Day 1 Naming Compounds Writing Formulas SWBAT Write

Day 1 Naming Compounds Writing Formulas

SWBAT Write and name binary and tertinary ionic compounds l Write and name covalent compounds l

Systematic Naming There are too many compounds to remember the names of them all. l Compound is made of two or more elements. l Put together atoms. l Name should tell us how many and what type of atoms. l

Periodic Table More than a list of elements. l Put in columns because of similar properties. l Each column is called a group. l
Metals
Transition metals l The Group B elements

Dull l Brittle l Nonconductorsinsulators l Non-metals

Metalloids or Semimetals Properties of both l Semiconductors l

5 Types of Chemical Compounds • Binary ionic - metal ion – nonmetal ion • Ternary ionic - at least one ion is a polyatomic ion • Binary molecular - two nonmetals

Atoms and ions Atoms are electrically neutral. l Same number of protons and electrons. l Ions are atoms, or groups of atoms, with a charge. l Different numbers of protons and electrons. l Only electrons can move. l Gain or lose electrons. l

Anion A negative ion. l Has gained electrons. l Non metals can gain electrons. l Charge is written as a superscript on the right. l 1 F 2 O Has gained one electron Has gained two electrons

Cations Positive ions. l Formed by losing electrons. l More protons than electrons. l Metals form cations. l 1+ K Has lost one electron 2+ Ca Has lost two electrons

Charge in groups 1 A, 2 A and 3 A is the group number 1+ 2+ in 5 A, 6 A and 7 A is 3+ 3 - 2 - 1 the group number 8

Naming Cations l l Just Write the name K 1+ Potassium ion

Naming Anions l Anions are always the same. l Change the element ending to – ide 1 l F Fluorin

Naming Anions l Anions are always the same. l Change the element ending to – ide 1 l F Fluorine

Naming Anions l Anions are always the same l Change the element ending to – ide 1 l F Fluori

Naming Anions l Anions are always the same l Change the element ending to – ide 1 l F Fluor

Naming Anions l Anions are always the same l Change the element ending to – ide 1 l F Fluori

Naming Anions l Anions are always the same l Change the element ending to – ide 1 l F Fluorid

Naming Anions l Anions are always the same l Change the element ending to – ide 1 l F Fluoride

Naming Binary Ionic Compounds Binary Compounds - 2 elements. l Ionic - a cation and an anion. l The name is just the names of the ions. l Cation first anion second l Easy with Group A elements. l Na. Cl = Na+ Cl- = sodium chloride l Mg. Br 2 = Mg 2+ Br- = magnesium bromide l Na 2 S l

Naming Binary Ionic Compounds The problem comes with the transition metals. l Cation name includes the charge. l The compound must be neutral. l same number of + and – charges. l Use the negative charge to find the charge on the positive ion. l

Naming Binary Ionic Compounds Write the name of Cu. O l Need the charge of Cu l O is 2 l copper must be 2+ l Copper(II) oxide l Name Co. Cl 3 l Cl is 1 - and there are three of them = 3 l Co must be 3+ l Cobalt(III) chloride l

Naming Binary Ionic Compounds Write the names of the following l KCl l Na 3 N l Cr. N l Sc 3 P 2 l Pb. O 2 l Na 2 Se l

Polyatomic ions l Groups of atoms that stay together and have a charge. l Covalently bonded l You must memorize these.

