DARIFENACIN OAB Syndrome Definition International Continence Society Symptom

DARIFENACIN

OAB Syndrome: Definition International Continence Society: • Symptom syndrome: lower urinary tract (LUT) dysfunction • Urgency, with or without urge incontinence, usually with frequency & nocturia • No proven infection / other obvious pathology 2 Abrams P, et al, Neurourol Urodyn, 2002; 21: 167 -178 / Wein AJ, et al, Urology, 2002; 60 (suppl 5 A); 7 -12
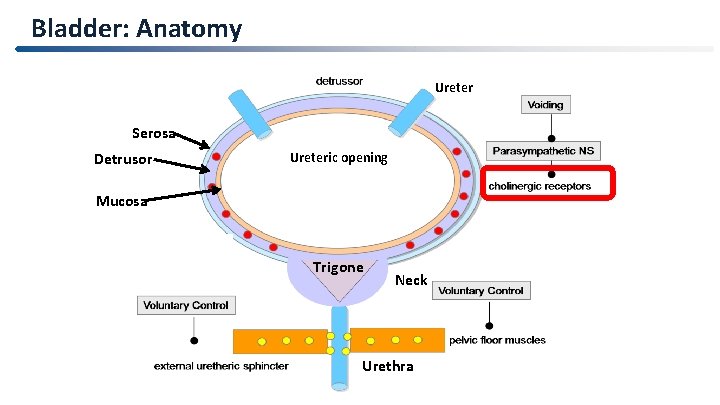
Bladder: Anatomy Ureter Serosa Detrusor Ureteric opening Mucosa Trigone Neck Urethra
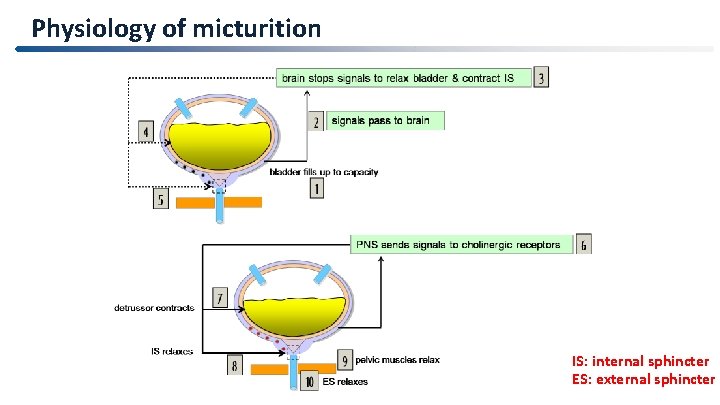
Physiology of micturition IS: internal sphincter ES: external sphincter

Symptoms of urinary incontinence… Urinate frequently, usually 8 or more times in 24 hrs Sudden urge Involuntary loss of urine to urinate that's difficult to control Significantly impacts Qo. L 1. Minassian VA, et al. Obstet Gynecol. 2008 Feb; 111(2 Pt 1): 324 -31/ OAB symptom ref: Mayo clinic Awaken 2 or more times in night to urinate (nocturia)
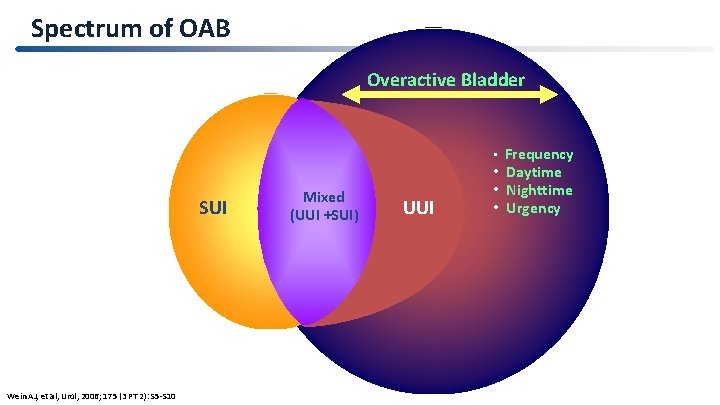
Spectrum of OAB Overactive Bladder • Frequency SUI z Mixed (UUI +SUI) UUI • Daytime • Nighttime • Urgency 6 Wein AJ, et al, Urol, 2006; 175 (3 PT 2): S 5 -S 10
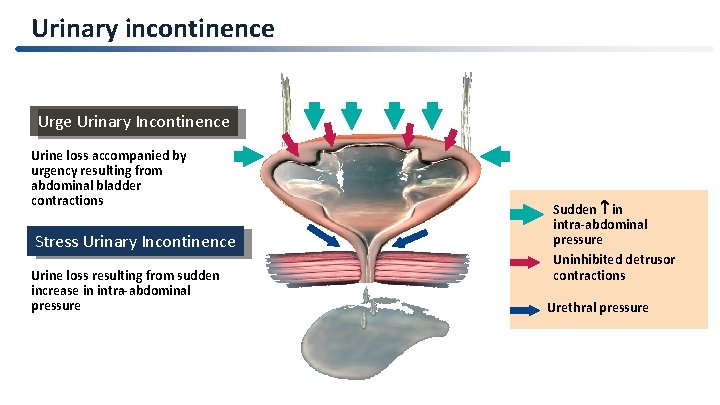
Urinary incontinence Urge Urinary Incontinence Urine loss accompanied by urgency resulting from abdominal bladder contractions Stress Urinary Incontinence Urine loss resulting from sudden increase in intra-abdominal pressure Sudden in intra-abdominal pressure Uninhibited detrusor contractions Urethral pressure 7

