Dal Mito alla Realt 31 Gennaio 2 Febbraio

Dal Mito alla Realtà 31 Gennaio – 2 Febbraio 2008 Atahotel Executive Milano Usare i markers della flogosi non migliora molto la terapia dell’asma Renato Cutrera Dir. U. O. C. Broncopneumologia Dipartimento Medicina Pediatrica (Prof. A. G. Ugazio) Ospedale Pediatrico Bambino Gesù IRCCS - Roma R. Cutrera, Milano, 2008

Domande che mi farei? E’ utile misurare l’infiammazione delle vie aeree in tutti i bambini con asma? E’ utile misurare l’infiammazione delle vie aeree routinariamente in tutti i bambini con asma? Tutti i pediatri che curano un bambino asmatico dovrebbero possedere la tecnologia per misurare l’infiammazione delle vie aeree? Ogni centro specialistico che segue bambini asmatici dovrebbe possedere la tecnologia per misurare l’infiammazione delle vie aeree? R. Cutrera, Milano, 2008

Asma bronchiale: definizione L’asma è una malattia infiammatoria cronica delle vie aeree caratterizzata da: • Episodi ricorrenti di dispnea, respiro sibilante, tosse e senso di costrizione toracica • Ostruzione bronchiale (di solito reversibile spontaneamente o dopo trattamento farmacologico) • Iperreattività bronchiale • Infiltrazione di cellule infiammatorie, rilascio di mediatori e rimodellamento strutturale delle vie aeree R. Cutrera, Milano, 2008
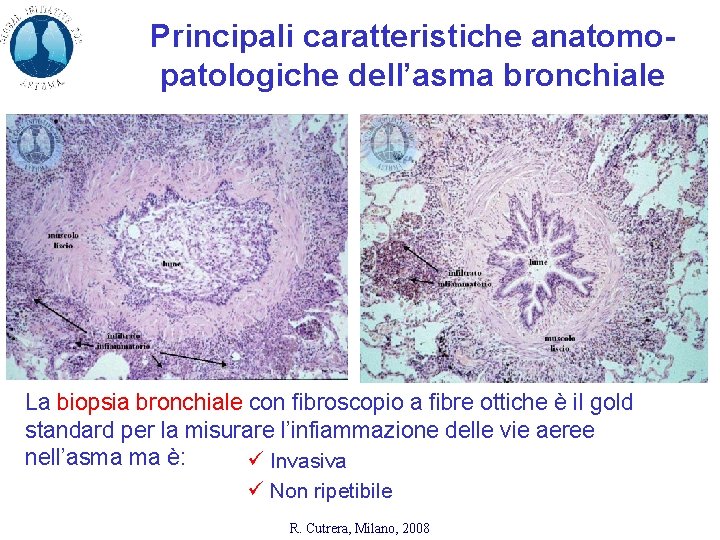
Principali caratteristiche anatomopatologiche dell’asma bronchiale La biopsia bronchiale con fibroscopio a fibre ottiche è il gold standard per la misurare l’infiammazione delle vie aeree nell’asma ma è: ü Invasiva ü Non ripetibile R. Cutrera, Milano, 2008

Altri metodi di analisi Challenge all’istamina o alla metacolina ü Interpretazione confusa dall’uso di broncodilatatori ü Difficile da attuare in pazienti gravi e bambini Metodo dell’espettorato indotto ü Abbastanza fastidioso ü Difficile da attuare in pazienti gravi e bambini ü Ripetibile non prima di 24 ore R. Cutrera, Milano, 2008

Markers dell’infiammazione nell’aria espirata ü Metodica non invasiva ü Facile da misurare ü Istantanea e ripetibile ü Attuabile in pazienti gravi e bambini ü Supera il concetto di sintomo ü Utile nella diagnosi differenziale ü Valuta la gravità della malattia ü Valuta la risposta al trattamento R. Cutrera, Milano, 2008