A Few Polyatomic Rules: PA ions which contain Oxygen contain endings of -ite ( the least amount of Oxygen )and –ate ( the greater amount). l For ex. l Sulfite SO 32 - and Sulfate SO 42 l Nitrite NO 21 - and Nitrate NO 31 l

Polyatomic Rules cont. Hydrogen is often indicated by an ions name when present. l For ex. : Hydrogen sulfate HSO 41 l The prefixes mono- and di- are sometimes used. l For ex. : Dihydrogen phosphate H 2 PO 41 l The prefix thio- means, replace an Oxygen with a Sulfur. l
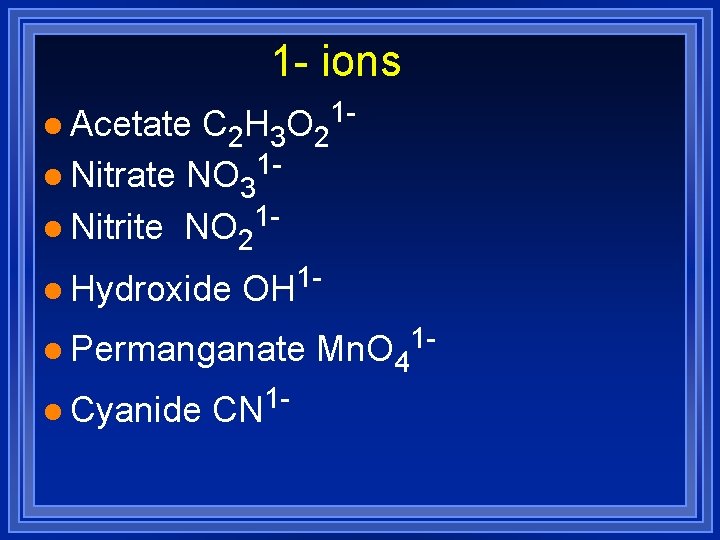
1 - ions C 2 H 3 O 21 l Nitrate NO 31 l Nitrite NO 21 l Acetate l Hydroxide 1 OH l Permanganate l Cyanide CN 1 - Mn. O 4 1 -

1 - ions Cl. O 41 l Chlorate Cl. O 31 l Chlorite Cl. O 21 l Perchlorate 1 l Hypochlorite

2 - ions l Sulfate 2 - SO 4 2 l Sulfite SO 3 2 l Carbonate CO 3 2 l Chromate Cr. O 4 l Dichromate Cr 2 O 72 l Silicate Si. O 32 -
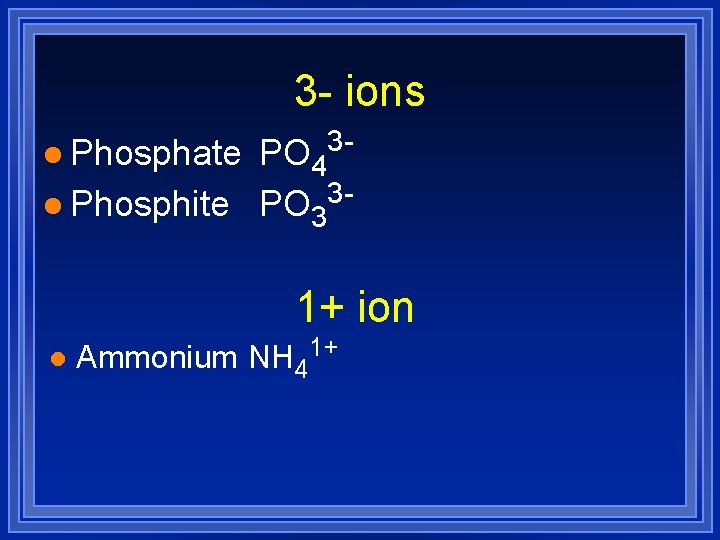
3 - ions l Phosphate 3 - PO 4 3 l Phosphite PO 3 1+ ion l Ammonium NH 41+

Adding Hydrogen to Polyatomics l Hydrogen ions are 1+ l Attach to other polyatomic ionschanges charge by one l Sulfate SO 42 l Hydrogen sulfate HSO 41 l Phosphate PO 43 l Hydrogen phosphate HPO 42 l Dihydrogen phosphate H 2 PO 41 -

Ternary Ionic Compounds Will have polyatomic ions l At least three elements (3 capital letters) l Still just name the ions l l Na. NO 3 l Ca. SO 4 l Cu. SO 3

