Environmental Factors Considerations: Ability to Regenerate Ability to Revascularize Site Ability to Fully Consolidate Ability to Minimize Healing Time
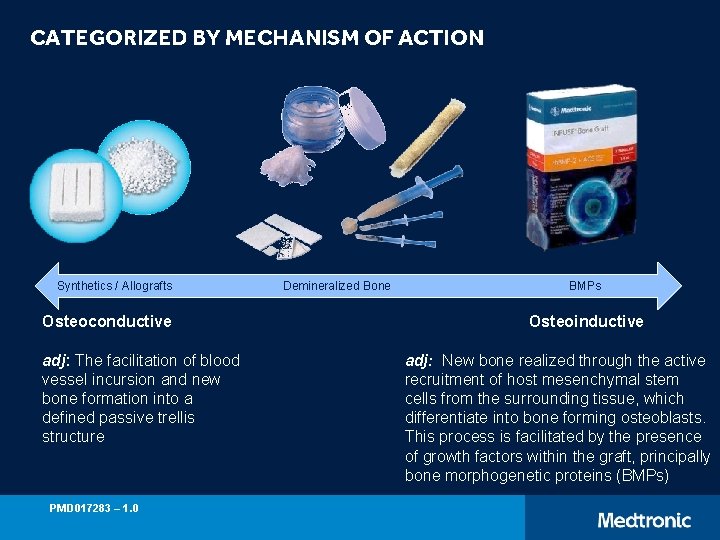
CATEGORIZED BY MECHANISM OF ACTION Synthetics / Allografts Osteoconductive adj: The facilitation of blood vessel incursion and new bone formation into a defined passive trellis structure PMD 017283 – 1. 0 Demineralized Bone BMPs Osteoinductive adj: New bone realized through the active recruitment of host mesenchymal stem cells from the surrounding tissue, which differentiate into bone forming osteoblasts. This process is facilitated by the presence of growth factors within the graft, principally bone morphogenetic proteins (BMPs)

COMMERCIAL DBM PRODUCTS • There are currently not less than • 35 different products • from at least 15 companies PMD 017283 – 1. 0

FREQUENTLY ASKED QUESTIONS ü Are they all the same? ü What makes them different? ü How do I choose a good DBM? PMD 017283 – 1. 0

HOW TO MEASURE DBM PERFORMANCE Osteoinduction: New bone realized through the active recruitment of host mesenchymal stem cells from the surrounding tissue, which differentiate into bone forming osteoblasts. This process is facilitated by the presence of growth factors within the graft, principally bone morphogenetic proteins (BMPs) = ü Accurate, reproducible ü Standard, validated method ü Indicative of performance PMD 017283 – 1. 0
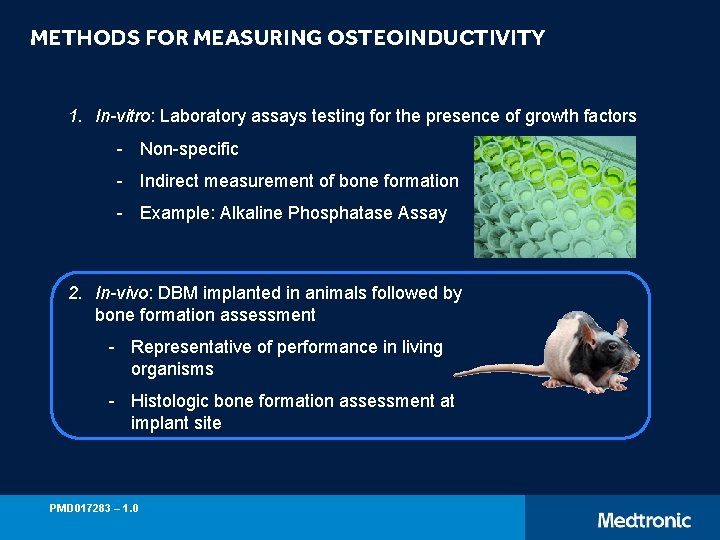
METHODS FOR MEASURING OSTEOINDUCTIVITY 1. In-vitro: Laboratory assays testing for the presence of growth factors - Non-specific - Indirect measurement of bone formation - Example: Alkaline Phosphatase Assay 2. In-vivo: DBM implanted in animals followed by bone formation assessment - Representative of performance in living organisms - Histologic bone formation assessment at implant site PMD 017283 – 1. 0