OAB: Treatment Goals • Eliminate or in urinary urge incontinence • urgency & frequency episodes • Ensure treatment compliance: long-term benefits • Meet patients treatment expectations Hegde SS. Br J Pharmacol. 2006; 147 (suppl 2): S 80 -S 87, Staskin DR, et al, Am J Med. 2006; 119 (suppl 3 A): 9 S-15 S, Marschall-Kehrel D, et al. Urology, 2006, 68 (suppl 2 A), Brubaker L, et al Urology. 2006; 68(suppl 2 A) 3 -8 8

OAB: Management • Behavioral therapies • Pharmacology therapy • Combined pharmacologic & behavioral therapy provides improved outcomes Fluid strategies Timed voiding 9 Mattiasson A. Urology. 2000; 55(suppl 5 a): 12 -13, Mattiasson A. Neuro Urodyn. 2001; 20: 403 -404, Burgio et al. JAGS. 2000; 48: 370 -374.

LAUREN HERSH. Am Fam Physician. 2013; 87(9): 634 -640.
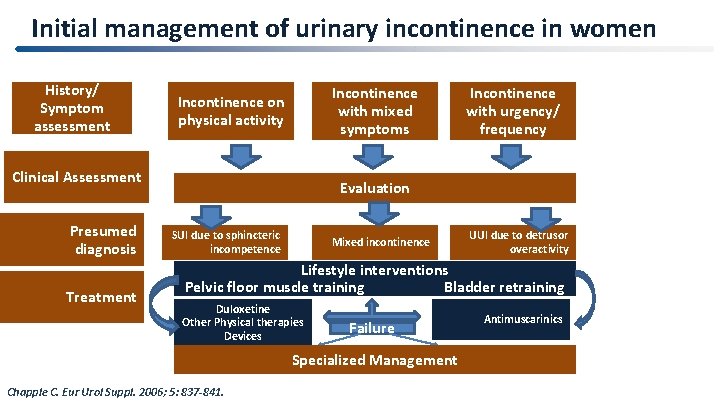
Initial management of urinary incontinence in women History/ Symptom assessment Incontinence with mixed symptoms Incontinence on physical activity Clinical Assessment Presumed diagnosis Treatment Incontinence with urgency/ frequency Evaluation SUI due to sphincteric incompetence Mixed incontinence UUI due to detrusor overactivity Lifestyle interventions Pelvic floor muscle training Bladder retraining Duloxetine Other Physical therapies Devices Failure Specialized Management Chapple C. Eur Urol Suppl. 2006; 5: 837 -841. Antimuscarinics
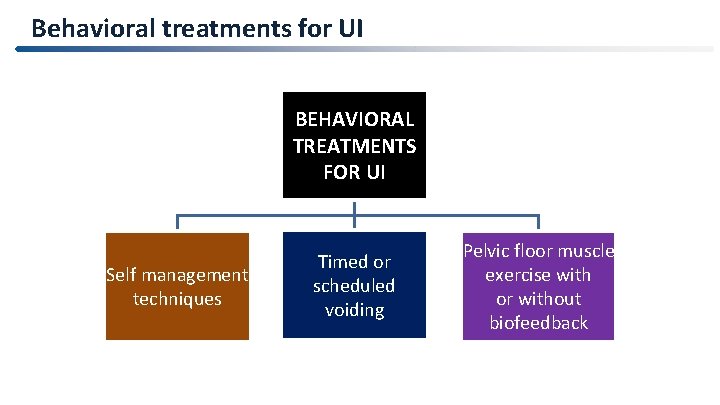
Behavioral treatments for UI BEHAVIORAL TREATMENTS FOR UI Self management techniques Timed or scheduled voiding Pelvic floor muscle exercise with or without biofeedback

OAB: Pharmacotherapy Drug therapy: becoming increasingly important & currently mainstay in treatment for OAB Antimuscarinic agents: Gold standard 13 Mattiasson A. Urology. 2000; 55(suppl 5 a): 12 -13, Mattiasson A. Neuro Urodyn. 2001; 20: 403 -404, Burgio et al. JAGS. 2000; 48: 370 -374.