Markers dell’infiammazione nell’aria espirata üOssido Nitrico (NO) ü Monossido di carbonio (CO) ü Idrocarburi esalati ü Breath-condensate R. Cutrera, Milano, 2008
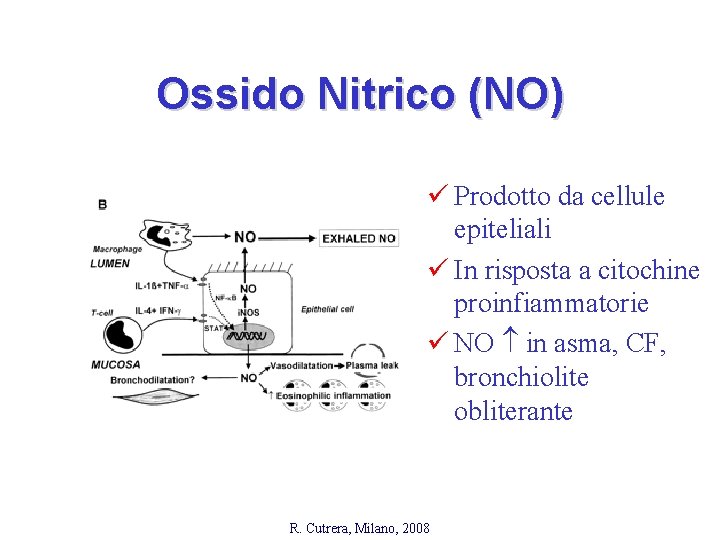
Ossido Nitrico (NO) ü Prodotto da cellule epiteliali ü In risposta a citochine proinfiammatorie ü NO in asma, CF, bronchiolite obliterante R. Cutrera, Milano, 2008
FUNZIONALITA’ RESPIRATORIA 4 Ossido d’Azoto (NO) marcatore di infiammazione R. Cutrera, Milano, 2008

Cosa sappiamo del Fe. NO nell’asma ü È correlato a infiammazione eosinofilica (Payne, AJRCCM 2001) ü È elevato nell’asma atopico (Alving, Eur Resp J, 1993) ü È ridotto da ICS (Kharitonov, Lancet 1994) ü È utile nel decidere a quali pazienti iniziare ICS (Smith, AJRCCM 2005) ü È utile nel decidere quando interrompere ICS (Pijnenburg, Thorax 2005) R. Cutrera, Milano, 2008

Exhaled nitric oxide measurements: clinical application and interpretation. Taylor, D R et al. Thorax 2006; 61: 817 -827 R. Cutrera, Milano, 2008

Exhaled nitric oxide measurements: clinical application and interpretation. Taylor, D R et al. Thorax 2006; 61: 817 -827 R. Cutrera, Milano, 2008

Ma abbiamo bisogno del Fe. NO come un infiammometro? Misurare spesso il Fe. NO con gli obbiettivi di: ü Predire e diminuire le riacutizzazioni ü Ottimizzare (diminuire) la dose di ICS ü Migliorare il calibro bronchiale? R. Cutrera, Milano, 2008

Use of Exhaled Nitric Oxide Measurements to Guide Treatment in Chronic Asthma Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DR- N Engl J Med 2005; 352: 2163– 2173. Single-blind, placebo-controlled trial 97 patients (12 -75 yrs) with chronic asthma Primary care setting Regularly receiving treatment with inhaled corticosteroids for 6 months or more Corticosteroid dose adjusted, in a stepwise fashion, on the basis of either Fe. NO measurements or an algorithm based on conventional guidelines (GINA) Use of long acting beta 2 agonist was discontinued Primary outcome: frequency of asthma exacerbations Secondary outcome: mean daily dose of inhaled steroid. R. Cutrera, Milano, 2008

Use of Exhaled Nitric Oxide Measurements to Guide Treatment in Chronic Asthma Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DR- N Engl J Med 2005; 352: 2163– 2173. R. Cutrera, Milano, 2008

