Dating Rocks Relative Absolute Relative Dating Nicolaus Stenonis

Dating Rocks Relative - Absolute

Relative Dating


Nicolaus Stenonis (1638 -1686).

Nicolaus Stenonis (1638 -1686). ". . . at the time when any given stratum was being formed, all the matter resting upon it was fluid, and, therefore, at the time when the lower stratum was being formed, none of the upper strata existed. ”

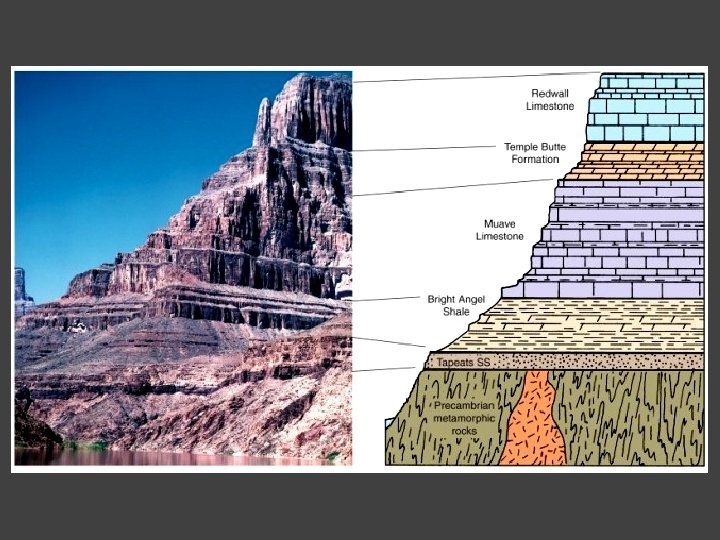
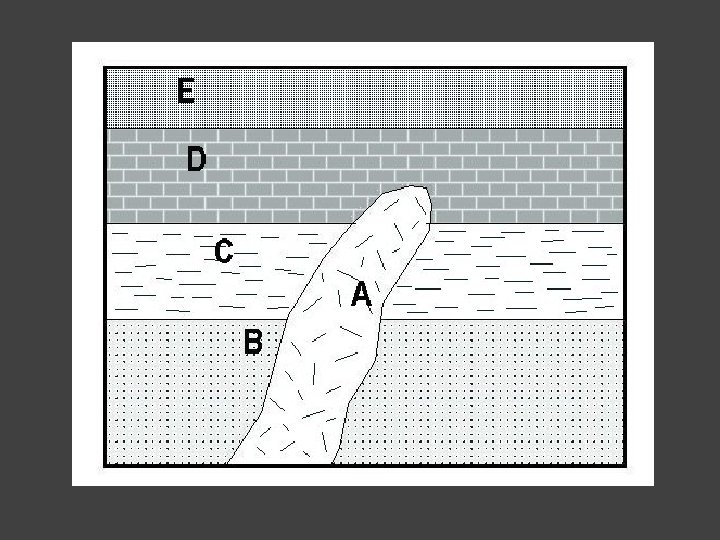
Steno’s Principle of Superposition (Prodromus, 1669) That is, in a sequence of strata, any stratum is younger than the sequence of strata on which it rests, and is older than the strata that rest upon it. ”

Nicolaus Stenonis’ Principle of superposition:

Nicolaus Stenonis’ Principle of superposition:



Quantitative Dating.

Quantitative dating: Structure of an atom.
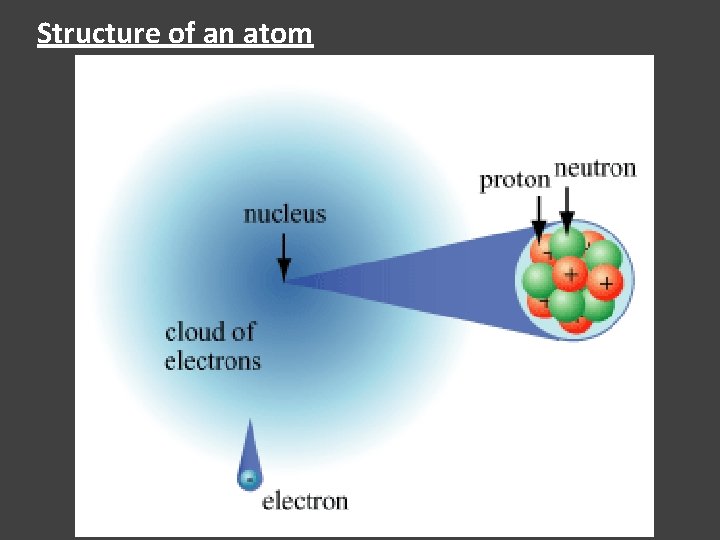
Structure of an atom

Inside the nucleus of an atom.