Anticholinergic agents have been 1 st line treatment for OAB for many years, efficacious pharmacologic management of this condition has been compromised by concerns regarding tolerability - Hesch K. Proc (Bayl Univ Med Cent) 2007; 20(3): 307– 314.
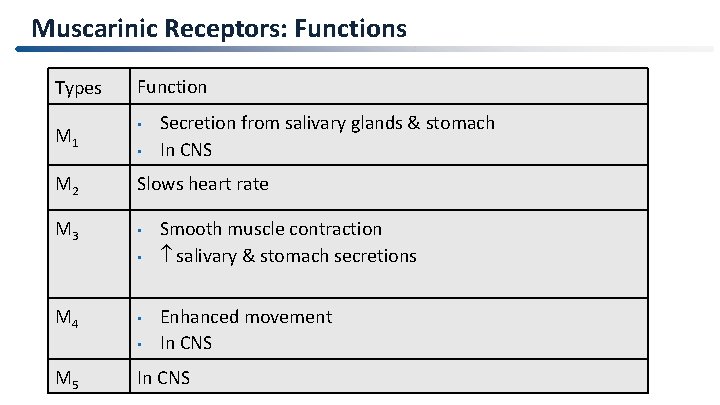
Muscarinic Receptors: Functions Types M 1 Function • • Secretion from salivary glands & stomach In CNS M 2 Slows heart rate M 3 • • M 4 • • M 5 Smooth muscle contraction salivary & stomach secretions Enhanced movement In CNS 15
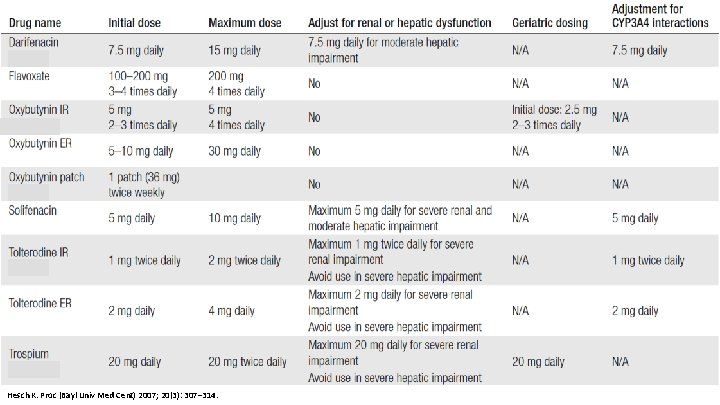
Hesch K. Proc (Bayl Univ Med Cent) 2007; 20(3): 307– 314.
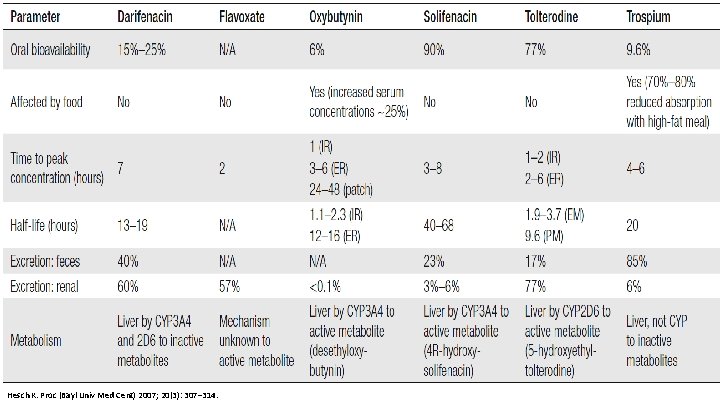
Hesch K. Proc (Bayl Univ Med Cent) 2007; 20(3): 307– 314.
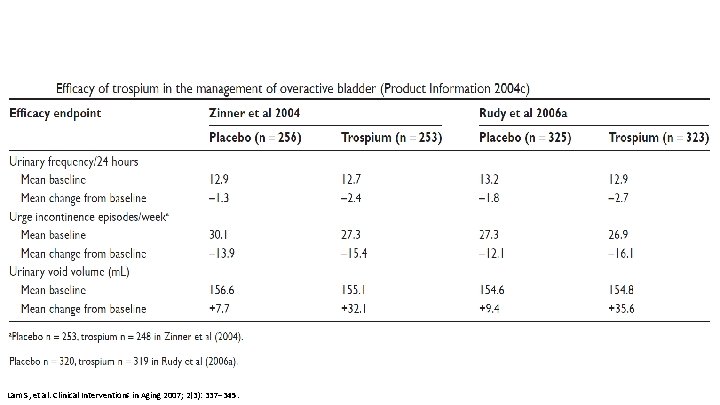
Lam S, et al. Clinical Interventions in Aging 2007; 2(3): 337– 345.
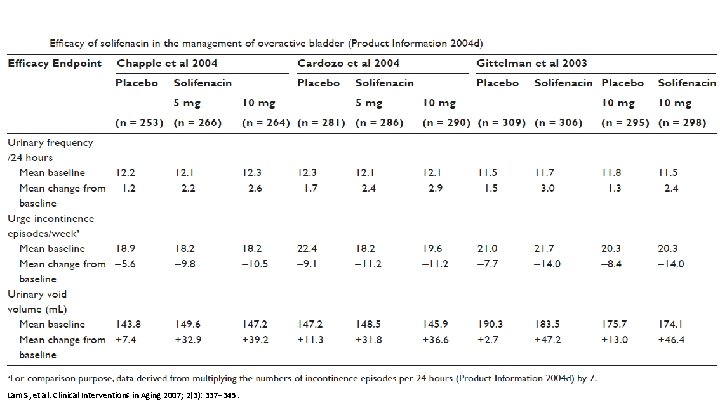
Lam S, et al. Clinical Interventions in Aging 2007; 2(3): 337– 345.
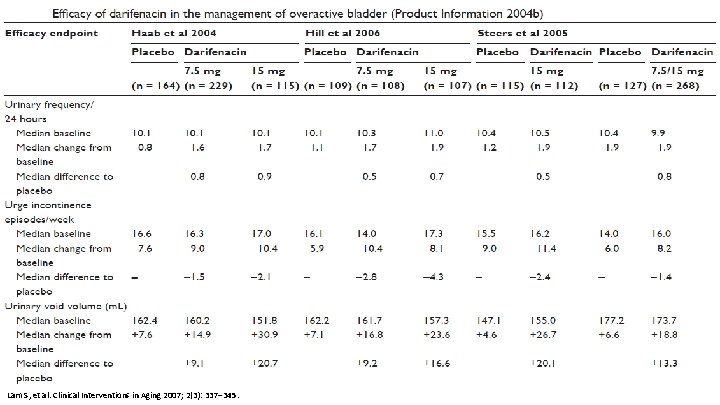
Lam S, et al. Clinical Interventions in Aging 2007; 2(3): 337– 345.

