DAL LABORATORIO ALLA DIAGNOSI Daniela Maria Cirillo MD

DAL LABORATORIO ALLA DIAGNOSI Daniela Maria Cirillo, MD, Ph. D WHO Collaborating Centre for TB Laboratory Strengtherning Supranational Reference Laboratory Milan Emerging Bacterial Pathogens Unit, San Raffaele Scientific Institute, Milan

SOMMARIO • • • INTRODUZIONE – La Diagnosi di Tubercolosi: un problema non risolto – NUOVI TEST PER MDR TB RACCOMANDATI DAL WHO Ruolo delle MIC nella pratica clinica Whole Genome Sequencing PER LA DIAGNOSI E SORVEGLIANZA DI MDRTB ANALISI MOLECOLARE PER INDIRIZZARE UN TRATTAMENTO APPROPRIATO Whole Genome Sequencing PER L’IDENTIFICAZIONE DELLE TRASMISSIONI CONCLUSIONI
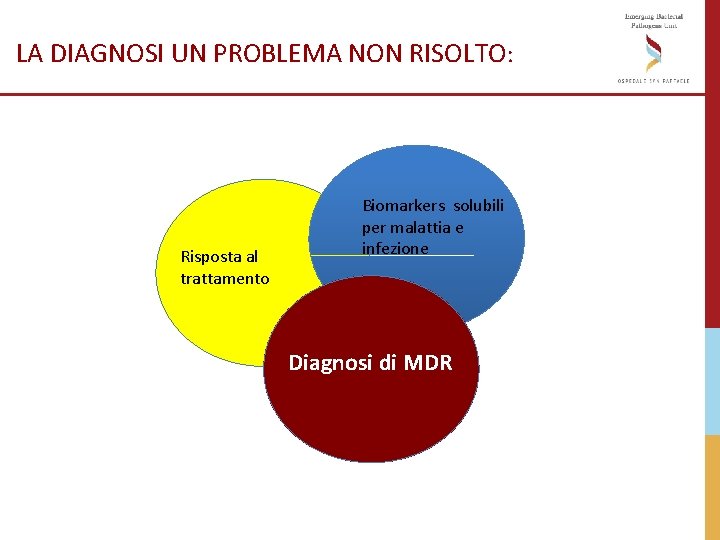
LA DIAGNOSI UN PROBLEMA NON RISOLTO: Risposta al trattamento Biomarkers solubili per malattia e infezione Diagnosi di MDR

LA DIAGNOSI DI MDRTB UN PROBLEMA NON RISOLTO: MDR-TB: necessita di diagnosi rapida e trattamento lungo e tossico MDR-TB è trasmessa in comunità La microbiologia convenzionale ha tempi incompatibili con la necessità di indirizzare il trattamento rapidamente Sono disponibili nuovi farmaci/regimi terapeutici e il “gap” rimane nel loro utilizzo corretto

MDR TB: DIAGNOSI E TRATTAMENTO TB incidence DS-TB MDR-TB XDR-TB WHO, Global TB Strategy 2015.

FENOTIPO O GENOTIPO? FENOTIPO • Lunghi tempi di crescita • Strumentazione, training, BSL 3 • Concentrazioni Critiche spesso non corrette GENOTIPO • Rapido • BSL 2 o BSL 1 • Completamente automatizzabile • Sensibilità?

DIAGNOSTICI MOLECOLARI RACCOMANDATI DA OMS Xpert ULTRA MTBDRplus • Integrated/automated q. PCR • Fully nested amplification • Results in 1 h 17 m • Technical expertise: none • TB detection by targeting two different multi-copy genes (IS 6110 & IS 1081) • High-resolution melt curve analysis for rpo. B detection • Additional PCR modifications to improve sensitivity and specificity • Reverse hybridization, colorimetric reaction • Results in 6 -7 h • Technical expertise: some • Biosafety lev 2 • Detect resistance in TB to Rifampicine (Rpo. B gene) and Isoniazide (Inh. A, Kat. G genes), MTBDRsl • Reverse hybridization, colorimetric reaction • Results in 6 -7 h • Technical expertise: some • Biosafety lev 2 • Detect resistance in TB to the fluoroquinolones (FQs; ofloxacin, moxifloxacin and levofloxacin) and the secondline injectable drugs (SLIDs; amikacin, kanamycin and capreomycin),

