DATA Metric System SI Taking Measurements Organization Presentation

































- Slides: 56

DATA Metric System (SI) Taking Measurements Organization Presentation

Data • Sometimes Your data are thealldata the seem records to you be wrong, have gathered but evenfrom then, an investigation. they are important and should be recorded accurately. • The types of data collected depend on the activity. • Remember that nature cannot be wrong, • Data may be a series of weights of volumes, a set of color changes, regardless of what you discover in the laboratory. or a list of scientific names. • Data that seem to be “wrong” are probably the result of the experimental error. • No matter which types of data are collected, all data must be treated carefully to ensure accurate results.

Metric System l l A decimal system of measurement whose units are based on certain physical standards and scaled on ten multiples of ___ Based on units of 10

Metric System Kilo (k) = 1000 l Hecta (h) = 100 l Deka (D) = 10 l Base = 1 l Deci (d) = 0. 1 l Centi (c) = 0. 01 l Milli (m) = 0. 001 l The system is easy to use because it is based on multiples of 10 1 centimeter kilogram liter kilometer = =1000 100 ==1000 milliliters cm 10 milligrams millimeters grams meters

Measurement in Science l Scientists use a revised form of the metric system called Le Systeme International d’Unites (International System of Units) or SI Units

S. I. Base Units The International System of Units (S. I. ) defines seven base units for the seven base quantities: Quantity Name Symbol length meter m mass kilogram kg time second s electric current ampere A thermodynamic temperature kelvin K luminous intensity candela cd amount of substance mol

S. I. Base Units

Length l The distance from one point to another l Meter – the SI unit of length l Tool used to measure length depends on the size of the object Frequently used SI units l l Larger objects (i. e. the room) 1 millimeter (mm) = 1000 micrometers (mm) l – meter stick 1 centimeter (cm) = 10 millimeters (mm) Length 1 meter (m) = length) 100 centimeters (cm) Smaller objects (i. e. pencil l 1 kilometer (km) = 1000 meters (m) – centimeter ruler 1 light-year = 9, 460, 000, 000 kilometers (km)

Measuring Length l Measure the distance from Point A to B l What distance is Point A? Point B? 4. 5 cm l 6. 4 cm What is the total distance between points? 6. 4 cm – 4. 5 cm = 1. 9 cm

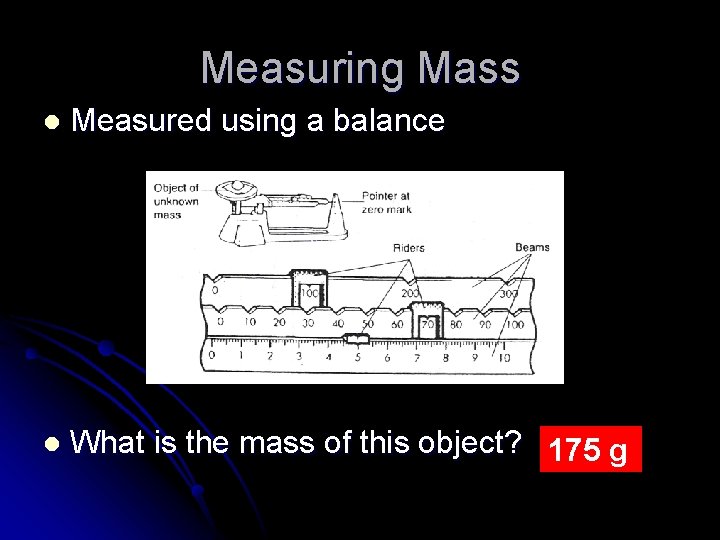
Mass l l l The amount of matter in something Mass is measured using a balance or triple beam balance Or an electronic balance

Mass l l The SI unit for mass is the kilogram (kg) The gram is the base unit However, some measurements of mass like the mass of vitamins or medicines are so small that we use milligrams (mg) to measure their mass Frequently used SI units Mass 1 gram (g) = 1000 milligrams (mg) 1 kilogram (kg) = 1000 grams (g) 1 metric ton = 1000 kilograms (kg)

Measuring Mass l Measured using a balance l What is the mass of this object? 175 g

Area is a physical quantity expressing the size of a part of a surface. l The term can also be used in a nonmathematical context to be mean "vicinity". l Surface area is the summation of the areas of the exposed sides of an object. l Frequently used SI units Area 1 square meter (m 2) = 10, 000 square centimeters (cm 2) 1 square kilometer (km 2) = 1, 000 square meters (m 2)

Measuring Area l Area = Length x Width 1 cm The square has an area of 4 square centimeters (4 cm 2) 1 cm Area = l x l 2 2 cm = 2 cm x 2 cm Area = 4 square centimeters (4 cm 2)

Measuring Area l Calculate the area of the following rectangles 2 cm 3 cm cm 5 cm 8 cm Area = 2 cm X 5 cm Area = 10 cm 2 Area = 3 cm X 8 cm Area = 24 cm 2