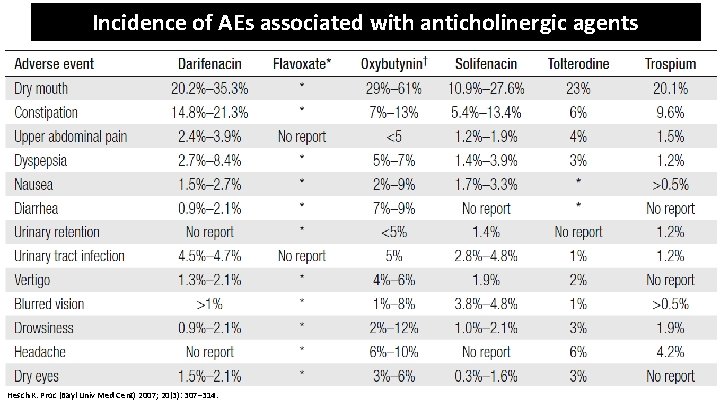
Incidence of AEs associated with anticholinergic agents Hesch K. Proc (Bayl Univ Med Cent) 2007; 20(3): 307– 314.

Ideal muscarinic receptor antagonist • Efficacious: – Inhibits involuntary bladder contractions – Does not adversely affect voluntary detrusor activity • Organ selective: – Preferentially affects bladder over other organs – Minimal side effects & improves tolerability • Tolerable: – Improves compliance 24

A drug with selectivity for muscarinic M 3 receptor, subtype primarily responsible for mediating human detrusor contraction might be effective in treating OAB symptoms & with narrower side-effect profile

Darifenacin • Darifenacin hydrobromide: Novel highly selective M 3 receptor antagonist • 59 -fold higher affinity for M 3 receptors Approved: US FDA 2004 • M 1/M 2 receptor sparing profile • Low incidence: Dry mouth & constipation • No impairment of cardiac function • No significant effect on cognitive function • No CNS safety concerns • AE withdrawal rates similar to placebo 26 Chapple CR. Expert Opin Investig Drugs. 2004 Nov; 13(11): 1493 -500.

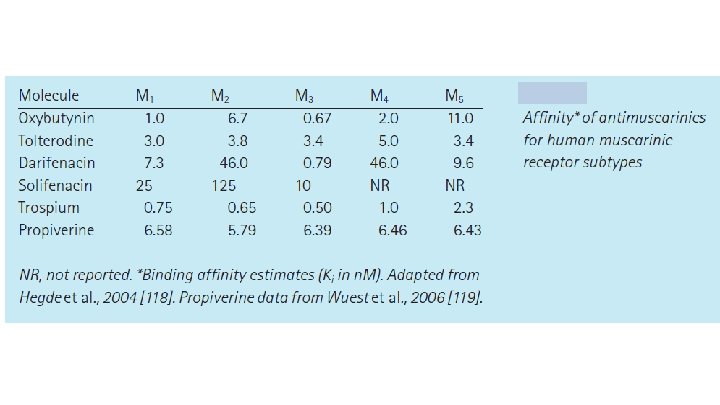
Darifenacin targets M 3 receptor in vitro M 3 over M 1 9 -fold greater M 3 over M 2 59 -fold greater M 3 receptors in bladder are primarily responsible for detrusor contraction 2* M 3 receptors involved in GI smooth muscle contraction, saliva production & iris sphincter function 2+ *Dry mouth, constipation & abnormal vision may be mediated through effects on M 3 receptors +Muscarinic M receptors are also found in other tissues but their role at these locations is as yet unclear 3 1. Napier C et al. Neurourol Urodyn 2002; 21(4): A 445/ 2. Abrams P et al. Br J Pharmacol 2006; 148: 565 -78.
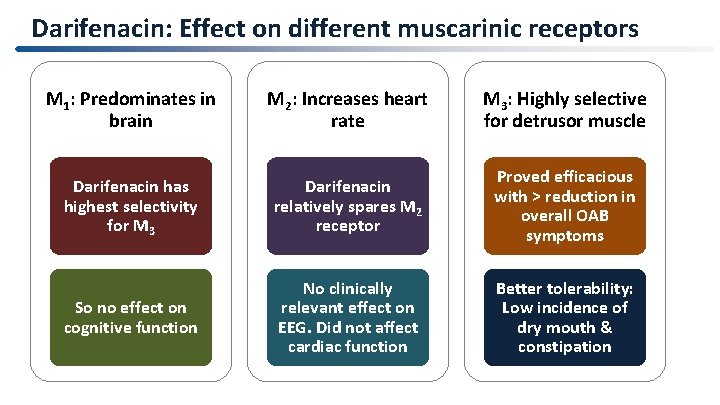
Darifenacin: Effect on different muscarinic receptors M 1: Predominates in brain M 2: Increases heart rate M 3: Highly selective for detrusor muscle Darifenacin has highest selectivity for M 3 Darifenacin relatively spares M 2 receptor Proved efficacious with > reduction in overall OAB symptoms So no effect on cognitive function No clinically relevant effect on EEG. Did not affect cardiac function Better tolerability: Low incidence of dry mouth & constipation 28
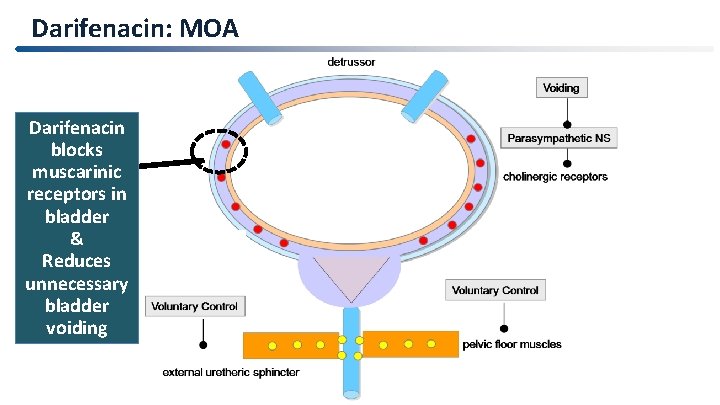
Darifenacin: MOA Darifenacin blocks muscarinic receptors in bladder & Reduces unnecessary bladder voiding 29