A standardised method for interpreting the association between mutations and phenotypic drug resistance in Mycobacterium tuberculosis Miotto, ERJ 2017
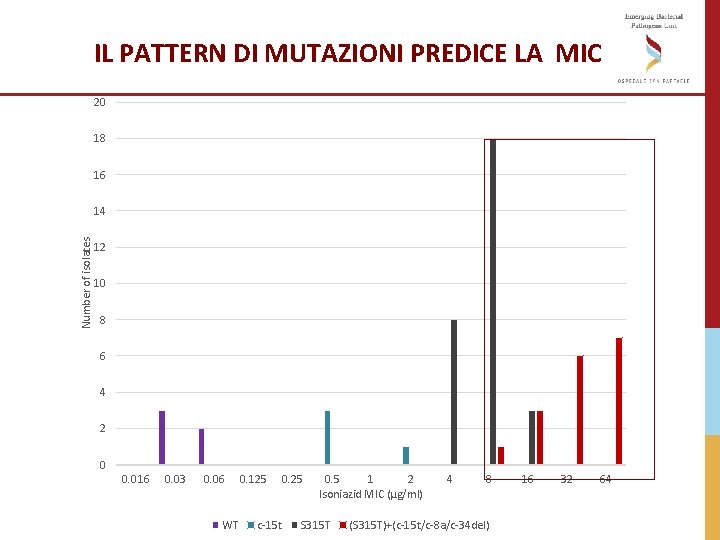
IL PATTERN DI MUTAZIONI PREDICE LA MIC 20 18 16 Number of isolates 14 12 10 8 6 4 2 0 0. 016 0. 03 0. 06 WT 0. 125 c-15 t 0. 25 0. 5 1 2 Isoniazid MIC (µg/ml) S 315 T 4 8 (S 315 T)+(c-15 t/c-8 a/c-34 del) 16 32 64
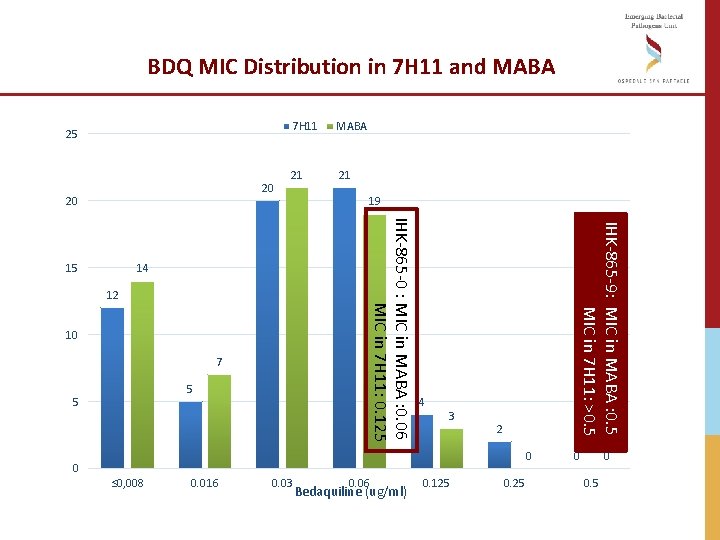
BDQ MIC Distribution in 7 H 11 and MABA 25 20 20 MABA 21 21 12 10 7 5 4 3 IHK-865 -9: MIC in MABA : 0. 5 MIC in 7 H 11: >0. 5 14 5 19 IHK-865 -0 : MIC in MABA : 0. 06 MIC in 7 H 11: 0. 125 15 7 H 11 2 0 0 ≤ 0, 008 0. 016 0. 03 0. 06 Bedaquiline (ug/ml) 0. 125 0 0 0. 5
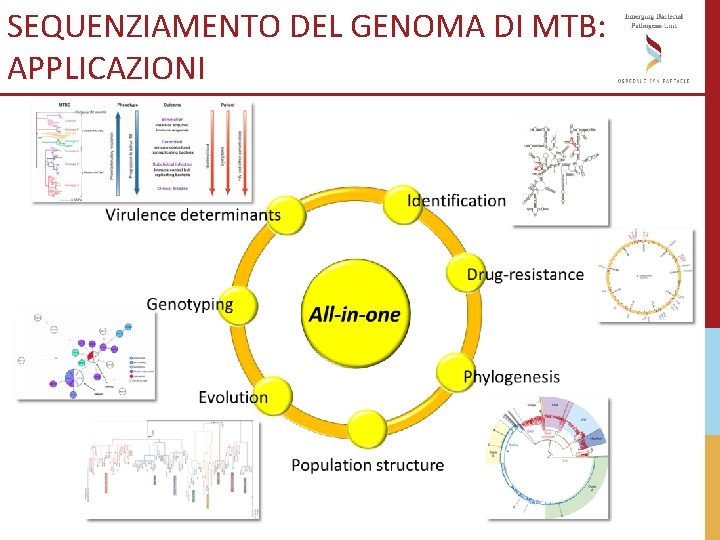
SEQUENZIAMENTO DEL GENOMA DI MTB: APPLICAZIONI