Use of Exhaled Nitric Oxide Measurements to Guide Treatment in Chronic Asthma Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DR- N Engl J Med 2005; 352: 2163– 2173. R. Cutrera, Milano, 2008
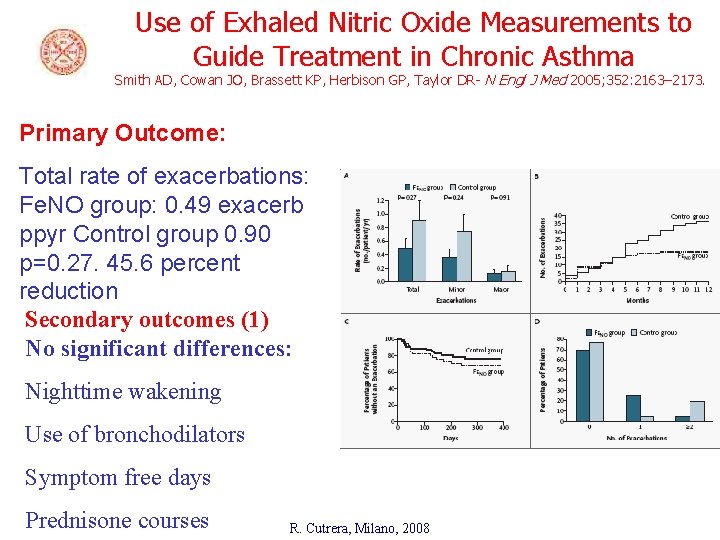
Use of Exhaled Nitric Oxide Measurements to Guide Treatment in Chronic Asthma Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DR- N Engl J Med 2005; 352: 2163– 2173. Primary Outcome: Total rate of exacerbations: Fe. NO group: 0. 49 exacerb ppyr Control group 0. 90 p=0. 27. 45. 6 percent reduction Secondary outcomes (1) No significant differences: Nighttime wakening Use of bronchodilators Symptom free days Prednisone courses R. Cutrera, Milano, 2008
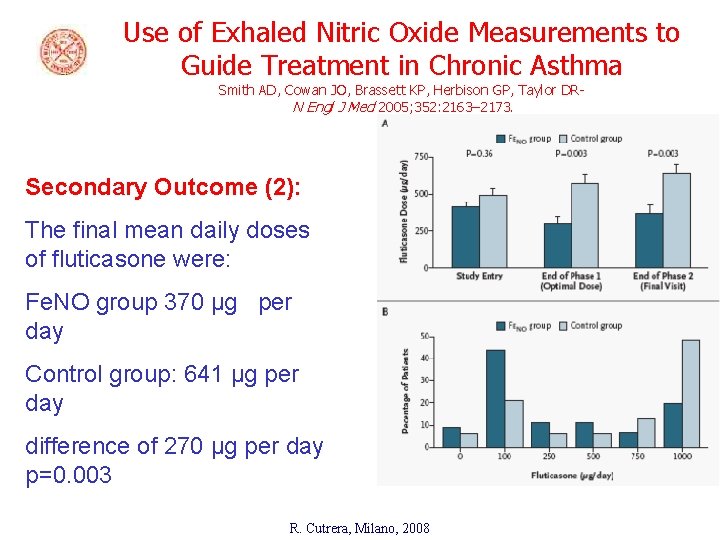
Use of Exhaled Nitric Oxide Measurements to Guide Treatment in Chronic Asthma Smith AD, Cowan JO, Brassett KP, Herbison GP, Taylor DRN Engl J Med 2005; 352: 2163– 2173. Secondary Outcome (2): The final mean daily doses of fluticasone were: Fe. NO group 370 μg per day Control group: 641 μg per day difference of 270 μg per day p=0. 003 R. Cutrera, Milano, 2008

Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. DBRCT tertiary care setting 85 children (6 -18 yrs) with atopic asthma, using inhaled steroids for 3 months or more, were allocated to: FENO group (n 39) in which treatment decisions were made on both FENO and symptoms, Symptom group (n 46) treated on symptoms only. Children were seen every 3 months over a 1 -year period. Beta 2 long acting permitted R. Cutrera, Milano, 2008

Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. Primary endpoint: Cumulative steroid dose Secondary endpoints: Mean daily symptom score Bronchodilator use Symptoms free days (last 4 weeks) Oral prednisone courses PD 20 at final visit FVC, FEV 1, MEF 25 at final visit R. Cutrera, Milano, 2008
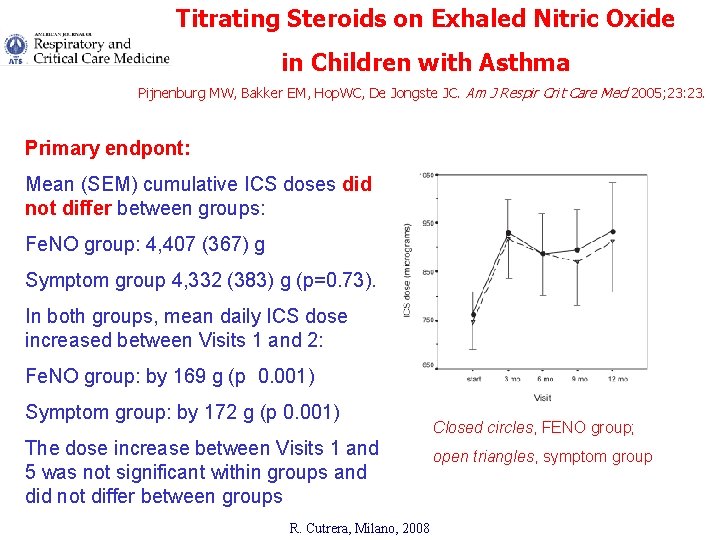
Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. Primary endpont: Mean (SEM) cumulative ICS doses did not differ between groups: Fe. NO group: 4, 407 (367) g Symptom group 4, 332 (383) g (p=0. 73). In both groups, mean daily ICS dose increased between Visits 1 and 2: Fe. NO group: by 169 g (p 0. 001) Symptom group: by 172 g (p 0. 001) The dose increase between Visits 1 and 5 was not significant within groups and did not differ between groups R. Cutrera, Milano, 2008 Closed circles, FENO group; open triangles, symptom group

Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. Miglioramento FEV 1 Secondary endponts: üNo differences in: Mean daily symptom score Bronchodilator use Symptoms free days (last 4 weeks) Oral prednisone courses üSignificant differences in: PD 20 FEV 1 Closed circles, FENO group; Fe. NO open triangles, symptom group R. Cutrera, Milano, 2008

Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. Miglioramento FEV 1 R. Cutrera, Milano, 2008 Miglioramento Fe. NO