Darifenacin: Dosage and Administration • For oral use • Adults (≥ 18 years): – Starting dose 7. 5 mg daily, after 2 weeks of therapy, should be reassessed • For greater symptom relief, dose may be increased to 15 mg daily • Darifenacin should be taken OD • Elderly patients (≥ 65 years): – Starting dose 7. 5 mg daily, after 2 weeks of therapy, to be reassessed • Patients who have acceptable tolerability profile but require greater symptom relief, dose may be increased to 15 mg daily 30
Ref: François Haab, et al Total 716 patients mean age - 57 years Darifenacin 7. 5 mg OD x 2 weeks then to titrate to 15 mg as desired 85% - women 31 Haab F, et al. BJU international. 2006; 98: 1025 -1032.
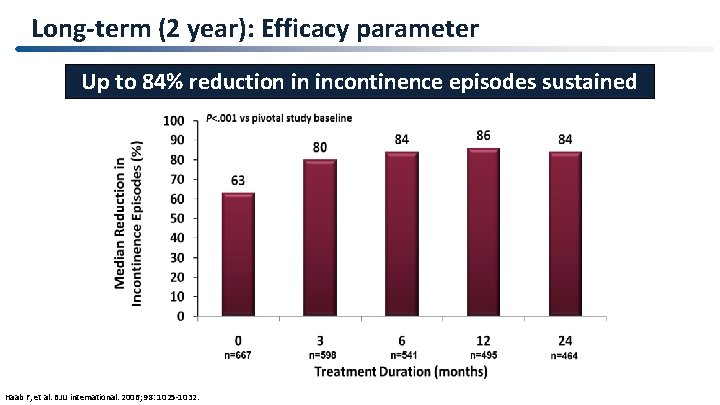
Long-term (2 year): Efficacy parameter Up to 84% reduction in incontinence episodes sustained Haab F, et al. BJU international. 2006; 98: 1025 -1032.
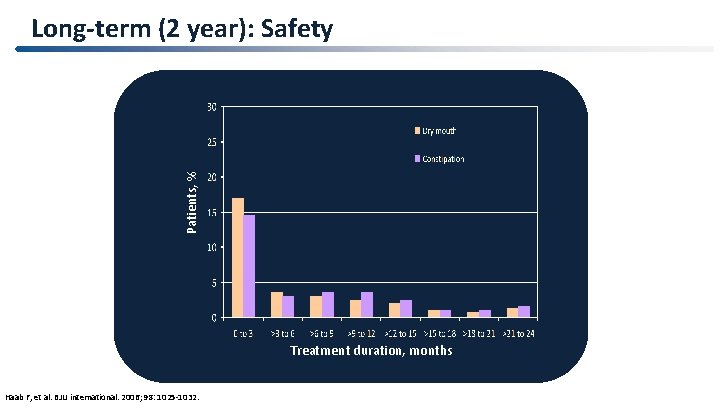
Patients, % Long-term (2 year): Safety Treatment duration, months 33 Haab F, et al. BJU international. 2006; 98: 1025 -1032.

