dBLOCK ELEMENTS No of lectures 12 Term 1

d-BLOCK ELEMENTS No. of lectures – 12 Term - 1 smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 1 1
INTRODUCTION OF D-BLOCK ELEMENTS smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 1 2
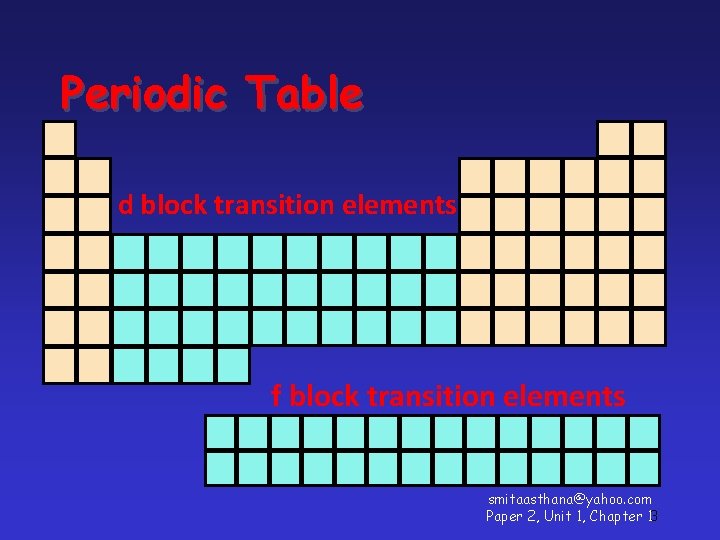
Periodic Table d block transition elements f block transition elements smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 13

d-Block Transition Elements IIIB IVB VB Sc Ti V Y La VIIIB VIIB IB IIB Cr Mn Fe Co Ni Cu Zn Zr Nb Mo Tc Ru Rh Pd Ag Cd Hf Ta Re Os Ir Pt Hg W Au Most have partially occupied d sub-shells in common oxidation states smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 14

Why are they called d-block elements? Their last electron enters the d-orbital. smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 15

Most d-block elements are also called transition metals. This is because they exhibit characteristics that ranges from s -block to p – block. Zinc group and Scandium group are NOT considered as transition metals, are called Non-typical Transition elements. smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 16

What is a transition metal? • For this reason, a transition metal is defined as being an element which forms at least one ion with a partially filled d orbital(s). smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 17

The d block: • The d block consists of three horizontal series in periods 4, 5 & 6 – 10 elements in each series – Chemistry is “different” from other elements – Differences within a group in the d block are less sharp than in s & p block • Similarities across a period are greater smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 18
![Electronic Arrangement Element Z 3 d 4 s Sc 21 [Ar] Ti 22 [Ar] Electronic Arrangement Element Z 3 d 4 s Sc 21 [Ar] Ti 22 [Ar]](http://slidetodoc.com/presentation_image_h2/cd7dd04100a90b6bb9a419e482fa541a/image-9.jpg)
Electronic Arrangement Element Z 3 d 4 s Sc 21 [Ar] Ti 22 [Ar] V 23 [Ar] Cr 24 [Ar] Mn 25 [Ar] Fe 26 [Ar] Co 27 [Ar] Ni 28 [Ar] Cu 29 [Ar] Zn 30 [Ar] smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 19

Electronic Configuration • Across the 1 st row of the d block (Sc to Zn) each element – has 1 more electron and 1 more proton – Each “additional” electron enters the 3 d sub-shell – The core configuration for all the 1 st series of transition elements is that of Ar • 1 s 22 p 63 s 23 p 6 smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 10 1

Chromium and Copper • At Cr – Orbital energies such that putting one e- into each 3 d and 4 s orbital gives lower energy than having 2 e- in the 4 s orbital • At Cu – Putting 2 e- into the 4 s orbital would give a higher energy than filling the 3 d orbitals smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 11 1
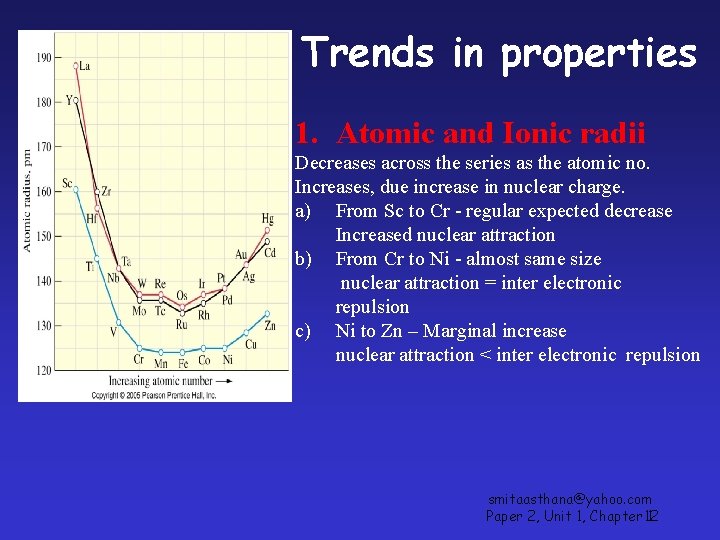
Trends in properties 1. Atomic and Ionic radii Decreases across the series as the atomic no. Increases, due increase in nuclear charge. a) From Sc to Cr - regular expected decrease Increased nuclear attraction b) From Cr to Ni - almost same size nuclear attraction = inter electronic repulsion c) Ni to Zn – Marginal increase nuclear attraction < inter electronic repulsion smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 12 1