IN VIVO RAT OSTEOINDUCTIVITY (OI) ASSAY Edwards, J. T. , et al CORR 357: 219 -228, 1998 ü Based upon classic Urist model ü Athymic (nude) rat (no thymus gland) ü 28 day implantation period ü Hindlimb intermuscular implantation ü Bone formation histological assessment *Animal Data may not be indicative of clinical performance. PMD 017283 – 1. 0
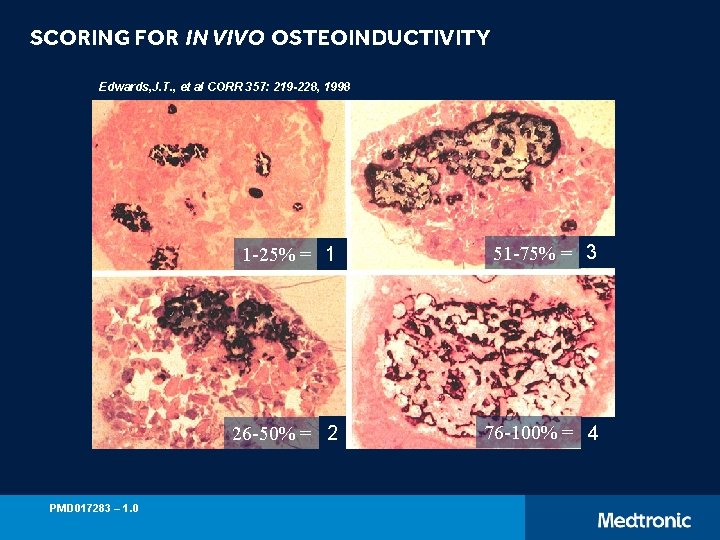
SCORING FOR IN VIVO OSTEOINDUCTIVITY Edwards, J. T. , et al CORR 357: 219 -228, 1998 PMD 017283 – 1. 0 1 -25% = 1 51 -75% = 3 26 -50% = 2 76 -100% = 4
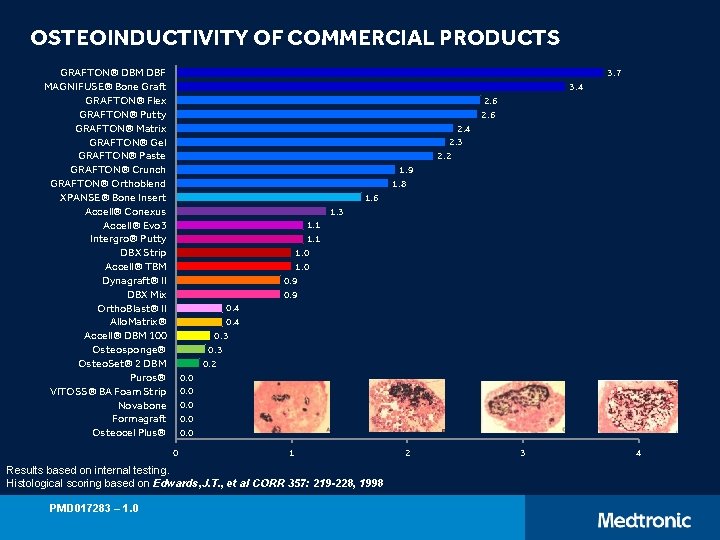
OSTEOINDUCTIVITY OF COMMERCIAL PRODUCTS GRAFTON® DBM DBF MAGNIFUSE® Bone Graft GRAFTON® Flex GRAFTON® Putty GRAFTON® Matrix GRAFTON® Gel GRAFTON® Paste GRAFTON® Crunch GRAFTON® Orthoblend XPANSE® Bone Insert Accell® Conexus Accell® Evo 3 Intergro® Putty DBX Strip Accell® TBM Dynagraft® II DBX Mix Ortho. Blast® II Allo. Matrix® Accell® DBM 100 Osteosponge® Osteo. Set® 2 DBM Puros® VITOSS® BA Foam Strip Novabone Formagraft Osteocel Plus® 3. 7 3. 4 2. 6 2. 4 2. 3 2. 2 1. 9 1. 8 1. 6 1. 3 1. 1 1. 0 0. 9 0. 4 0. 3 0. 2 0. 0 0 1 Results based on internal testing. Histological scoring based on Edwards, J. T. , et al CORR 357: 219 -228, 1998 PMD 017283 – 1. 0 2 3 4