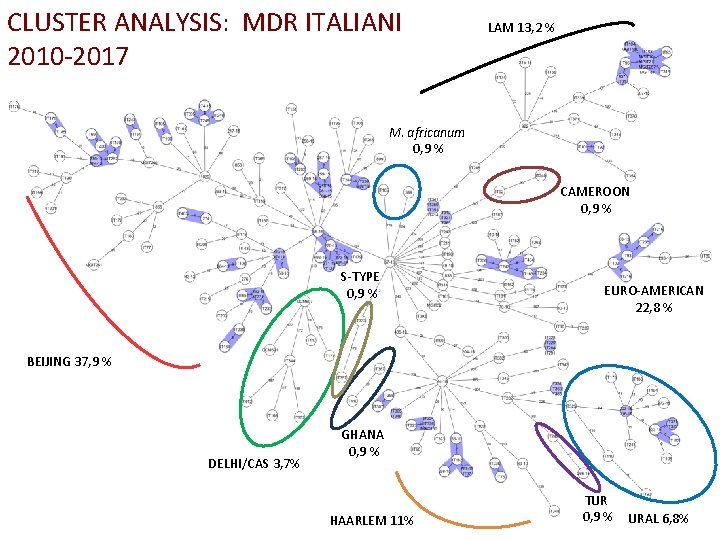
CLUSTER ANALYSIS: MDR ITALIANI 2010 -2017 LAM 13, 2 % M. africanum 0, 9 % CAMEROON 0, 9 % S-TYPE 0, 9 % EURO-AMERICAN 22, 8 % BEIJING 37, 9 % DELHI/CAS 3, 7% GHANA 0, 9 % HAARLEM 11% TUR 0, 9 % URAL 6, 8%
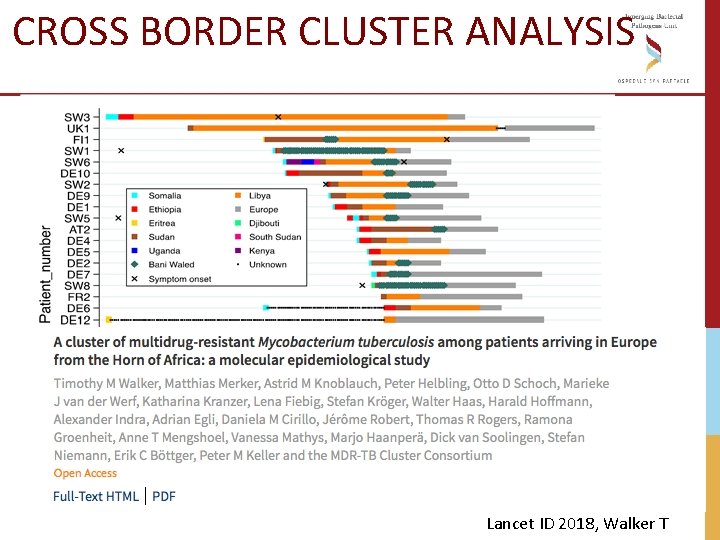
CROSS BORDER CLUSTER ANALYSIS 2016 Milano 2017 Lamezia Terme Lancet ID 2018, Walker T