# protons of a nuclues define the elemental nature.
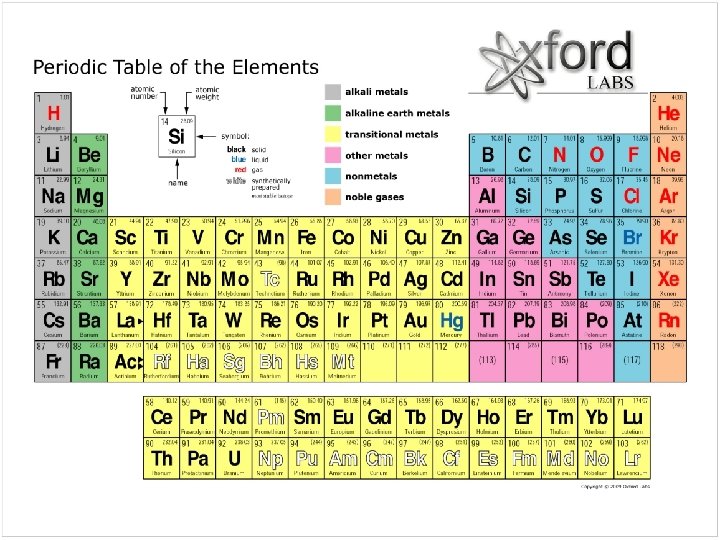

Elements are defined by - atom number ( = # protons), - Atom mass (# protons + # neutrons).

Isotopes of an element.
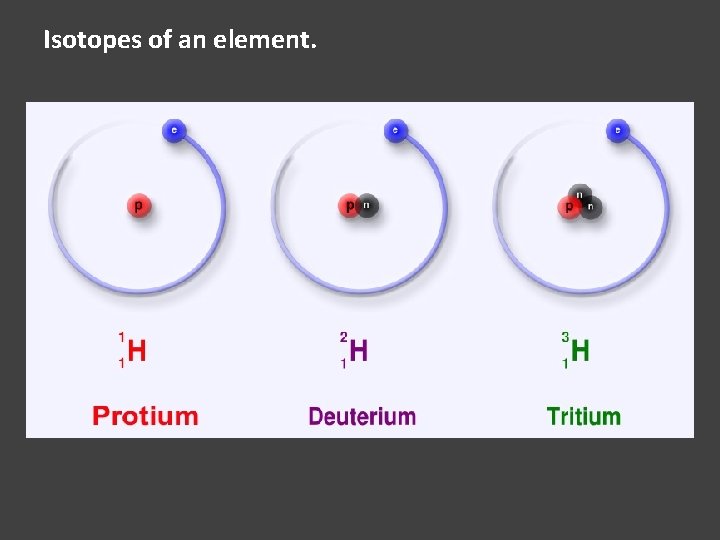
Isotopes of an element.

Isotopes of Carbon:
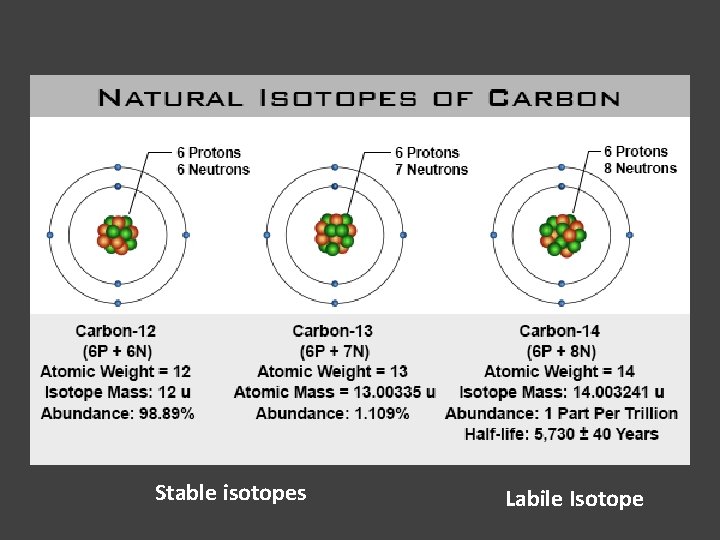
Stable isotopes Labile Isotope

Labile isotopes and radio-active decay:

a -decay:

b –decay (loss of an electron):

Reverse b –decay (gain of an electron):

a Decay 1 Helium nucleus = 2 neutron + 2 electron. An atom nucleus looses 1 Helium nucleus (= a decay): ( atom number -2, atom mass -4)