OSTEOINDUCTIVITY OF COMMERCIAL PRODUCTS Animal testing is not necessarily indicative of human clinical outcome. Histological scoring table based on images from Edwards, JT et al. Clin Orthop Relat Res. 1998; 357: 219 -228 Accell®, Dynagraft®, and Ortho. Blast® are registered trademarks of Integra Life. Sciences Corporation. Allomatrix® and Osteo. Set® are registered trademarks of Wright Medical Technology. Inter. Gro® is a registered trademark of Biomet, Inc. Osteocel® is a registered trademark of Nuvasive. Puros® is a registered trademark of Zimmer, Inc. Data on File from Medtronic internal testing: GRAFTON® Putty, ongoing final product testing (2006 -2014); ACCELL® Connexus, three manufacturing lots tested on 2005; ACCELL® EVO 3, three manufacturing lots tested on 2010/2014; INTERGRO® Putty, one manufacturing lot tested on 2004; ACCELL® TBM, two manufacturing lots tested on 2010; DBX® Strip, three manufacturing lots tested on 2010; DBX® Mix, two manufacturing lots tested on 2010; DYNAGRAFT® II, one manufacturing lot tested on 2003; ALLOMATRIX®, five manufacturing lots tested on 1999/2005; ORTHOBLAST® II, two manufacturing lots tested on 2003/2005; ACCELL® 100, two manufacturing lots tested on 2003/2005; OSTEOSET® 2 DBM, two manufacturing lots tested on 2008; OSTEOCEL® Plus, three manufacturing lots tested on 2011; PUROS®, one manufacturing lot tested on 2010. PMD 017283 – 1. 0 12 Presentation Title (Edit on Slide Master) | June 1, 2015 | Confidential, for Internal Use Only
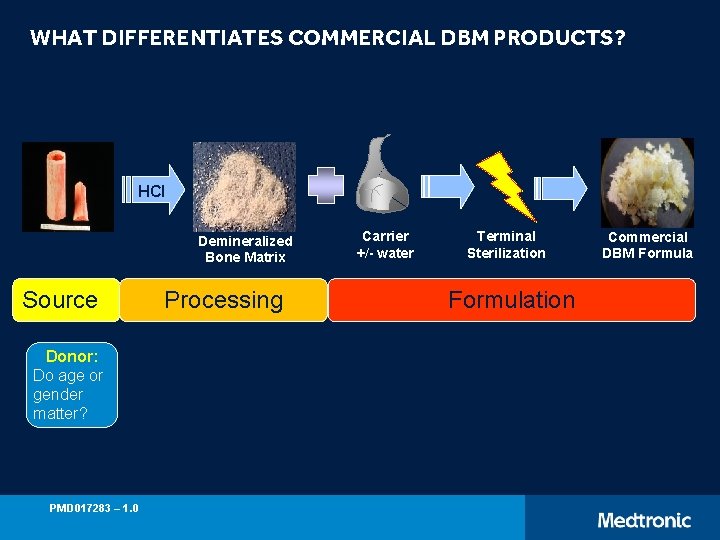
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Source Donor: Do age or gender matter? PMD 017283 – 1. 0 Processing Carrier +/- water Terminal Sterilization Formulation Commercial DBM Formula
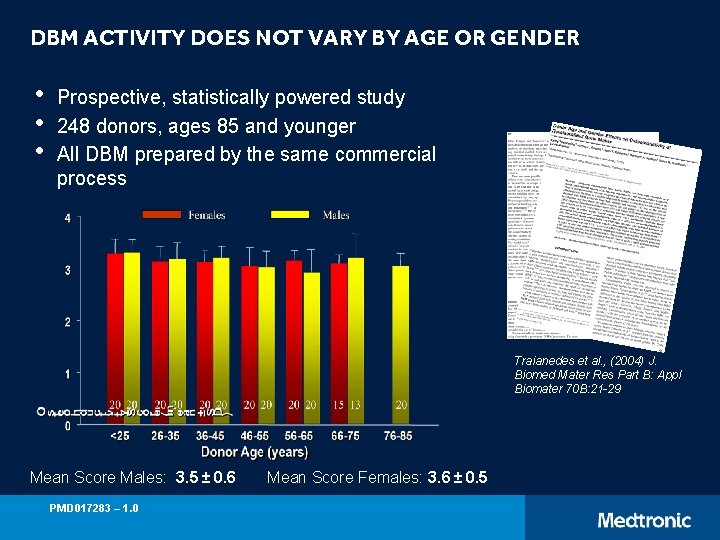
DBM ACTIVITY DOES NOT VARY BY AGE OR GENDER • • • Prospective, statistically powered study 248 donors, ages 85 and younger All DBM prepared by the same commercial process Traianedes et al. , (2004) J. Biomed Mater Res Part B: Appl Biomater 70 B: 21 -29 Mean Score Males: 3. 5 ± 0. 6 PMD 017283 – 1. 0 Mean Score Females: 3. 6 ± 0. 5
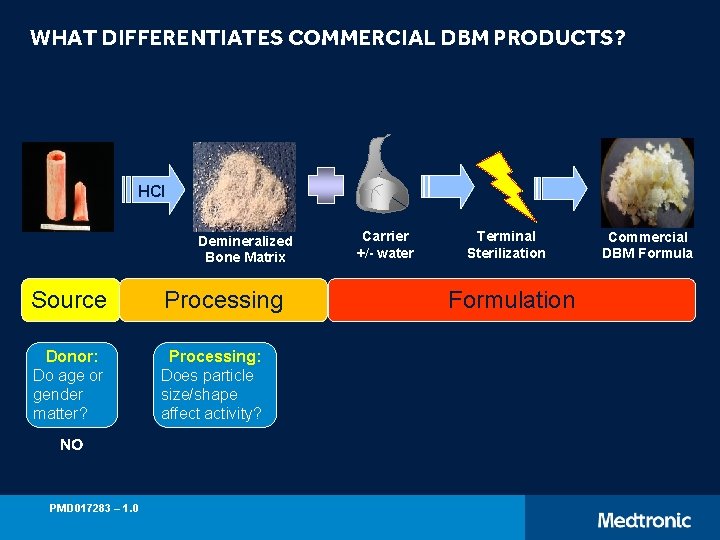
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Source Processing Donor: Do age or gender matter? Processing: Does particle size/shape affect activity? NO PMD 017283 – 1. 0 Carrier +/- water Terminal Sterilization Formulation Commercial DBM Formula