Summary of the long term study (2 years) • Up to 84% reduction in incontinence episodes sustained • Favourable safety & tolerability • Low discontinuation rates Darifenacin (7. 5 & 15 mg OD): Effective in treatment of patients with OAB Haab F, et al. BJU international. 2006; 98: 1025 -1032.
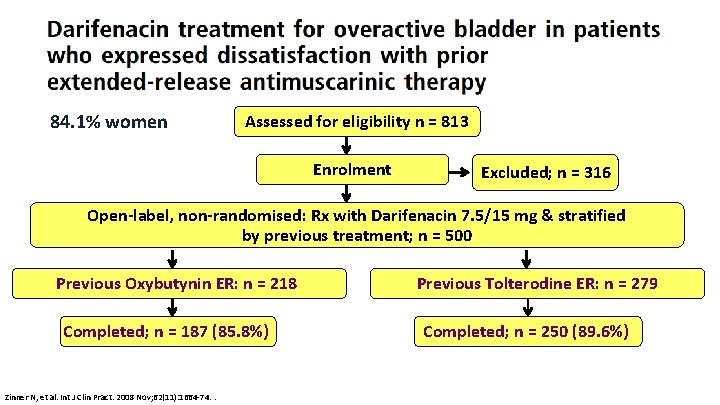
84. 1% women Assessed for eligibility n = 813 Enrolment Excluded; n = 316 Open-label, non-randomised: Rx with Darifenacin 7. 5/15 mg & stratified by previous treatment; n = 500 Previous Oxybutynin ER: n = 218 Completed; n = 187 (85. 8%) Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74. . Previous Tolterodine ER: n = 279 Completed; n = 250 (89. 6%)
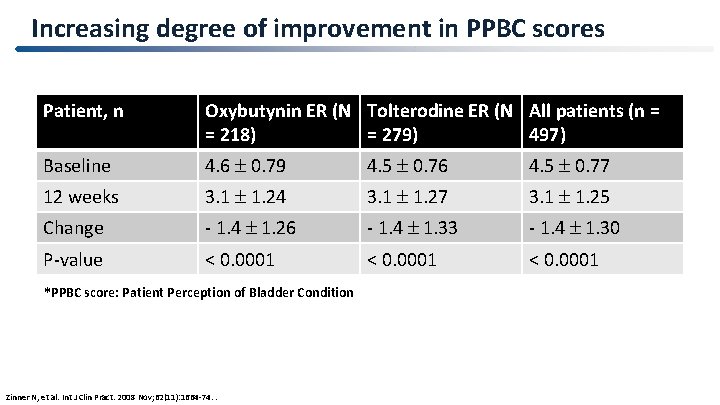
Increasing degree of improvement in PPBC scores Patient, n Oxybutynin ER (N Tolterodine ER (N All patients (n = = 218) = 279) 497) Baseline 4. 6 0. 79 4. 5 0. 76 4. 5 0. 77 12 weeks 3. 1 1. 24 3. 1 1. 27 3. 1 1. 25 Change - 1. 4 1. 26 - 1. 4 1. 33 - 1. 4 1. 30 P-value < 0. 0001 *PPBC score: Patient Perception of Bladder Condition Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74. .
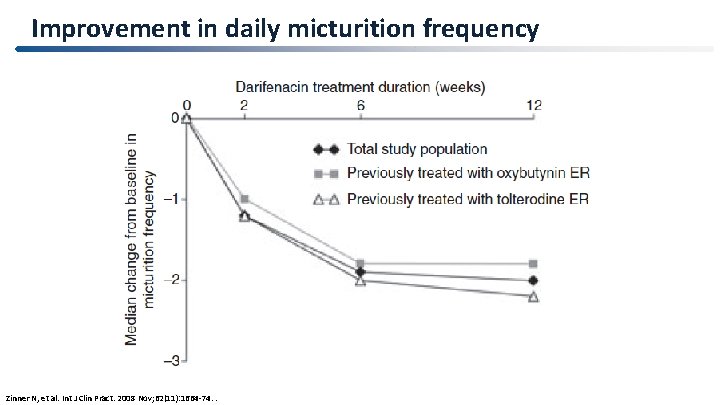
Improvement in daily micturition frequency Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74. .
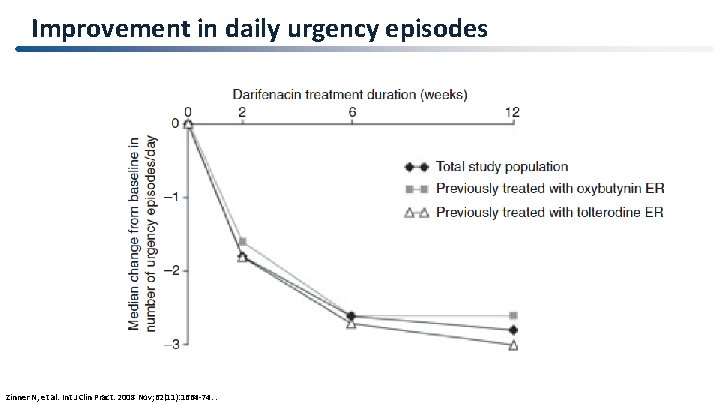
Improvement in daily urgency episodes Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74. .
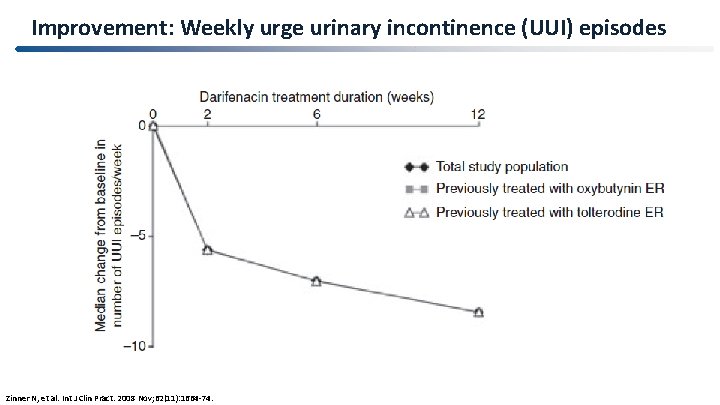
Improvement: Weekly urge urinary incontinence (UUI) episodes Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74.

Conclusion In patients who were dissatisfied with previous extended release antimuscarinic treatment (oxybutynin / tolterodine) • PPBC score & OAB symptoms were significantly improved • Satisfaction was high during treatment with darifenacin (7. 5 ⁄ 15 mg) Zinner N, et al. Int J Clin Pract. 2008 Nov; 62(11): 1664 -74.