Volume l l The amount of space a substance takes up Volume of a liquid is found using a graduated cylinder l Unit is liters (L) or milliliters (m. L) Frequently used SI units Volume 1 milliliter (m. L) = 1 cubic centimeter (cc) or (cm 3) 1 liter (L) = 1000 milliliters (m. L) or 1 dm 3

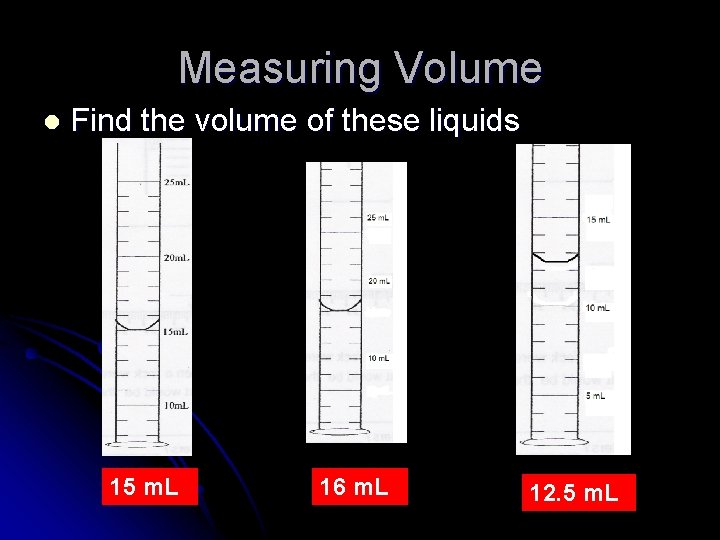
Measuring Volume l Liquid volume measured using graduated cylinder l Read volume at meniscus ( downward curve of water) l What is the volume of this liquid? l 45 m. L

Measuring Volume l Find the volume of these liquids 15 m. L 16 m. L 12. 5 m. L

Volume l Volume of a solid is found using the volume equation l length x width x height l In this equation, we are also multiplying the units together so units for the volume of a solid are… l m x m = m 3 OR cm x cm = cm 3 l Also know that 1 m. L = 1 cm 3 = 1 cc (cubic centimeter)

Measuring Volume l Solid volume measured using volume equation (l x w x h) What is the volume of a cube that is 2 cm x 2 cm? Volume = l 1 x l 2 x l 3 = 2 cm x 2 cm Volume = 8 cubic centimeters (8 cm 3)

Measuring Volume l Calculate the volume of the box Volume = l x w x h Volume = 6 mm X 5 mm X 2 mm 6 mm Volume = 60 mm 3 2 mm 5 mm

Water Displacement Some solid samples, such as an irregularly shaped rock cannot have their volume measured easily by using the volume equation (length x width x height) l For these solids, scientists use a technique called Water Displacement l

Water Displacement Steps of Water Displacement l 1. Add water to a graduated cylinder and record its volume (ex: 7 ml) l 2. Place the irregularly shaped solid into the graduated cylinder already containing water and record the new volume (ex: 9 ml) l

Water Displacement l l 3. Subtract the smaller volume (combined volume) from the larger volume (water only) to get the volume of the irregularly shaped solid. (ex: 9 ml – 7 ml = 2 ml) 4. We would say that the irregularly shaped solid takes up 2 ml of space. Since it is a solid, we need to state the volume using cm 3 so we would say that its volume is 2 cm 3

Water Displacement Volume = V 2 – V 1 Volume = 30 – 20 Volume = 10 m. L = 10 cm 3

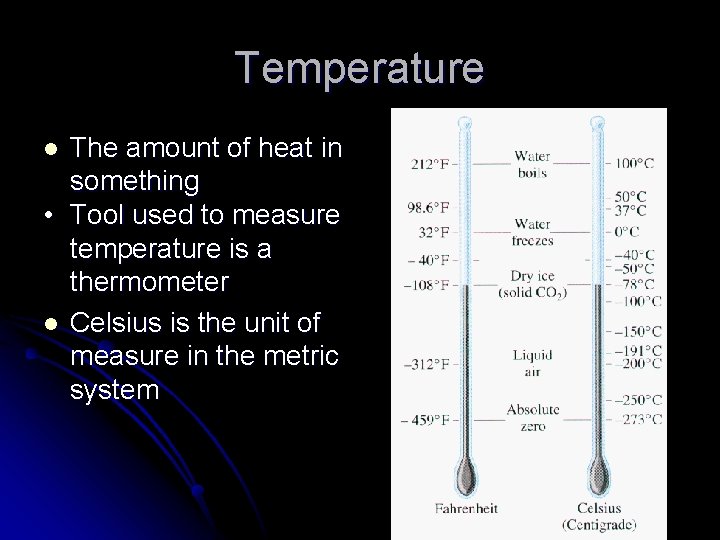
Temperature The amount of heat in something • Tool used to measure temperature is a thermometer l Celsius is the unit of measure in the metric system l