Titrating Steroids on Exhaled Nitric Oxide in Children with Asthma Pijnenburg MW, Bakker EM, Hop. WC, De Jongste JC. Am J Respir Crit Care Med 2005; 23: 23. Conclusions: we have shown that a treatment algorithm using Fe. NO for ICS dose titration every 3 months for 1 year is superior to conventional treatment guided by symptoms, and leads to similar clinical asthma control and less airway hyperresponsiveness, obstruction, and inflammation with a similar ICS dose. Conclusioni (personali): Il primary endpoint dello studio (diminuzione dello steroide) è negativo I secondary endpoints clinici sono negativi I secondary endpoint funzionali sono significativi Utilizzando il Fe. NO non diminuiremo lo steroide necessario, i sintomi non miglioreranno, ma avremo meno infiammazione, ostruzione e BHR R. Cutrera, Milano, 2008
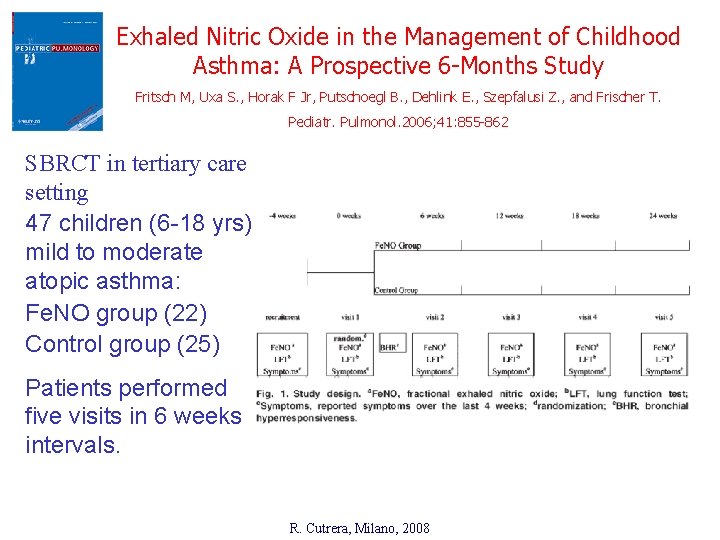
Exhaled Nitric Oxide in the Management of Childhood Asthma: A Prospective 6 -Months Study Fritsch M, Uxa S. , Horak F Jr, Putschoegl B. , Dehlink E. , Szepfalusi Z. , and Frischer T. Pediatr. Pulmonol. 2006; 41: 855 -862 SBRCT in tertiary care setting 47 children (6 -18 yrs) mild to moderate atopic asthma: Fe. NO group (22) Control group (25) Patients performed five visits in 6 weeks intervals. R. Cutrera, Milano, 2008
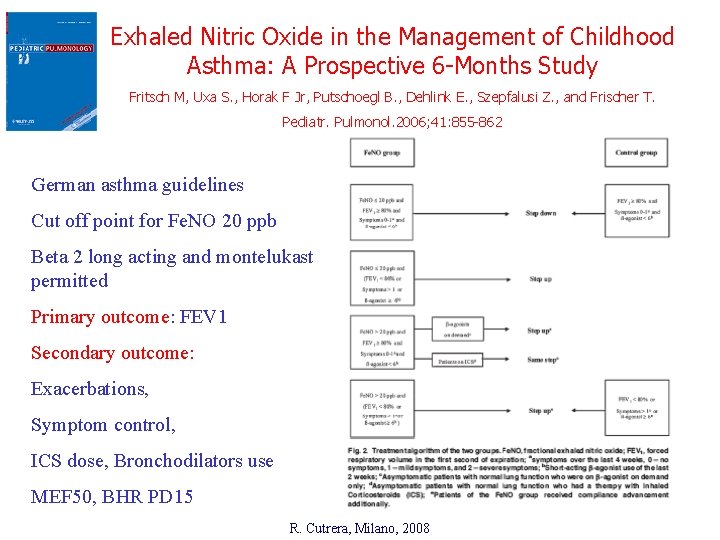