1 proton = neutron + electron. An atom nucleus looses an electron (= b decay): 1 Neutron 1 proton. ( atom number +1, atom mass not changed) An atom nucleus gains an electron: (= - b decay, or reverse b decay) 1 proton 1 neutron. ( atom number -1, atom mass not changed)
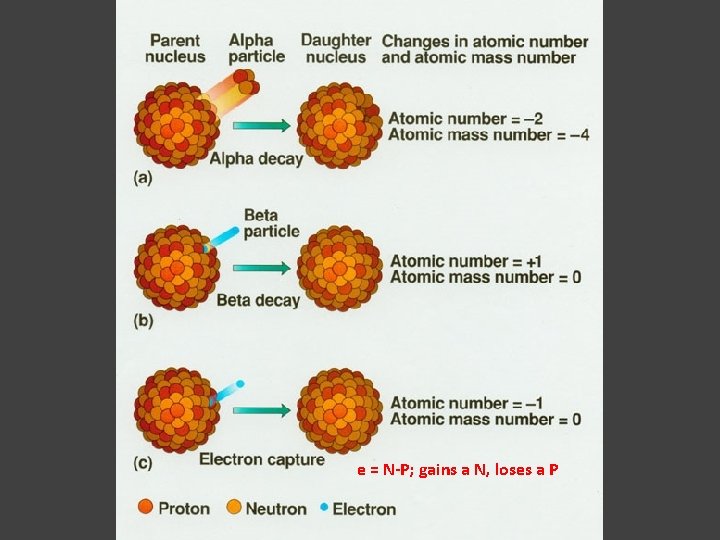
e = N-P; gains a N, loses a P

Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium--236 Uranium-238 Thorium-232 Radon-222

Radioactive Decay: Loss of an a particle Samarium-147 Americium Plutonium Uranium Thorium Radon

Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium Uranium Thorium Radon
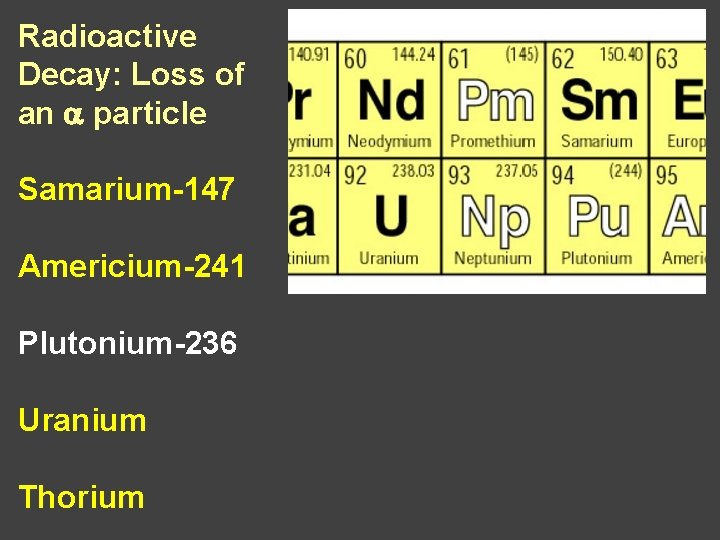
Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium-236 Uranium Thorium

Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium-236 Uranium-238 Thorium Radon

Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium-236 Uranium-238 Thorium-232 Radon

Radioactive Decay: Loss of an a particle Samarium-147 Americium-241 Plutonium-236 Uranium-238 Thorium-232 Radon-222

Radioactive Decay: Loss of an a particle Samarium Americium Plutonium Uranium Thorium Radon

Radioactive Decay: Loss of an b particle (= loss of an electron)

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Bismuth-210 Rhenium-187 Iodine-129 Rubidium-87 Chlorine-39 Carbon-14

Radioactive Decay: Loss of a b particle. Rubidium Rhenium Iodine Carbon Bismuth chlorine

Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11

Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11

Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11

Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11

Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11
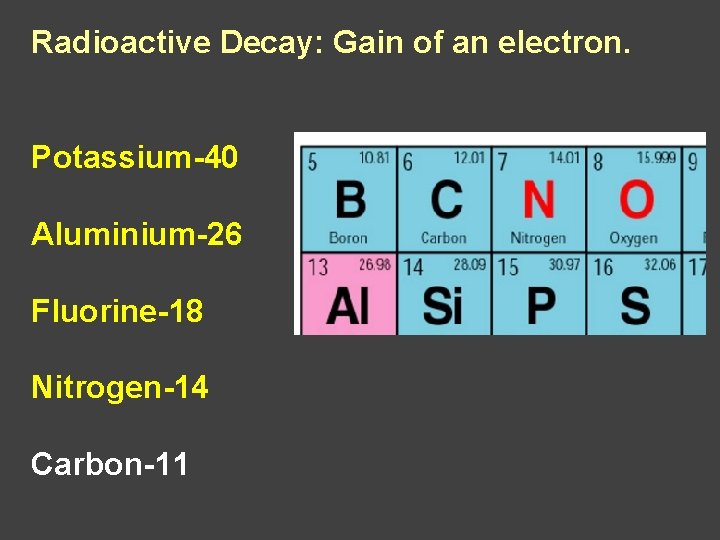
Radioactive Decay: Gain of an electron. Potassium-40 Aluminium-26 Fluorine-18 Nitrogen-14 Carbon-11

Radioactive Decay: Gain of a b particle Potassium Aluminium Nitrogen Carbon Fluorine

Radioactive decay Series: Radon-219 to Lead-207. 219 ----> Po 215 ----> Pb 211 ----> Rn 86 84 82 211 ----> Tl 207 ----> Pb 207 Bi 83 81 82 Rn: Radon Po: Polonium Pb: Lead Bi: Bismuth Tl: Thallium
- Slides: 57