Temperature l SI unit for temperature is the Kelvin (K) l l Notice that there is no degree sign used! Kelvin scale is easier for scientists to use To calculate Kelvin you add 273 to the Celsius temperature Ex. 23 ○C = 23 + 273 = 296 K

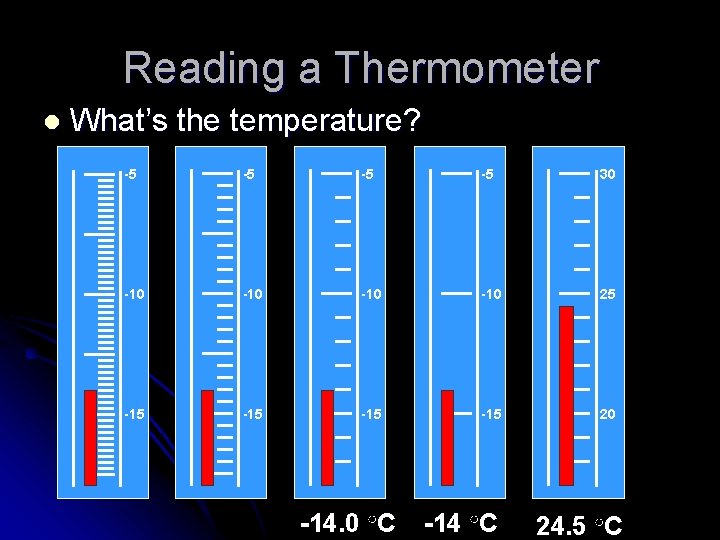
Reading a Thermometer l What’s the temperature? -5 -5 30 -10 -10 25 -15 -15 20 -14. 0 ○C -14 ○C 24. 5 ○C

Time l l l The period between 2 events The SI unit of time is the second (s) Measurement tool of time is a stopwatch

Recording and Organizing Laboratory Data • There are many ways to record and organize data, including: • data tables, charts, and graphs. • It is important to include the appropriate units when you record data. • Remember that DATA are measurements or observations, not merely numbers. • Data tables, graphs, and diagrams should have descriptive titles to ensure that another person can understand them without having been present during the investigation. Many important scientific discoveries have been made accidentally in the course of an often unrelated laboratory activity. Scientists who keep very careful and complete records sometimes notice unexpected trends in and relationships among data long after the work is completed. The laboratory notebooks of working scientists are studded with diagrams and notes; every step of every procedure is carefully recorded.

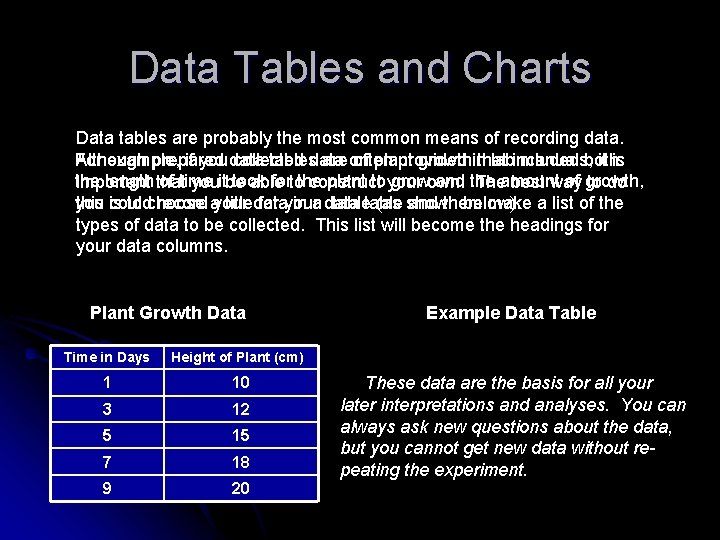
Data Tables and Charts Data tables are probably the most common means of recording data. For example, if you data collected plant growthinthat Although prepared tablesdata are on often provided labincluded manuals, both it is the length that of time took fortothe plant to your growown. and the of growth, important youitbe able construct Theamount best way to do you could record ayour in a data table (as shown below). this is to choose titledata for your and them make a list of the types of data to be collected. This list will become the headings for your data columns. Plant Growth Data Time in Days Height of Plant (cm) 1 10 3 12 5 15 7 18 9 20 Example Data Table These data are the basis for all your later interpretations and analyses. You can always ask new questions about the data, but you cannot get new data without repeating the experiment.