DBM PRODUCTS: PARTICLES VS FIBERS Grind Mill PMD 017283 – 1. 0
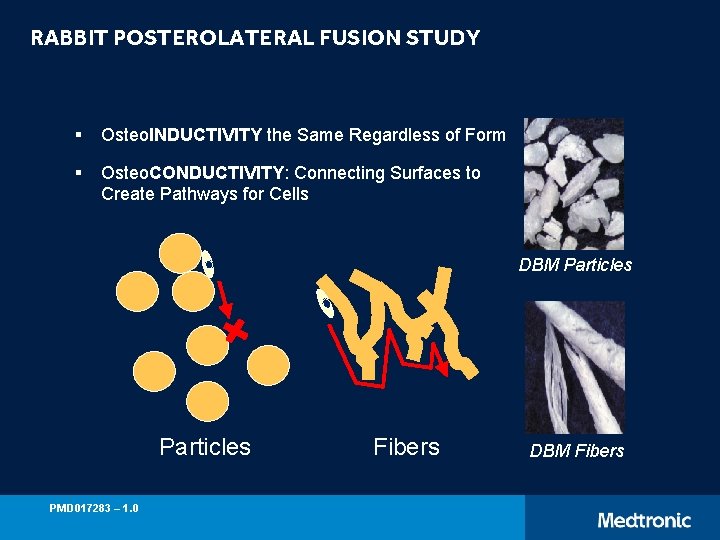
RABBIT POSTEROLATERAL FUSION STUDY § Osteo. INDUCTIVITY the Same Regardless of Form § Osteo. CONDUCTIVITY: Connecting Surfaces to Create Pathways for Cells DBM Particles PMD 017283 – 1. 0 Fibers DBM Fibers