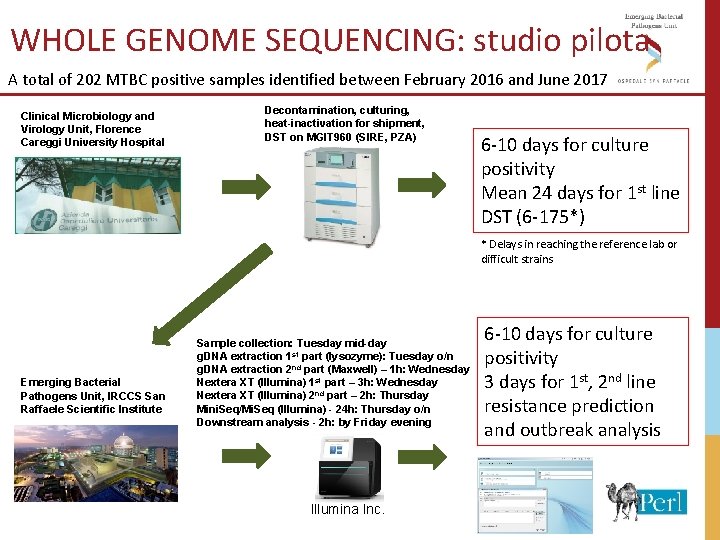
WHOLE GENOME SEQUENCING: studio pilota A total of 202 MTBC positive samples identified between February 2016 and June 2017 Clinical Microbiology and Virology Unit, Florence Careggi University Hospital Decontamination, culturing, heat-inactivation for shipment, DST on MGIT 960 (SIRE, PZA) 6 -10 days for culture positivity Mean 24 days for 1 st line DST (6 -175*) * Delays in reaching the reference lab or difficult strains Emerging Bacterial Pathogens Unit, IRCCS San Raffaele Scientific Institute Sample collection: Tuesday mid-day g. DNA extraction 1 st part (lysozyme): Tuesday o/n g. DNA extraction 2 nd part (Maxwell) – 1 h: Wednesday Nextera XT (Illumina) 1 st part – 3 h: Wednesday Nextera XT (Illumina) 2 nd part – 2 h: Thursday Mini. Seq/Mi. Seq (Illumina) - 24 h: Thursday o/n Downstream analysis - 2 h: by Friday evening Illumina Inc. 6 -10 days for culture positivity 3 days for 1 st, 2 nd line resistance prediction and outbreak analysis
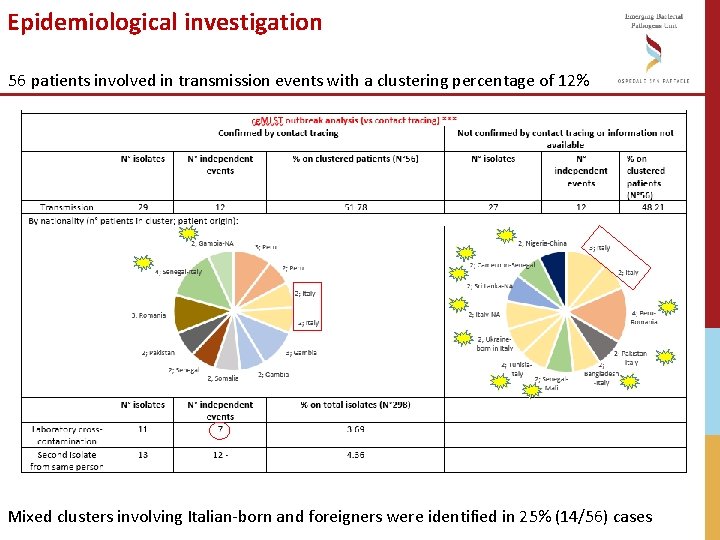
Epidemiological investigation 56 patients involved in transmission events with a clustering percentage of 12% Mixed clusters involving Italian-born and foreigners were identified in 25% (14/56) cases