41 Chappel C, et al. BJU International. 2005; 95: 993 -1001.

Study Design Total 1058 patients After 4 -wk washout period, randomized 12 weeks study Darifenacin 7. 5 mg OD Darifenacin 15 mg OD Placebo OD n = 337 n = 335 n = 388 Primary efficacy: Incontinence episodes per week Secondary efficacy: No of significant leaks per week Voiding frequency Bladder capacity Frequency & severity of urgency Nocturnal wakenings Responder rates Chappel C, et al. BJU International. 2005; 95: 993 -1001. 42
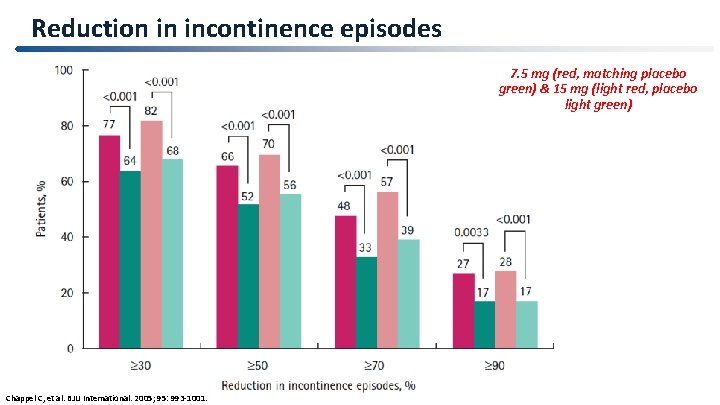
Reduction in incontinence episodes 7. 5 mg (red, matching placebo green) & 15 mg (light red, placebo light green) 43 Chappel C, et al. BJU International. 2005; 95: 993 -1001.
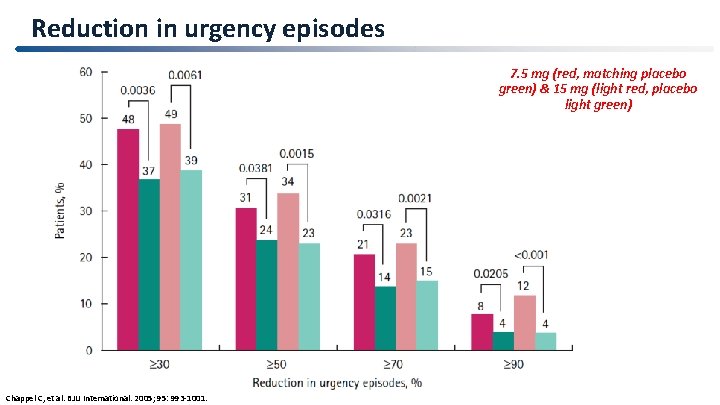
Reduction in urgency episodes 7. 5 mg (red, matching placebo green) & 15 mg (light red, placebo light green) 44 Chappel C, et al. BJU International. 2005; 95: 993 -1001.
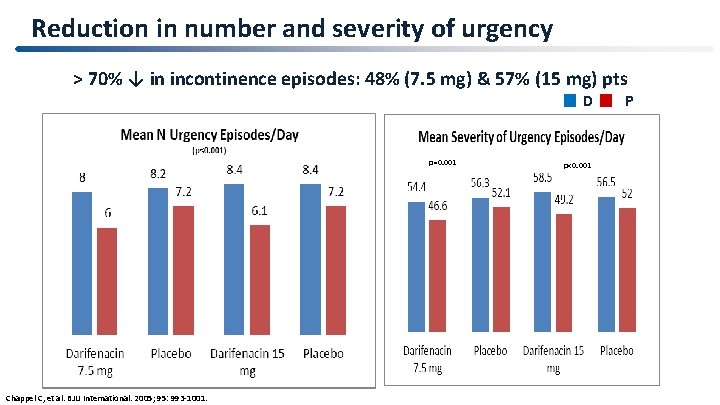
Reduction in number and severity of urgency > 70% ↓ in incontinence episodes: 48% (7. 5 mg) & 57% (15 mg) pts D p=0. 001 Chappel C, et al. BJU International. 2005; 95: 993 -1001. p<0. 001 P

Percentage responder rates % of patients responding Treatment group ≥ 3 dry days/week ≥ 7 consecutive dry days Darifenacin 7. 5 Placebo 55 43 19 15 0. 001 0. 108 Darifenacin 15 61 24 Placebo 48 16 P vs Placebo <0. 001 0. 011 P vs Placebo Darifenacin was well tolerated with no CNS or CV safety concerns 46 Chappel C, et al. BJU International. 2005; 95: 993 -1001.

Summary of the clinical study • Significant ↓ median no. of incontinence episodes/wk Darifenacin (7. 5 & 15 mg OD): • Significant ↓ frequency & severity of Effective treatment for OAB urgency, voiding frequency & no. of leaks (incontinence episodes>> change of clothing/ pads) • ↓ no. of nocturnal awakenings/wk • ↑ bladder capacity Chappel C, et al. BJU International. 2005; 95: 993 -1001.

Chapple et al: Curr Med Res Opin. 2007; 23(10): 2347 -58.