Exhaled Nitric Oxide in the Management of Childhood Asthma: A Prospective 6 -Months Study Fritsch M, Uxa S. , Horak F Jr, Putschoegl B. , Dehlink E. , Szepfalusi Z. , and Frischer T. Pediatr. Pulmonol. 2006; 41: 855 -862 German asthma guidelines Cut off point for Fe. NO 20 ppb Beta 2 long acting and montelukast permitted Primary outcome: FEV 1 Secondary outcome: Exacerbations, Symptom control, ICS dose, Bronchodilators use MEF 50, BHR PD 15 R. Cutrera, Milano, 2008
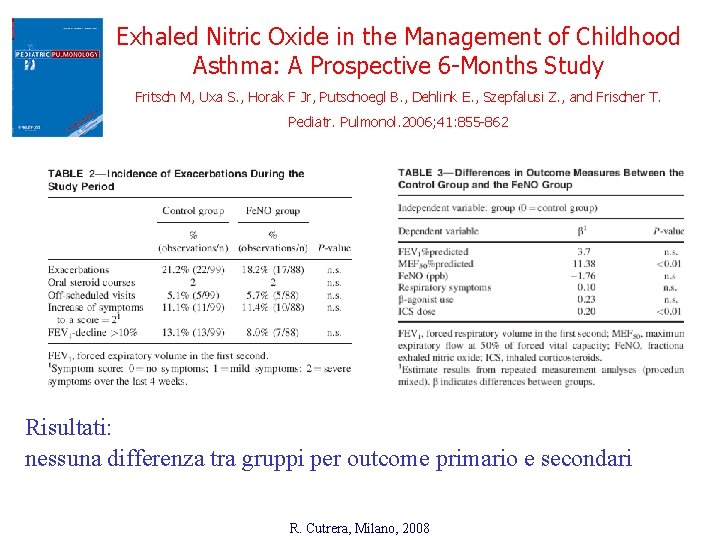
Exhaled Nitric Oxide in the Management of Childhood Asthma: A Prospective 6 -Months Study Fritsch M, Uxa S. , Horak F Jr, Putschoegl B. , Dehlink E. , Szepfalusi Z. , and Frischer T. Pediatr. Pulmonol. 2006; 41: 855 -862 Risultati: nessuna differenza tra gruppi per outcome primario e secondari R. Cutrera, Milano, 2008
The Use of Exhaled Nitric Oxide to Guide. Asthma Management: A Randomized Controlled Trial Shaw DE, . Berry MA, Thomas M, Green RH, Brightling CE, Wardlaw AJ, and Pavord ID Am J Respir Crit Care Med Vol 176. pp 231– 237, 2007 118 adults with a primary care diagnosis of asthma were randomized to a SBRCT of corticosteroid therapy based on: FENO measurements (n=58) BTS guidelines (n= 60). Assessed monthly for 4 months then every 2 months for a further 8 months. The primary outcomes: severe asthma exacerbations dosage of ICS (BDP equivalent) R. Cutrera, Milano, 2008