ROLE OF FIBERS IN DBM ACTIVITY Martin et al; Spine 24(7), 1999 pp: 637 -645* ü Rabbit Posterolateral fusion of the spine (L 5 -L 6) ü Study Groups: § Grafton Gel (Particles) § Grafton Putty (Fibers) ü Eight week implantation period ü Fusion and new bone formation assessed by: § Manual Palpation § X-rays § Histology Gold standard of comparison: Autograft ~ 70% Study Group Gel (Particles) DBM (100%) 7/12 (58%) 10/12 (83%) 0/12 (0%) 4/12 (33%) Inactivated DBM (no osteoinductivity) Fibers More Osteoconductive than Particles Residual fusion observed attributed to Osteoconduction *Animal Data may not be indicative of clinical performance. PMD 017283 – 1. 0 Putty (Fibers)
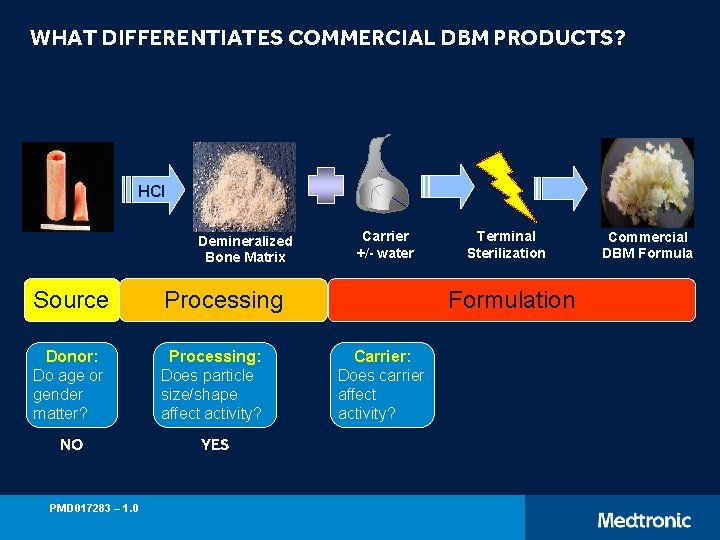
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Source Processing Donor: Do age or gender matter? Processing: Does particle size/shape affect activity? NO YES PMD 017283 – 1. 0 Carrier +/- water Terminal Sterilization Formulation Carrier: Does carrier affect activity? Commercial DBM Formula
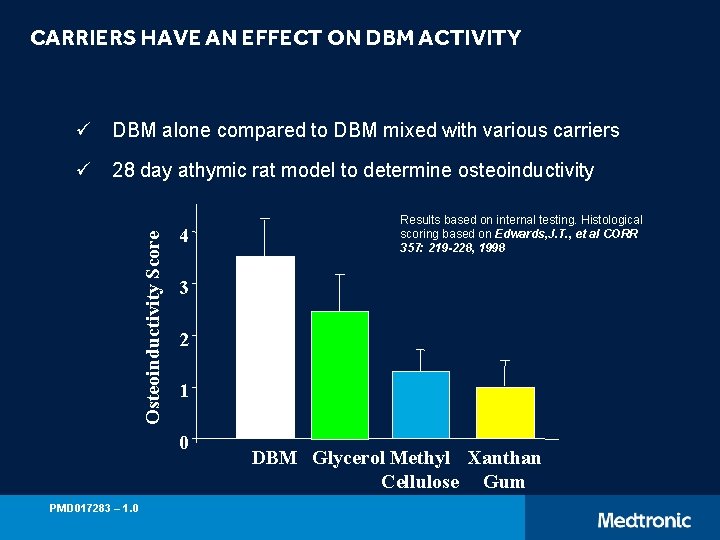
CARRIERS HAVE AN EFFECT ON DBM ACTIVITY ü DBM alone compared to DBM mixed with various carriers Osteoinductivity Score ü 28 day athymic rat model to determine osteoinductivity 4 3 2 1 0 PMD 017283 – 1. 0 Results based on internal testing. Histological scoring based on Edwards, J. T. , et al CORR 357: 219 -228, 1998 DBM Glycerol Methyl Xanthan Cellulose Gum
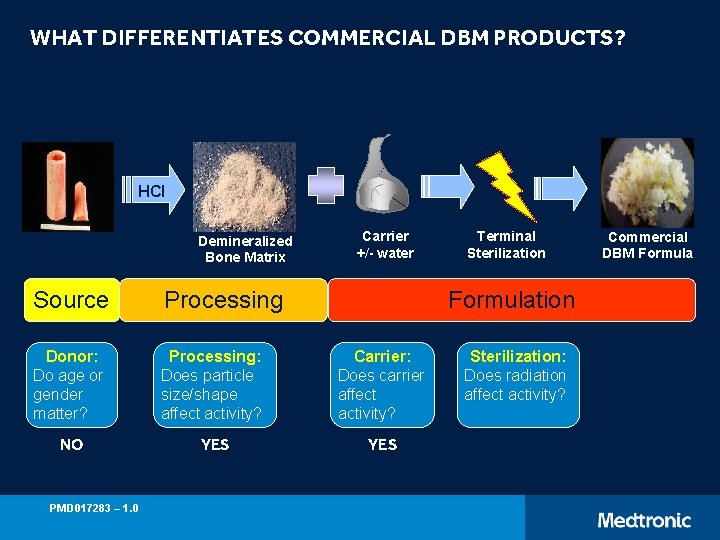
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Carrier +/- water Source Processing Donor: Do age or gender matter? Processing: Does particle size/shape affect activity? Carrier: Does carrier affect activity? NO YES PMD 017283 – 1. 0 Terminal Sterilization Formulation Sterilization: Does radiation affect activity? Commercial DBM Formula
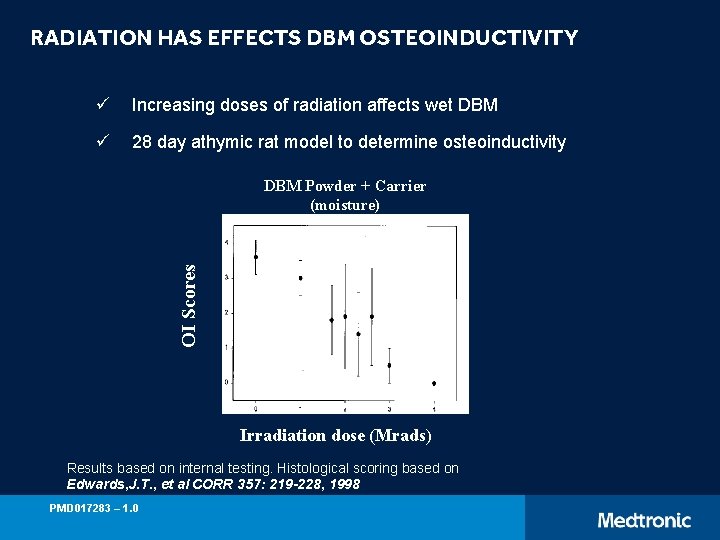
RADIATION HAS EFFECTS DBM OSTEOINDUCTIVITY ü Increasing doses of radiation affects wet DBM ü 28 day athymic rat model to determine osteoinductivity OI Scores DBM Powder + Carrier (moisture) Irradiation dose (Mrads) Results based on internal testing. Histological scoring based on Edwards, J. T. , et al CORR 357: 219 -228, 1998 PMD 017283 – 1. 0
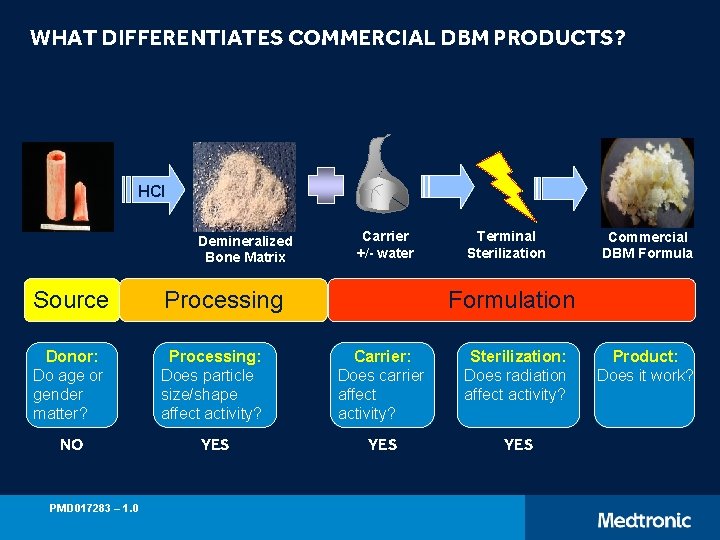
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Carrier +/- water Terminal Sterilization Source Processing Donor: Do age or gender matter? Processing: Does particle size/shape affect activity? Carrier: Does carrier affect activity? Sterilization: Does radiation affect activity? NO YES YES PMD 017283 – 1. 0 Commercial DBM Formulation Product: Does it work?