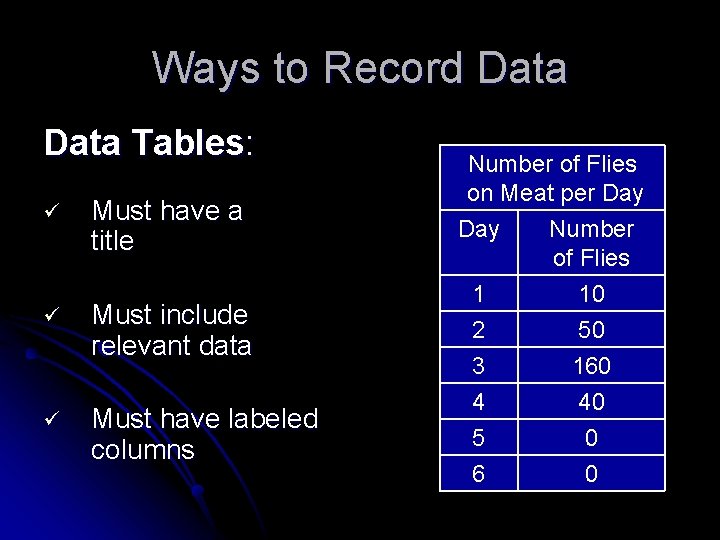
Ways to Record Data Tables: ü Must have a title ü Must include relevant data ü Must have labeled columns Number of Flies on Meat per Day Number of Flies 1 10 2 50 3 160 4 40 5 0 6 0

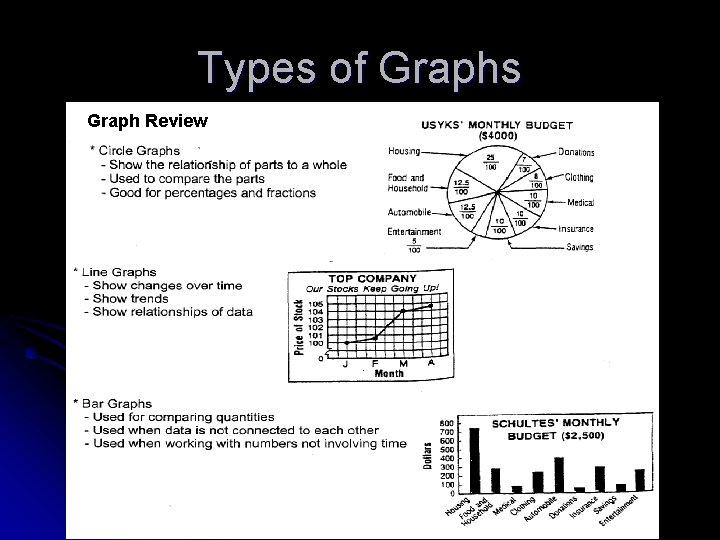
Graphs After data are collected, you must determine how to display them l One way of showing your results is to use a graph l Three types of graphs are commonly used l l Pie (circle) graphs Line graphs Bar graphs

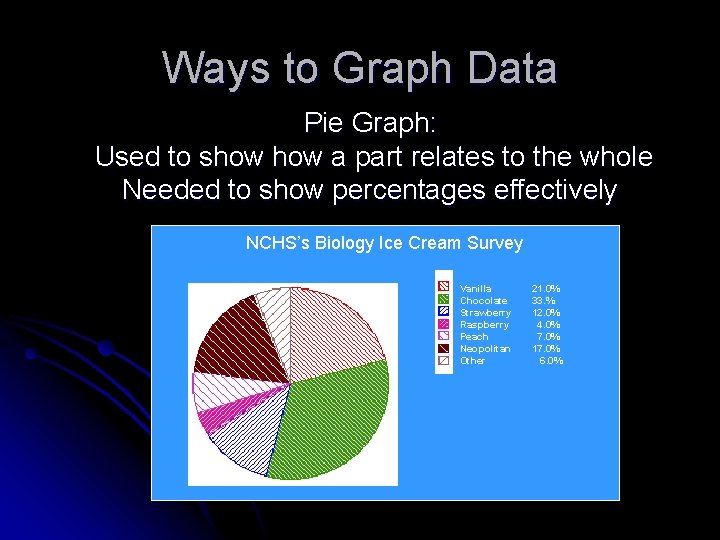
Ways to Graph Data Pie Graph: Used to show a part relates to the whole Needed to show percentages effectively NCHS’s Biology Ice Cream Survey Vanilla Chocolate Strawberry Raspberry Peach Neopolitan Other 21. 0% 33. % 12. 0% 4. 0% 7. 0% 17. 0% 6. 0%

Ways to Graph Data (Continued) l Line graph: used to show change over time l Bar graph: used to compare quantities in different categories l Be sure to include: ü Title ü Axis labels ü Key