The Use of Exhaled Nitric Oxide to Guide. Asthma Management: A Randomized Controlled Trial Shaw DE, . Berry MA, Thomas M, Green RH, Brightling CE, Wardlaw AJ, and Pavord ID Am J Respir Crit Care Med Vol 176. pp 231– 237, 2007 In the Fe. NO group, treatment Asthma control: Juniper asthma control was adjusted following a set questionnaire (scores asthma control protocol according to both the from 0 to 6) Fe. NO and Juniper scores. score of greater than 1. 57 was used to If the Fe. NO was greater than 26 identify poorly controlled asthma. ppb, inhaled corticosteroid In the control group treatment was doubled if the score was more than 1. 57, and treatment was halved if the score was less than 1. 57 for 2 consecutive months treatment was increased; if the Fe. NO was less than 16 ppb or less than 26 ppb on two consecutive occasions, treatment was decreased. R. Cutrera, Milano, 2008
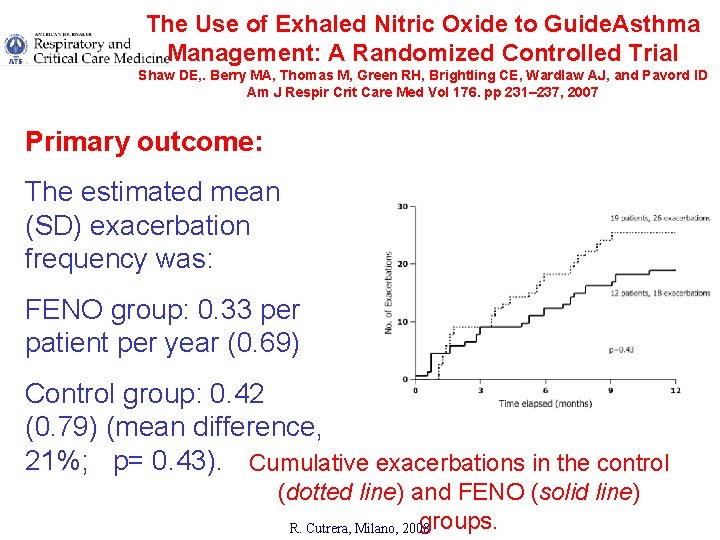
The Use of Exhaled Nitric Oxide to Guide. Asthma Management: A Randomized Controlled Trial Shaw DE, . Berry MA, Thomas M, Green RH, Brightling CE, Wardlaw AJ, and Pavord ID Am J Respir Crit Care Med Vol 176. pp 231– 237, 2007 Primary outcome: The estimated mean (SD) exacerbation frequency was: FENO group: 0. 33 per patient per year (0. 69) Control group: 0. 42 (0. 79) (mean difference, 21%; p= 0. 43). Cumulative exacerbations in the control (dotted line) and FENO (solid line) groups. R. Cutrera, Milano, 2008
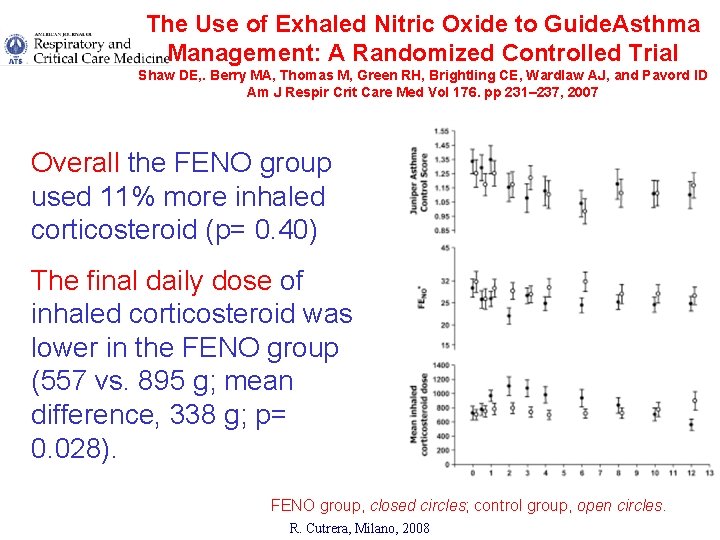
The Use of Exhaled Nitric Oxide to Guide. Asthma Management: A Randomized Controlled Trial Shaw DE, . Berry MA, Thomas M, Green RH, Brightling CE, Wardlaw AJ, and Pavord ID Am J Respir Crit Care Med Vol 176. pp 231– 237, 2007 Overall the FENO group used 11% more inhaled corticosteroid (p= 0. 40) The final daily dose of inhaled corticosteroid was lower in the FENO group (557 vs. 895 g; mean difference, 338 g; p= 0. 028). FENO group, closed circles; control group, open circles. R. Cutrera, Milano, 2008