WHEN AND HOW ARE DBM PRODUCTS TESTED FOR OI? HCl Demineralized Bone Matrix Source Processing Carrier +/- water Terminal Sterilization Commercial DBM Formulation ? ? In vitro In vivo PMD 017283 – 1. 0

WHEN AND HOW ARE DBM PRODUCTS TESTED FOR OI? PMD 017283 – 1. 0
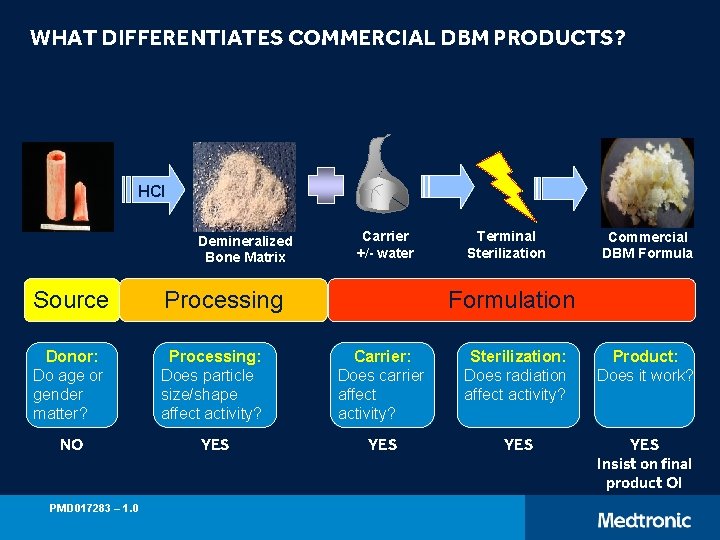
WHAT DIFFERENTIATES COMMERCIAL DBM PRODUCTS? HCl Demineralized Bone Matrix Carrier +/- water Terminal Sterilization Commercial DBM Formula Source Processing Donor: Do age or gender matter? Processing: Does particle size/shape affect activity? Carrier: Does carrier affect activity? Sterilization: Does radiation affect activity? Product: Does it work? NO YES YES Insist on final product OI PMD 017283 – 1. 0 Formulation

MANY CLINICAL STUDIES PUBLISHED PMD 017283 – 1. 0

DBM PERFORMANCE SUMMARY ü Are they all the same? • No, final DBM product performance varies widely ü What makes them different? • Differences in processing, carrier, and sterilization all effect osteoinductivity of DBM products ü How do I choose a good DBM? • Insist on final product OI data from your rep • Understand your product and how it’s made • Make sure there is peer- reviewed clinical evidence PMD 017283 – 1. 0

Thank You

Important Safety Information GRAFTON® DBM Family Indications GRAFTON® DBM and GRAFTON PLUS® DBM are intended for use as a bone graft extender, bone graft substitute, and bone void filler in bony voids or gaps of the skeletal system (i. e. , spine, pelvis, and extremities) not intrinsic to the stability of the bony structure. The voids or gaps may be surgically created defects or defects created by traumatic injury to the bone. GRAFTON® DBM and GRAFTON® PLUS DBM may be used alone in a manner comparable to autogenous bone chips or allograft bone particulate (demineralized freeze dried bone), or they may be mixed with either allograft or autograft bone or bone marrow as a bone graft extender. GRAFTON® DBM and GRAFTON® PLUS DBM are indicated only for bony voids or gaps that are not intrinsic to the stability of the bony structure. GRAFTON® DBM and GRAFTON® PLUS DBM are absorbed/remodeled and replaced by host bone during the healing process. MAGNIFUSE® Bone Graft Indications MAGNIFUSE® Bone Graft is intended for use as a bone graft substitute in bony voids or gaps of the skeletal system (i. e. , spine, pelvis and extremities) not intrinsic to the stability of the bony structure. The voids or gaps may be surgically created defects or defects created by traumatic injury to the bone. MAGNIFUSE® Bone Graft may be used in a manner comparable to autogenous bone or allograft bone. MAGNIFUSE® Bone Graft may be mixed with fluid such as bone marrow aspirate, blood, sterile water, or sterile saline in order to adjust the consistency and handling characteristics of the bone graft material. MAGNIFUSE® Bone Graft is resorbed/remodeled and replaced by host bone during the healing process. Risks of GRAFTON® DBM Family and MAGNIFUSE® Bone Graft The following are contraindications for the use of MAGNIFUSE® Bone Graft, GRAFTON® DBM and GRAFTON PLUS® DBM: • The presence of infection at the transplantation site. • Treatment of spinal insufficiency fractures.

Important Safety Information continued PROGENIX® DBM Family Indications PROGENIX® Putty is intended for use as a bone graft substitute in bony voids or gaps of the skeletal system not intrinsic to the stability of the bony structure (i. e. spine, pelvis and extremities). The voids or gaps may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. PROGENIX® Putty provides a bone void filler that is resorbed/remodeled and is replaced by host bone during the healing process. When used in the extremities or pelvis, the device is used by itself. When used in the spine, the device must be mixed with autograft bone and used as a bone graft extender. PROGENIX® Plus is intended for use as a bone graft substitute in bony voids or gaps of the skeletal system not intrinsic to the stability of the bony structure (i. e. spine, pelvis and extremities). The voids or gaps may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. PROGENIX® Plus provides a bone void filler that is resorbed/remodeled and is replaced by host bone during the healing process. The device may either be use alone or mixed with autograft bone and used as a bone graft extender. Risks of PROGENIX® DBM Family PROGENIX® Putty and PROGENIX® Plus are contraindicated, and should not be used, in patients who have known allergies to bovine collagen, sodium alginate or polymyxim and/or bacitracin antibiotics. These products do not possess sufficient mechanical strength to support the reduction of a defect site prior to soft and hard tissue ingrowth. These products are contraindicated where stabilization of the defect is not possible. XPANSE® Bone Insert Information XPANSE® Bone Insert can be used in orthopedic or reconstructive bone grafting procedures. The product can also be used in bone grafting procedures in combination with autologous bone or other forms of allograft bone, or alone as a bone graft. The presence of infection at the transplantation site is a contraindication for the use of this allograft.