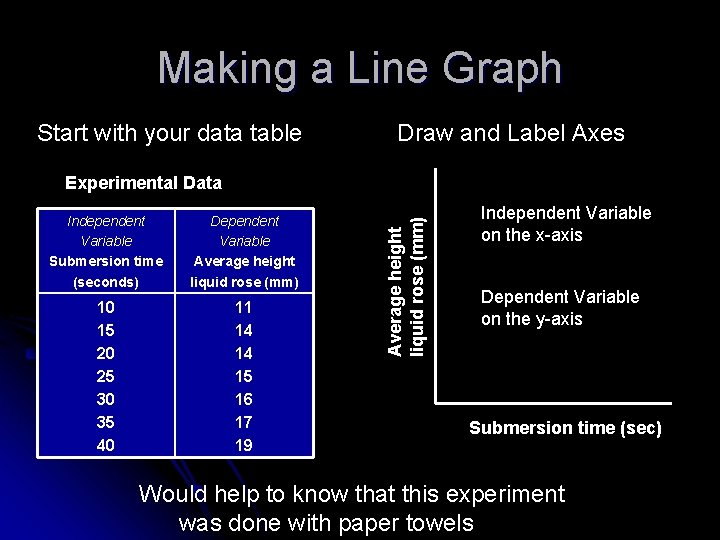
Making a Line Graph Start with your data table Draw and Label Axes Independent Variable Submersion time (seconds) Dependent Variable Average height liquid rose (mm) 10 15 20 25 30 35 40 11 14 14 15 16 17 19 Average height liquid rose (mm) Experimental Data Independent Variable on the x-axis Dependent Variable on the y-axis Submersion time (sec) Would help to know that this experiment was done with paper towels

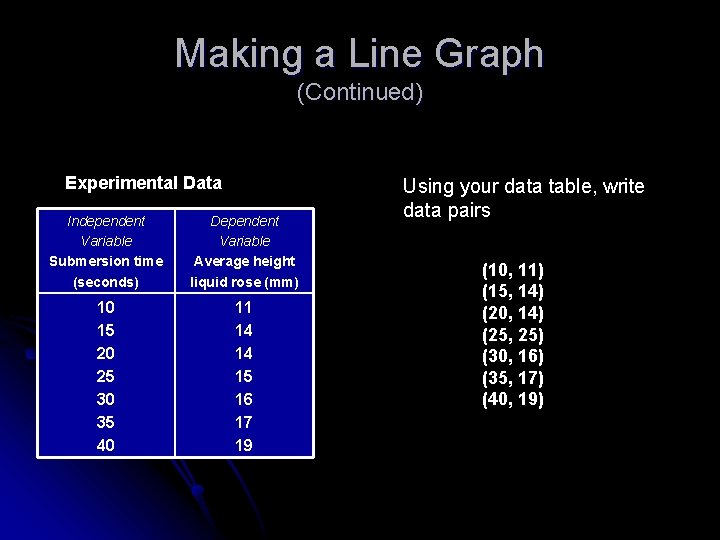
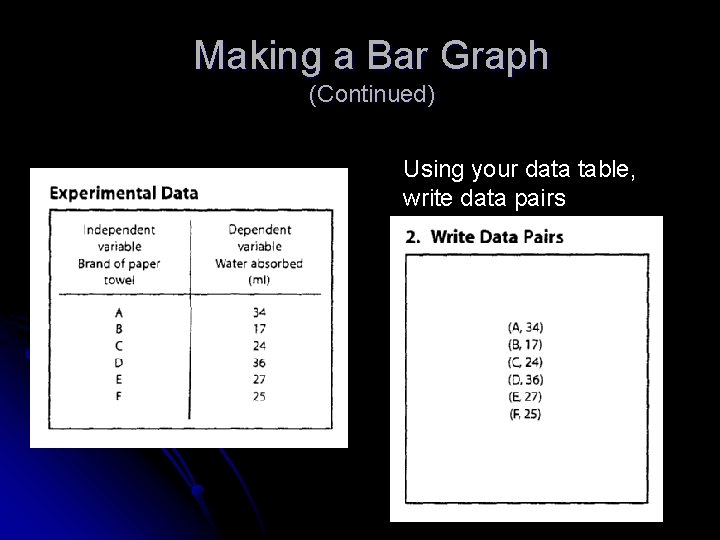
Making a Line Graph (Continued) Experimental Data Independent Variable Submersion time (seconds) Dependent Variable Average height liquid rose (mm) 10 15 20 25 30 35 40 11 14 14 15 16 17 19 Using your data table, write data pairs (10, 11) (15, 14) (20, 14) (25, 25) (30, 16) (35, 17) (40, 19)