Ternary Ionic Compounds l (NH 4)2 O l Fe(OH)3 l Li. CN l (NH 4)2 CO 3 l Ni. PO 4

Writing Formulas The charges have to add up to zero. l Get charges on pieces. l Cations from name or periodic table. l Anions from periodic table or polyatomic. l Balance the charges by adding subscripts. l Put polyatomics in parenthesis if there is more than one of them l

Writing Formulas Write the formula for calcium chloride. l Calcium is Ca 2+ l Chloride is Cl 1 l Ca 2+ Cl 1 - would have a 1+ charge. l Need another Cl 1 l Ca 2+ Cl 21 l

Crisscross l Switch the numerical value of the charges 3 32 2+ Ba N Ba 3 N 2 l Reduce ratio if possible

Write the formulas for these Lithium sulfide l tin (II) oxide l tin (IV) oxide l Copper (II) sulfate l Iron (III) phosphide l gallium nitrate l Iron (III) sulfide l ammonium sulfide l

Write the formulas for these Ammonium chloride l barium nitrate l
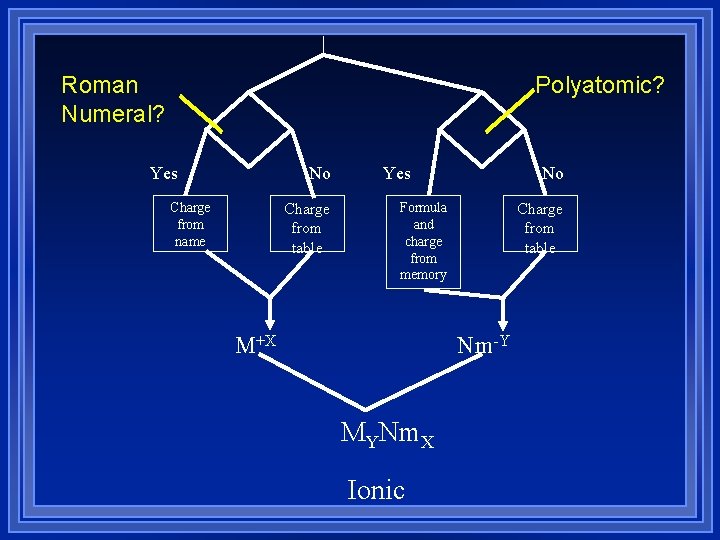
Polyatomic? Roman Numeral? Yes No Charge from name Charge from table Yes No Formula and charge from memory M+X Charge from table Nm-Y MYNm. X Ionic

Things to look for If cations have (), the number is their charge. Not how many. l If anions end in -ide they are probably off the periodic table (Monoatomic) l If anion ends in -ate or -ite it is polyatomic l The positive piece always gets written first l Hydrogen- it depends on where it’s at – If it is second, it’s a nonmetal -hydride l

Molecular Compounds Writing names and Formulas

Molecular compounds made of just nonmetals l smallest piece is a molecule l can’t be held together because of opposite charges. l can’t use charges to figure out how many of each atom l

Easier l Ionic compounds use charges to determine how many of each. – Have to figure out charges. – Have to figure out numbers. l Molecular compounds name tells you the number of atoms. l Uses prefixes to tell you the number

Prefixes 1 monol 2 dil 3 tril 4 tetral 5 pental 6 hexal 7 heptal 8 octal 9 nonal 10 decal

Naming l To write the name write two words Prefix name -ide Exception - we don’t write mono- if there is only one of the first element. l No ao oo double vowels when writing name, io, oi, and ai are okay. l

Name These N 2 O l NO 2 l Cl 2 O 7 l CBr 4 l CO 2 l Ba. Cl 2 l

Write formulas for these diphosphorus pentoxide l tetraiodine nonoxide l sulfur hexaflouride l nitrogen trioxide l Carbon tetrahydride l phosphorus trifluoride l aluminum chloride l diagram l
- Slides: 49