Important Safety Information continued MASTERGRAFT® Family of Products MASTERGRAFT® Granules is indicated only for bony voids or gaps that are not intrinsic to the stability of the bony structure. Additionally, MASTERGRAFT® Granules can be used with autograft as a bone graft extender. MASTERGRAFT® Granules is to be gently packed into bony voids or gaps of the skeletal system (e. g. , the posterolateral spine, pelvis, ilium and/or extremities). These defects may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. MASTERGRAFT® Granules provides a bone void filler that resorbs and is replaced with bone during the healing process. MASTERGRAFT® Putty combined with either autogenous bone marrow, and/or sterile water, and/or autograft is indicated as a bone void filler for bony voids or gaps that are not intrinsic to the stability of the bony structure. Additionally, MASTERGRAFT® Putty can be used with autograft as a bone graft extender. MASTERGRAFT® Putty is to be gently packed into bony voids or gaps of the skeletal system (e. g. , the posterolateral spine, pelvis, ilium, and/or extremities). These defects may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. MASTERGRAFT® Putty resorbs and is replaced with bone during the healing process. MASTERGRAFT® Strip and MASTERGRAFT® Matrix EXT are to be combined with autogenous bone marrow and are indicated for bony voids or gaps that are not intrinsic to the stability of the bony structure and can be used as bone graft extenders. These devices are to be gently packed into bony voids or gaps of the skeletal system (i. e. , the posterolateral spine, pelvis, ilium, and/or extremities). These defects may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. These devices resorb and are replaced with bone during the healing process.

Important Safety Information continued Risks of MASTERGRAFT® Family of Products MASTERGRAFT® is not intended to provide structural support during the healing process. Supplemental fixation should be used if structural support is required. Therefore, MASTERGRAFT® is contraindicated where the device is intended as structural support in the skeletal system. Conditions representing relative contraindications include: • Severe neurological or vascular disease. • Uncontrolled diabetes. • Hypercalcemia. • Pregnancy. • Where stabilization of fracture is not possible. • Segmental defects without supplemental fixation. • Where there is significant vascular impairment proximal to the graft site. • When there are systemic and/or metabolic disorders that affect the bone or wound healing. • Any patient unwilling to follow postoperative instructions. • Any case not described in the indications. MASTERGRAFT® should not be used in patients with a known history of hypersensitivity to bovine derived materials.

Warnings and Precautions – INFUSE® BONE GRAFT This device has not been tested in pregnant women to determine if there is any effect on a developing fetus. This device has also not been studied in nursing mothers. When tested in female rabbits that received the rh. BMP-2, a component of the device, developed an immune response and later became pregnant, the following was seen: » The antibodies developed by the mother were able to reach the developing rabbit fetus. The effect of these antibodies on the developing rabbit fetus is not currently known. » Some bone formation abnormalities were observed in a small number of the rabbit fetuses tested. It is not known if these changes would disappear as the rabbit fetus continued to develop or at some time after birth. This device should not be used immediately prior to or during pregnancy. Women of child-bearing potential should be advised not to get pregnant for one year following treatment with the device. Women of child bearing potential should be warned of potential risk to a fetus and should discuss other possible orthopedic treatments with their surgeon. BMP-2 plays a critical role during fetal development in humans and other animals. It is not known whether a pregnant woman, previously exposed to BMP-2 by implantation with the device, might develop a second immune response to BMP-2 from the developing fetus with adverse effects for the woman or baby. In a rabbit pregnancy study to investigate this issue, no increase in anti-BMP-2 antibodies was observed.

Brief Summary – INFUSE® BONE GRAFT BRIEF SUMMARY OF INDICATIONS, CONTRAINDICATIONS, WARNINGS, AND PRECAUTION FOR INFUSE® BONE GRAFT FOR CERTAIN ORAL MAXILLOFACIAL AND DENTAL REGENERATIVE USES INFUSE® Bone Graft is indicated as an alternative to autogenous bone graft for sinus augmentations, and for localized alveolar ridge augmentations for defects associated with extraction sockets. The INFUSE® Bone Graft consists of two components – recombinant human Bone Morphogenetic Protein-2 (rh. BMP-2) placed on an absorbable collagen sponge (ACS). These components must be used as a system for the prescribed indication. The bone morphogenetic protein solution component must not be used without the carrier/scaffold component or with a carrier/scaffold component different from the one described in the package insert. INFUSE® Bone Graft is contraindicated for patients with a known hypersensitivity to recombinant human Bone Morphogenetic Protein-2, bovine Type 1 collagen or to other components of the formulation and should not be used in the vicinity of a resected or extant tumor, in patients with any active malignancy or patients undergoing treatment for a malignancy, in pregnant women, or patients with an active infection at the operative site. There are no adequate and well-controlled studies in human pregnant women. In an experimental rabbit study, rh. BMP-2 has been shown to elicit antibodies that are capable of crossing the placenta. Women of child bearing potential should be warned by their surgeon of potential risk to a fetus and informed of other possible dental treatments. The safety and effectiveness of this device has not been established in nursing mothers. Women of child-bearing potential should be advised to not become pregnant for one year following treatment with this device. INFUSE® Bone Graft has not been studied in patients who are skeletally immature (<18 years of age or no radiographic evidence of epiphyseal closure). Please see the package insert for the complete list of indications, warnings, precautions, adverse events, clinical results, and other important medical information.