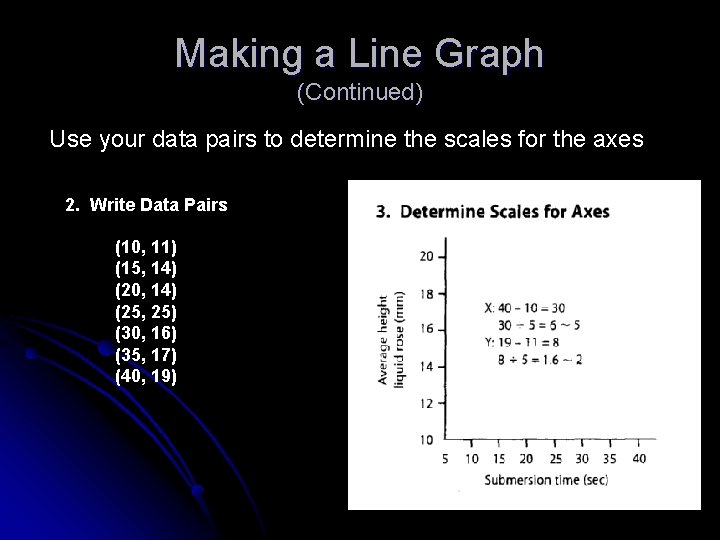
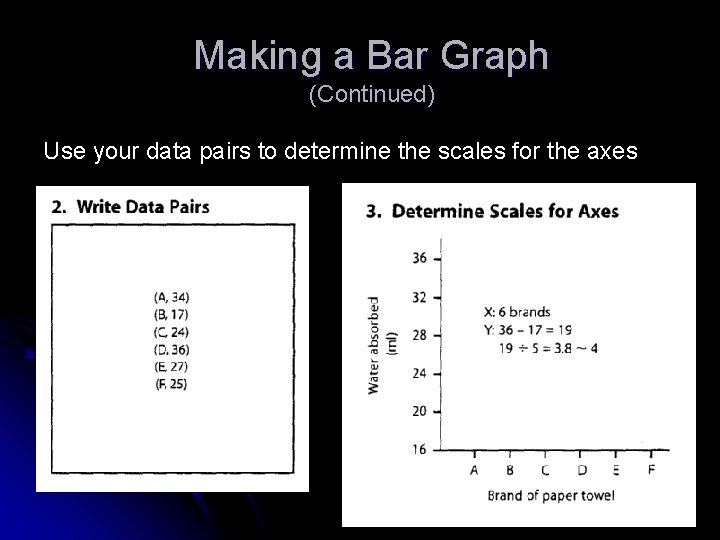
Making a Line Graph (Continued) Use your data pairs to determine the scales for the axes 2. Write Data Pairs (10, 11) (15, 14) (20, 14) (25, 25) (30, 16) (35, 17) (40, 19)

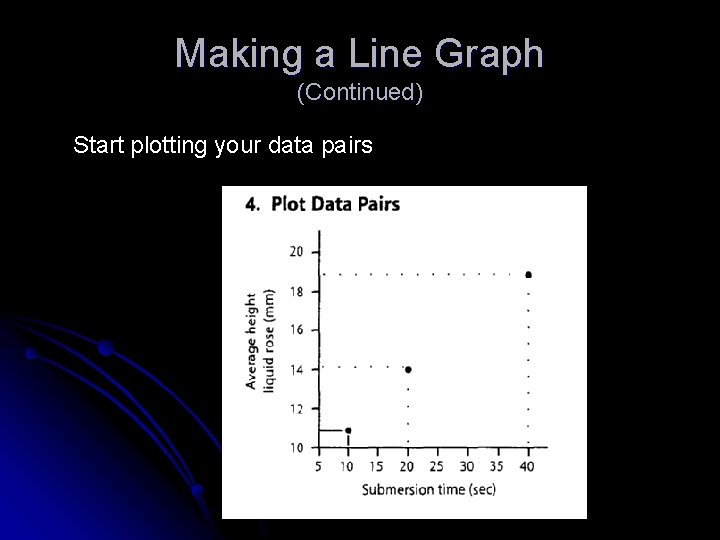
Making a Line Graph (Continued) Start plotting your data pairs

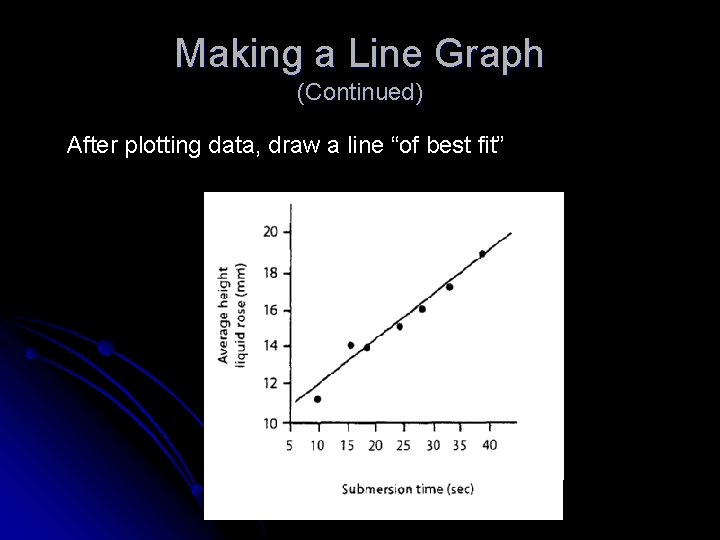
Making a Line Graph (Continued) After plotting data, draw a line “of best fit”