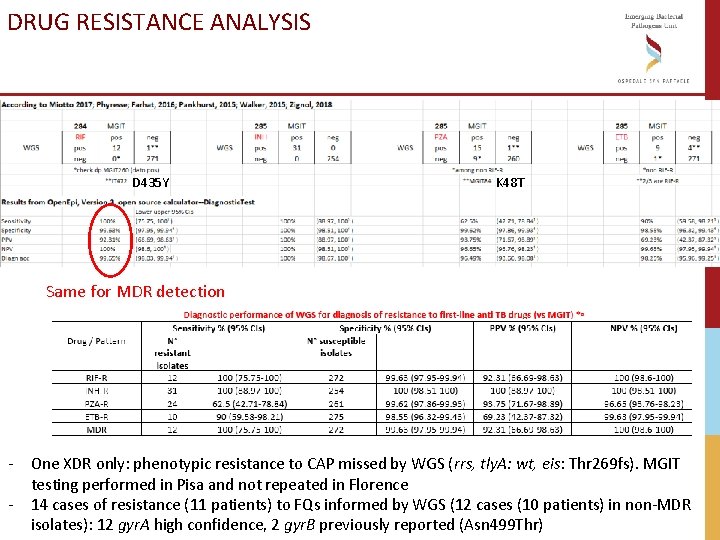
DRUG RESISTANCE ANALYSIS D 435 Y K 48 T Same for MDR detection - One XDR only: phenotypic resistance to CAP missed by WGS (rrs, tly. A: wt, eis: Thr 269 fs). MGIT testing performed in Pisa and not repeated in Florence - 14 cases of resistance (11 patients) to FQs informed by WGS (12 cases (10 patients) in non-MDR isolates): 12 gyr. A high confidence, 2 gyr. B previously reported (Asn 499 Thr)
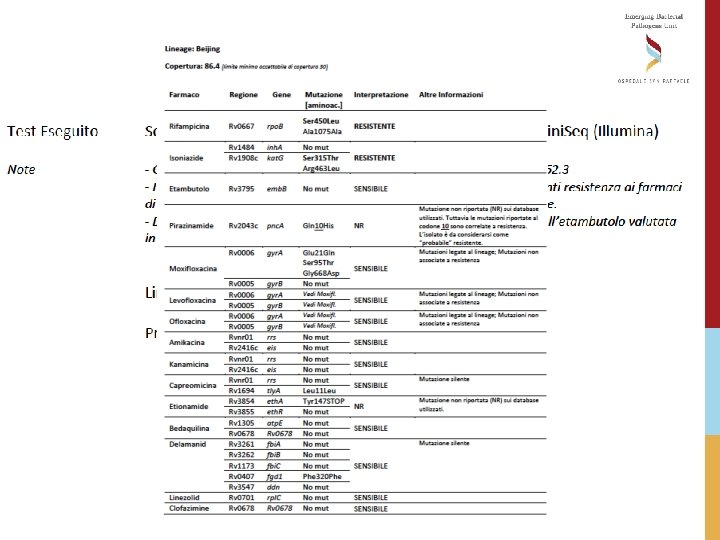
REPORTING WGS
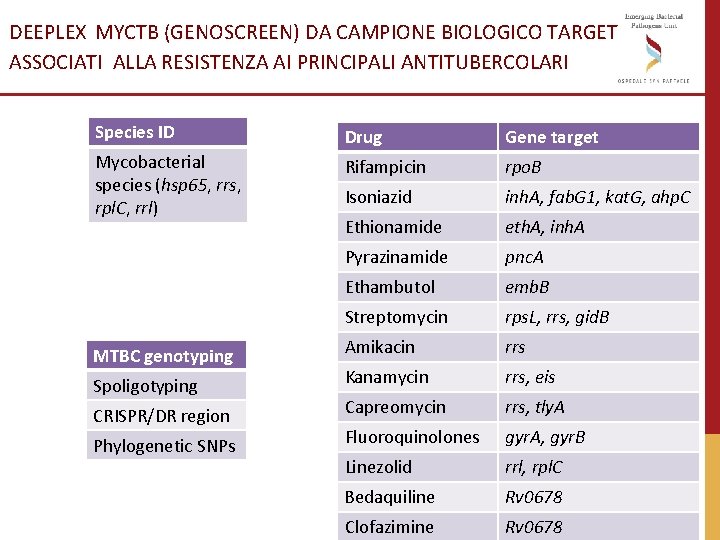
DEEPLEX MYCTB (GENOSCREEN) DA CAMPIONE BIOLOGICO TARGET ASSOCIATI ALLA RESISTENZA AI PRINCIPALI ANTITUBERCOLARI Species ID Drug Gene target Mycobacterial species (hsp 65, rrs, rpl. C, rrl) Rifampicin rpo. B Isoniazid inh. A, fab. G 1, kat. G, ahp. C Ethionamide eth. A, inh. A Pyrazinamide pnc. A Ethambutol emb. B Streptomycin rps. L, rrs, gid. B Amikacin rrs Kanamycin rrs, eis Capreomycin rrs, tly. A Fluoroquinolones gyr. A, gyr. B Linezolid rrl, rpl. C Bedaquiline Rv 0678 Clofazimine Rv 0678 MTBC genotyping Spoligotyping CRISPR/DR region Phylogenetic SNPs