Darifenacin treatment of patients >or= 65 yrs with OAB • Evaluating efficacy, tolerability, safety & Qo. L • Patients (n = 400, mean age 72 yrs) with OAB randomized (2: 1) • 12 wks, double-blind darifenacin (7. 5 mg OD for 2 wks, then optional titration to 15 mg daily) or placebo • Marked improvements in OAB symptoms can be achieved in patients >or= 65 yrs with significant treatment differences in micturition frequency • Qo. L appears to be sensitive & relevant patient-oriented measure of treatment effect Chapple et al: Curr Med Res Opin. 2007; 23(10): 2347 -58.
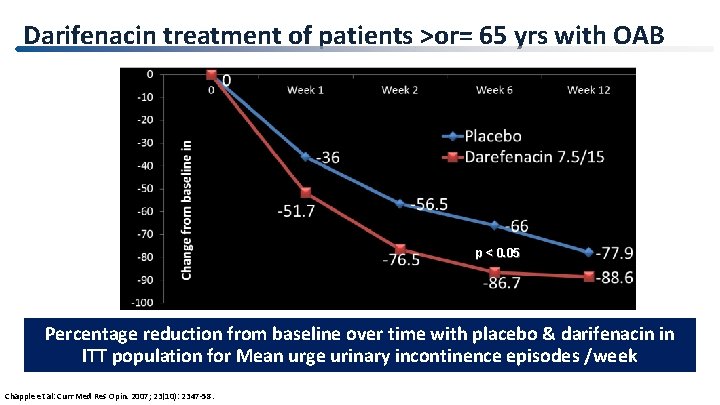
Darifenacin treatment of patients >or= 65 yrs with OAB p < 0. 05 Percentage reduction from baseline over time with placebo & darifenacin in ITT population for Mean urge urinary incontinence episodes /week Chapple et al: Curr Med Res Opin. 2007; 23(10): 2347 -58.
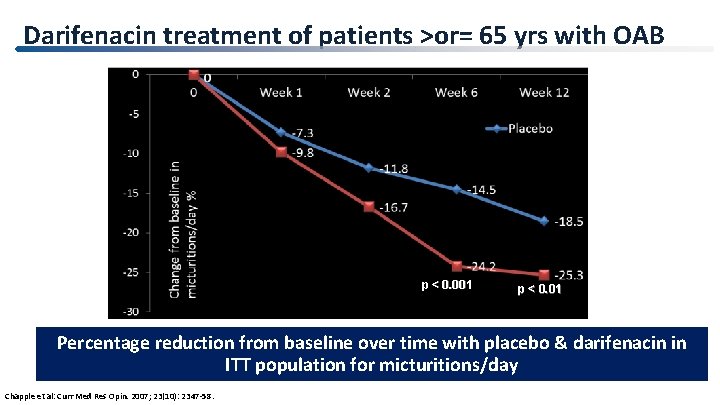
Darifenacin treatment of patients >or= 65 yrs with OAB p < 0. 001 p < 0. 01 Percentage reduction from baseline over time with placebo & darifenacin in ITT population for micturitions/day Chapple et al: Curr Med Res Opin. 2007; 23(10): 2347 -58.

Darifenacin treatment of patients >or= 65 yrs with OAB 0 -10 -20 -30 -40 -50 -60 -70 Change from baseline in urgency episodes/day % 0 0 Week 1 Week 2 Week 6 -22 Week 12 Placebo Darefenacin 7. 5/15 -30. 5 -39. 6 -42. 9 -41 -55. 5 -62. 7 p < 0. 001 p < 0. 01 -69. 6 -80 Percentage reduction from baseline over time with placebo & darifenacin in ITT population for urinary urgency episodes/day ITT population for urinary Chapple et al: Curr Med Res Opin. 2007; 23(10): 2347 -58.

Safety and tolerability • During clinical development: 7, 363 pts & volunteers (doses: 3. 75 to 75 mg OD) • Safety evaluated: Phase II & III trials in 8, 830 pts; 6, 001 with Darifenacin • Of this total 1, 069 pts: In 3 Phase III, 12 -wk, fixed-dose efficacy & safety trials • All long-term trials combined: 1, 216 & 672 pts for at least 24 & 52 wks, resp. • All placebo-controlled trials combined: Incidence of serious AEs for 7. 5 mg, 15 mg & placebo similar BJU Int. 2006 Nov; 98(5): 1025 -32/BJU Int. 2005 Nov; 96(7): 1055 -62/ J Clin Pharmacol. 2005 Sep; 45(9): 1038 -47/ J Urol. 2005 Feb; 173(2): 493 -8/ Eur Urol. 2004 Apr; 45(4): 420 -9.

Safety and tolerability • All fixed-dose Phase III studies combined: 3. 3% of pts discontinued due to all AEs vs. 2. 6% in placebo • Most frequently reported AEs: Dry mouth & constipation • Majority AEs: Mild or moderate in severity & most occurred during 1 st 2 wks of treatment • No significant CNS (primarily M 1 -receptor mediated) AEs or cardiac (primarily M 2 -receptor mediated) AEs • Does not affect cognitive, cardiac or visual function • Does not prolong QT/QTc interval BJU Int. 2006 Nov; 98(5): 1025 -32/BJU Int. 2005 Nov; 96(7): 1055 -62/ J Clin Pharmacol. 2005 Sep; 45(9): 1038 -47/ J Urol. 2005 Feb; 173(2): 493 -8/ Eur Urol. 2004 Apr; 45(4): 420 -9.

Darifenacin: Other benefits • mean, median & minimum warning time compared with placebo, allowing subjects more time to reach toilet & potentially avoiding embarrassing experience of incontinence – Cardozo L, et al. J Urol. 2005 Apr; 173(4): 1214 -8. • Darifenacin: improved tolerability compared to oxybutynin • Zinner N, et al. World J Urol. 2005 Sep; 23(4): 248 -52. • Long-term treatment: associated with significant & clinically meaningful improvements in HRQo. L for patients with 'wet' OAB over 2 yrs • Dwyer P, et al. Neurourol Urodyn. 2008; 27(6): 540 -7.
Summary of darifenacin Selectivity • 59 fold higher selectivity for M 3 receptors • M 1/M 2 sparing profile • Low incidence of dry mouth & constipation Efficacy Tolerability • Significant • No. of incontinence episodes • No. & severity of urgency • Voiding frequency • Nocturnal awakenings • Proven long term safety & tolerability profile (2 yr) • Improvement in PPBC scores & OAB symptoms in patients previously treated with oxybutynin & tolterodine 56

57
- Slides: 57