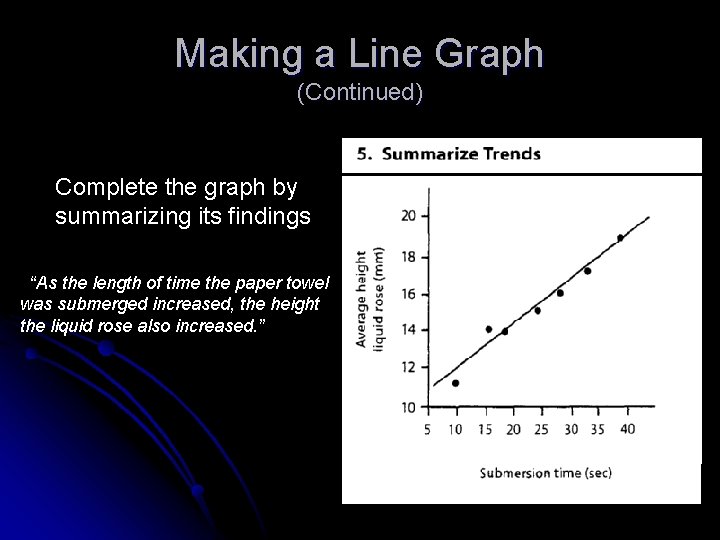
Making a Line Graph (Continued) Complete the graph by summarizing its findings “As the length of time the paper towel was submerged increased, the height the liquid rose also increased. ”

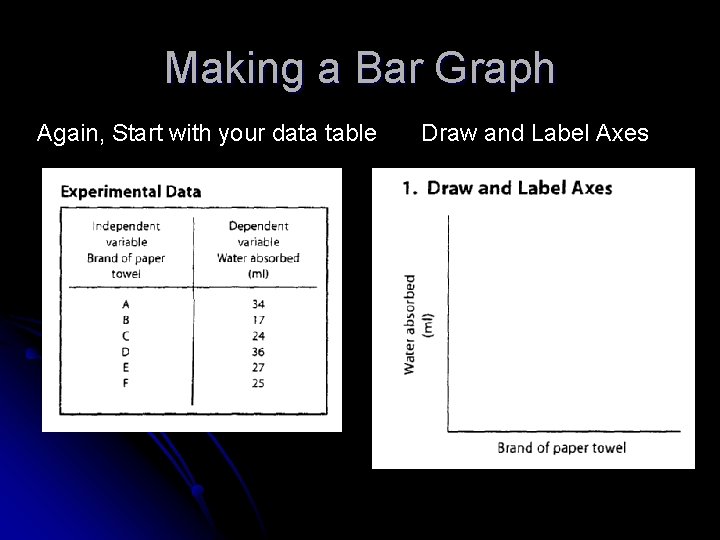
Making a Bar Graph Again, Start with your data table Draw and Label Axes

Making a Bar Graph (Continued) Using your data table, write data pairs

Making a Bar Graph (Continued) Use your data pairs to determine the scales for the axes

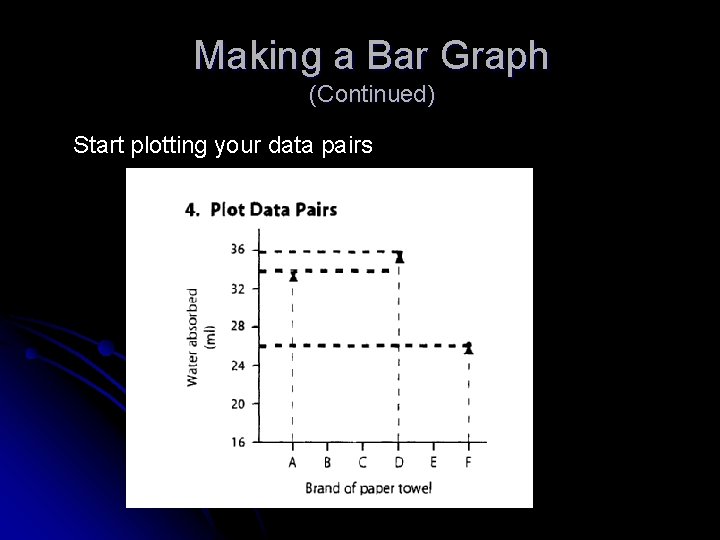
Making a Bar Graph (Continued) Start plotting your data pairs