Brief Summary – INFUSE® BONE GRAFT continued BRIEF SUMMARY OF INDICATIONS, CONTRAINDICATIONS, AND WARNINGS FOR: INFUSE® BONE GRAFT/LT-CAGE® LUMBAR TAPERED FUSION DEVICE INFUSE® BONE GRAFT/INTER FIX™ THREADED FUSION DEVICE INFUSE® BONE GRAFT/INTER FIX™ RP THREADED FUSION DEVICE The INFUSE® Bone Graft/Medtronic Titanium Threaded Interbody Fusion Device is indicated for spinal fusion procedures in skeletally mature patients with degenerative disc disease (DDD) at one level from L 2 – S 1, who may also have up to Grade 1 spondylolisthesis or Grade 1 retrolisthesis at the involved level. The INFUSE® Bone Graft/LT-CAGE® Lumbar Tapered Fusion Device is to be implanted via an anterior open or an anterior laparoscopic approach. INFUSE® Bone Graft with either the INTER FIX™ or INTER FIX™ RP Threaded Fusion Device is to be implanted via an anterior open approach. The INFUSE® Bone Graft/Medtronic Titanium Threaded Interbody Fusion Device consists of two components containing three parts– a metallic spinal fusion cage, a recombinant human bone morphogenetic protein and a carrier/scaffold for the bone morphogenetic protein and resulting bone. These components must be used as a system for the prescribed indication described above. The bone morphogenetic protein solution component must not be used without the carrier/scaffold component or with a carrier/scaffold component different from the one described in this document. The INFUSE® Bone Graft component must not be used without the Medtronic Titanium Threaded Interbody Fusion Device component. NOTE: The INTER FIX™ Threaded Fusion Device and the INTER FIX™ RP Threaded Fusion Device may be used together to treat a spinal level. LT-CAGE® Lumbar Tapered Fusion Device implants are not to be used in conjunction with either the INTER FIX™ or INTER FIX™ RP implants to treat a spinal level. The INFUSE® Bone Graft/Medtronic Titanium Threaded Interbody Fusion Device is contraindicated for patients with a known hypersensitivity to recombinant human Bone Morphogenetic Protein-2, bovine Type I collagen or to other components of the formulation and should not be used in the vicinity of a resected or extant tumor; in patients with any active malignancy or patients undergoing treatment for a malignancy; in patients who are skeletally immature; in pregnant women; or in patients with an active infection at the operative site or with an allergy to titanium or titanium alloy.

Brief Summary – INFUSE® BONE GRAFT continued There are no adequate and well-controlled studies in human pregnant women. In an experimental rabbit study, rh. BMP-2 has been shown to elicit antibodies that are capable of crossing the placenta. Women of child bearing potential should be warned by their surgeon of potential risk to a fetus and informed of other possible orthopedic treatments. The safety and effectiveness of this device has not been established in nursing mothers. Women of child-bearing potential should be advised to not become pregnant for one year following treatment with this device. Please see the package insert for the complete list of indications, warnings, precautions, adverse events, clinical results, definition of DDD, and other important medical information. The package insert also matches the sizes of those sized devices that are indicated for use with the appropriate INFUSE® Bone Graft kit. CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician with appropriate training or experience.

Brief Summary – INFUSE® BONE GRAFT continued BRIEF SUMMARY OF INDICATIONS, CONTRAINDICATIONS, AND WARNINGS FOR: INFUSE® BONE GRAFT INFUSE® Bone Graft is indicated for treating acute, open tibial shaft fractures that have been stabilized with IM nail fixation after appropriate wound management. INFUSE® Bone Graft must be applied within 14 days after the initial fracture. Prospective patients should be skeletally mature. INFUSE® Bone Graft consists of two components – recombinant human Bone Morphogenetic Protein-2 solution and a carrier/scaffold for the bone morphogenetic protein solution and resulting bone. These components must be used as a system. The bone morphogenetic protein solution component must not be used without the carrier/scaffold component or with a carrier/scaffold component different from the one described in this document. INFUSE® Bone Graft is contraindicated for patients with a known hypersensitivity to recombinant human Bone Morphogenetic Protein-2, bovine Type 1 collagen or to other components of the formulation and should not be used in the vicinity of a resected or extant tumor, in patients with an active malignancy or patients undergoing treatment for a malignancy. INFUSE® Bone Graft should also not be used in patients who are skeletally immature, in patients with an inadequate neurovascular status, in patients with compartment syndrome of the affected limb, in pregnant women, or in patients with an active infection at the operative site. There are no adequate and well controlled studies in human pregnant women. In an experimental rabbit study, rh. BMP-2 has been shown to elicit antibodies that are capable of crossing the placenta. Women of child bearing potential should be warned by their surgeon of potential risk to a fetus and informed of other possible orthopedic treatments. The safety and effectiveness of this device has not been established in nursing mothers. Women of child-bearing potential should be advised to not become pregnant for one year following treatment with this device. Please see the package insert for the complete list of indications, warnings, precautions, adverse events, clinical results, and other important medical information. CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician with appropriate training or experience.
- Slides: 38