The Use of Exhaled Nitric Oxide to Guide. Asthma Management: A Randomized Controlled Trial Shaw DE, . Berry MA, Thomas M, Green RH, Brightling CE, Wardlaw AJ, and Pavord ID Am J Respir Crit Care Med Vol 176. pp 231– 237, 2007 Conclusions: An asthma treatment strategy based on the measurement of Fe. NO did not result in a large reduction in asthma exacerbations or in the total amount of inhaled corticosteroid therapy used over 12 mo, when compared with current asthma guidelines. R. Cutrera, Milano, 2008
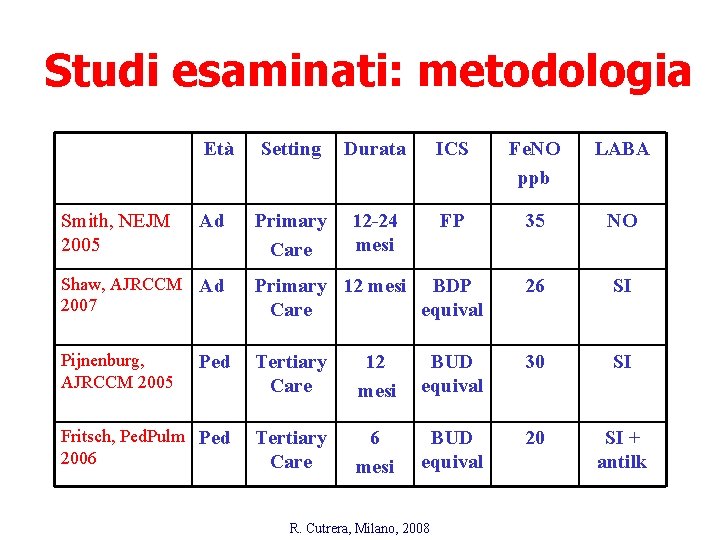
Studi esaminati: metodologia Età Smith, NEJM 2005 Ad Setting Durata ICS Fe. NO ppb LABA Primary Care 12 -24 mesi FP 35 NO Shaw, AJRCCM Ad 2007 Primary 12 mesi BDP Care equival 26 SI Pijnenburg, AJRCCM 2005 Ped Tertiary Care 12 mesi BUD equival 30 SI Fritsch, Ped. Pulm Ped 2006 Tertiary Care 6 mesi BUD equival 20 SI + antilk R. Cutrera, Milano, 2008
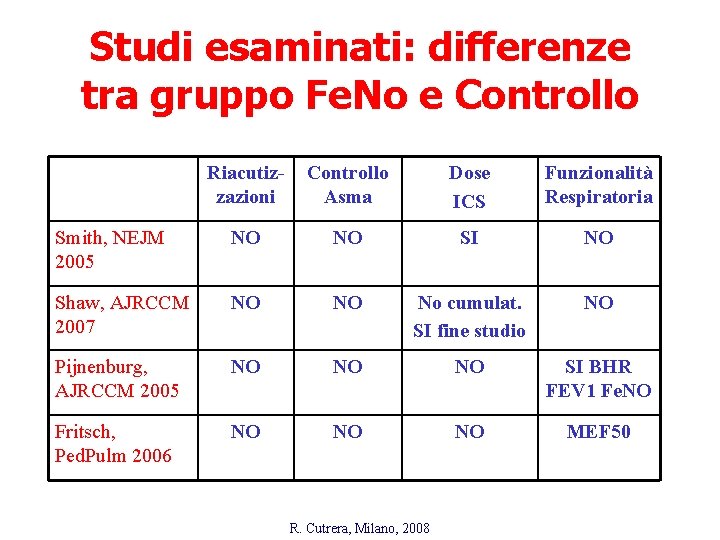
Studi esaminati: differenze tra gruppo Fe. No e Controllo Riacutizzazioni Controllo Asma Dose ICS Funzionalità Respiratoria Smith, NEJM 2005 NO NO SI NO Shaw, AJRCCM 2007 NO NO No cumulat. SI fine studio NO Pijnenburg, AJRCCM 2005 NO NO NO SI BHR FEV 1 Fe. NO Fritsch, Ped. Pulm 2006 NO NO NO MEF 50 R. Cutrera, Milano, 2008

Problemi aperti ü ü Differenti fenotipi di asma Algoritmi di trattamento differenti Utilizzo di farmaci concomitanti Valori di normalità del Fe. No (età altezza, inquinamento atmosferico) ü Cut point “patologico” su cui agire ü Correlazione tra Fe. No e eosinofili bronchiali ü Coesistenza di infiammazione eosinofila e neutrofila nello stesso momento R. Cutrera, Milano, 2008

Risposte che provo a dare E’ utile misurare l’infiammazione delle Si vie aeree? E’ utile misurare l’infiammazione delle vie aeree in tutti i bambini con asma? Si, almeno una volta, e sempre nelle asma che non rispondono alla terapia E’ utile misurare l’infiammazione delle vie aeree routinariamente in tutti i bambini con asma? No allo stato attuale delle conoscenze Tutti i pediatri che curano un bambino asmatico dovrebbero possedere la tecnologia per misurare l’infiammazione delle vie aeree? No Ogni centro specialistico che segue bambini asmatici dovrebbe possedere la tecnologia per misurare l’infiammazione delle vie aeree? Si R. Cutrera, Milano, 2008
- Slides: 36