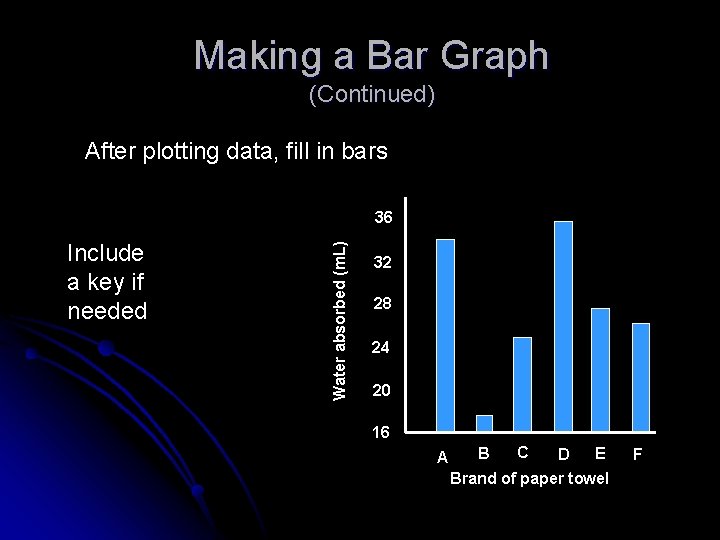
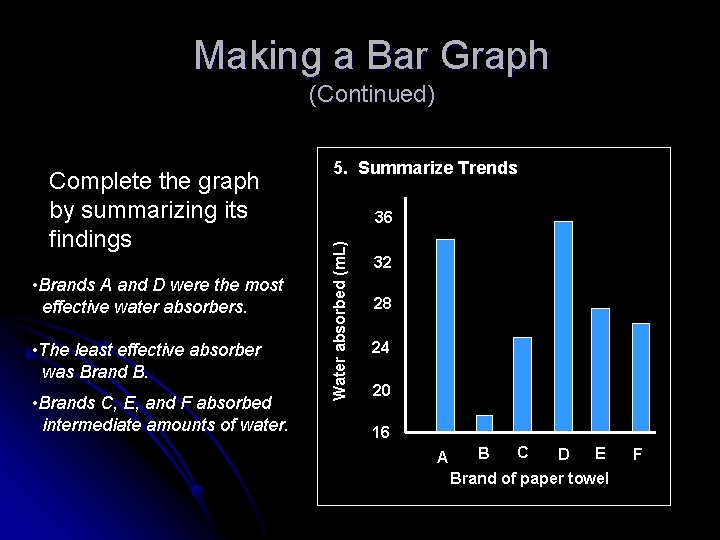
Making a Bar Graph (Continued) After plotting data, fill in bars Include a key if needed Water absorbed (m. L) 36 32 28 24 20 16 A B C E D Brand of paper towel F

Making a Bar Graph (Continued) • Brands A and D were the most effective water absorbers. • The least effective absorber was Brand B. • Brands C, E, and F absorbed intermediate amounts of water. 36 Water absorbed (m. L) Complete the graph by summarizing its findings 5. Summarize Trends 32 28 24 20 16 A B C E D Brand of paper towel F

Graphing Review l Purposes: l l l Organize and analyze data Show patterns Communicate information Allow scientists to make predictions Components: l l l Appropriate title Equal units on each axis with good use of spacing Labels on each axis including units l l (example, mm, sec. , kg. ) Neatly created using a variety of color

Types of Graphs Graph Review

Statistical Methods & Analysis of Errors

ERROR l May be defined as a deviation from the absolute value or from the true average of a large number of results. TYPES OF ERRORS 1. Determinate error - constant 2. Indeterminate error – random or accidental

Determinate Error l l Usually present in each measurement and affect all observations in a series. Example: l l l l Errors in calibration Operation in measuring Impurities in the reagents or drugs Biased personal errors Pouring or mixing Weighing operations Matching colors Making calculations

Indeterminate Error Occur by chance l Vary from one measurement to the next l Yield a random pattern around an average l Occur also in the filling of capsules l Random fluctuations in temperature and other factors l

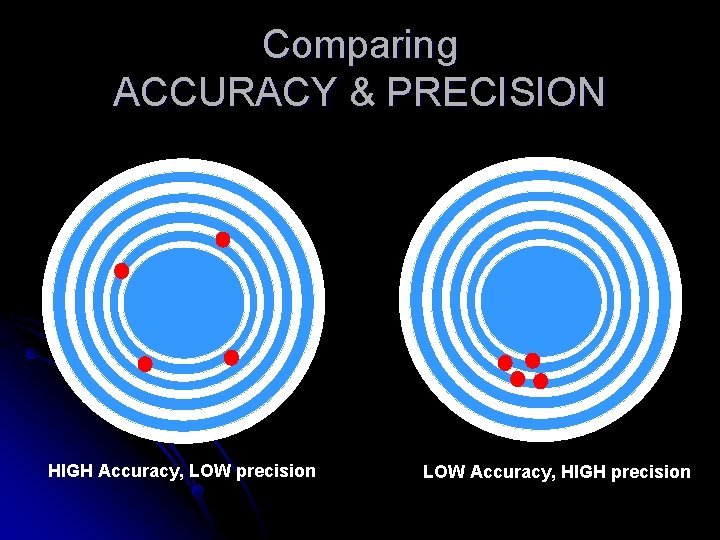
PRECISION & ACCURACY – is the degree of closeness of a measured or calculated quantity to its actual (true) value. PRECISION – also called REPRODUCIBILITY the degree to which measurements or calculations show the same similar results.

Comparing ACCURACY & PRECISION HIGH Accuracy, LOW precision LOW Accuracy, HIGH precision

Prepare for an exam next meeting … This presentation can be viewed or downloaded at: http: //roselynnaranjo. yolasite. com