2. Ionization Potential smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 13 1

3. Variable Oxidation States ØD-block elements exhibit variable oxidation states. ØThis means that they can form two or more different types of cations. Ø Examples: ØIron can form both Fe²⁺ and Fe ³⁺ ØManganese can Mn²⁺, Mn³⁺, Mn⁴⁺, Mn⁶⁺ and Mn⁷⁺ smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 14 1

Oxidation States of TM’s Sc Ti V Cr Mn Fe Co Ni Cu Zn +1 +2 +2 +3 +3 +4 +4 +5 +5 +5 +6 +6 +7 +2 +2 +6 smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 15 1

Oxidation States of TM’s 1. No of OS’s shown by an element increases from Sc to Mn – In each of these elements highest OS is equal to no. of 3 d and 4 s e- • After Mn decrease in no. of OS’s shown by an element – Highest OS shown becomes lower and less stable – Seems increasing nuclear charge binds 3 d emore strongly, hence harder to remove smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 16 1

Stability of OS’s • General trends 1. Higher OS’s become less stable relative to lower ones on moving from left to right across the series 2. Compounds containing TM’s in high OS’s tend to be oxidising agents e. g Mn. O 43. Compounds with TM’s in low OS’s are often reducing agents e. g V 2+ & Fe 2+ smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 17 1

Oxidation States of TM’s • In the following table – Most important OS’s in boxes – OS = +1 only important for Cu – – OS = +2, where 4 s e- lost shown by all except for Sc and Ti – OS = +3, shown by all except Zn smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 18 1

Oxidation States of TM’s • Nature of bonds – Ionic and covalent – Lower OS’s found in ionic compounds • E. g. compounds containing Cr 3+, Mn 2+, Fe 3+, Cu 2+ ions – TM’s in higher OS’s usually covalently bound to electronegative element such as O or F • E. g VO 3 -, vanadate(V) ion; Mn. O 4 -, manganate(VII) ion • Simple ions with high OS’s such as V 5+ & Mn 7+ are not formed smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 19 1

Oxidising and reducing nature – lower oxidation states are highly reducing • E. g. V 2+(aq) & Cr 2+(aq) strong reducing agents – higher oxidation states are oxidising in nature • E. g. Co 3+ is a strong oxidising agent, • KMn. O 4 - OS +7, K 2 Cr 2 O 7 - OS +6 are oxidising agents Acidic and basic nature • Higher oxidation states are acidic in nature • Lower oxidation states become increasingly basic via amphoteric nature ü H 2 Cr. O 4 is strong acid – Cr OS +6 smitaasthana@yahoo. com üMn 2 O 3– basic (+3), Mn. O 2– amphoteric(+4), KMn. O 4 – 2, acidic(+7) Paper Unit 1, Chapter 20 1

Formation of coloured ions The compounds of the d-block metal ions are usually colored, except, those of d 0 and d 10 metal ions. The colors are due to: Electronic transitions of d-electrons within the d sub-shell. These are known as d→d transitions. When light passes through these compounds, electrons from a lower energy d-orbital absorb a photon of energy and are promoted to higher energy d-orbitals. The energy absorbed is equivalent to the energy difference between the two sets of orbitals. Electron while returning from the excited state gives away the energy which falls in visible range of spectrum and the substance appears coloured. Since light of a certain frequency is absorbed, the light that comes out looks coloured because it lacks some colour. The colour of the compound is the complementary of the one that was absorbed smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 21 1

Ferromagnetism and Paramagnetism EOS smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 22 1

Magnetic behaviour Electron is a micromagnet, moves 1. On its axis – Spin moment 2. In the orbitals – Orbital moment Total magnetic moment = Spin moment + Orbital moment µ(S + L) = √ 4 S (S+1) + L( L + 1) Orbital moment is negligible, µ eff. = √ n(n+2) B. M. smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 1 23
![Titanium [Ar] 4 s 23 d 2 Quite unreactive Zirconium [Kr] 5 s 2 Titanium [Ar] 4 s 23 d 2 Quite unreactive Zirconium [Kr] 5 s 2](http://slidetodoc.com/presentation_image_h2/cd7dd04100a90b6bb9a419e482fa541a/image-24.jpg)
Titanium [Ar] 4 s 23 d 2 Quite unreactive Zirconium [Kr] 5 s 2 4 d 2 Fairly inactive element Hafnium [Xe] 6 s 2 4 f 14 5 d 2 Not very reactive Rutherfordium [Rn] 7 s 2 5 f 14 6 d 2 Highly radioactive smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 24 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 25 1
![[Ar] 4 s 13 d 5 [Kr] 5 s 1 4 d 5 [Xe] [Ar] 4 s 13 d 5 [Kr] 5 s 1 4 d 5 [Xe]](http://slidetodoc.com/presentation_image_h2/cd7dd04100a90b6bb9a419e482fa541a/image-26.jpg)
[Ar] 4 s 13 d 5 [Kr] 5 s 1 4 d 5 [Xe] 6 s 1 4 f 14 5 d 5 smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 26 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 27 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 28 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 29 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 30 1

smitaasthana@yahoo. com Paper 2, Unit 1, Chapter 31 1
- Slides: 31