CASO CLINICO • Bimba di 2 a in attesa di trapianto per ADA SCID con ascessi cutanei multipli agli arti ZN positivi • Padre con Mantoux 20 mm • Deeplex da materiale • Identificazione come BCG e profilo di resistenza

CONCLUSIONI • La sensibilità di alcuni test molecolari si sta avvicinando a quella della coltura liquida. • La disponibilità immediata di dati molecolari precisi indirizza il trattamento MDR • Piattaforme WGS based con analisi standardizzate dei dati permettono di ottenere dati utilizzabili per: – Ottimizzazione della terapia a livello individuale – Sorveglianza delle resistenze – Sorveglianza delle trasmissioni a livello locale, nazionale ed internazionale RETE DI SORVEGLIANZA MOLECOLARE NAZIONALE E INTERNAZIONALE

RINGRAZIAMENTI Riccardo Alagna, Lucia Barcellini Simone Battaglia Emanuele Borroni Andrea Cabibbe Matteo Chiacchiaretta Maria Rosaria De Filippo, Lucinda Furci, Arash Ghodousi, Floriana Gona, Giovanna Graziano, Riccardo Ligresti, Paola Mantegani Paolo Miotto Camilla Riva Marco Rossi Giovanna Stancanelli Elisa Tagliani Enrico Tortoli Alberto Trovato Emerging Bacterial Pathogens Unit Federica Cugnata Paola Rancoita Clelia di Serio CUSSB C. Boheme All SRL network C. Denkinger Antwerp, Stockholm, Amsterdam T. Rodwell Borstel, Gauting FIND OSR Genomic Center D. Goletti E Girardi IMNI A. Matteelli University of Brescia WHOCC TRADATE GB Migliori NDWG co-chair and core group And TF leaders A. Varga Consortium members New tools WG chair and co-chair WHO HQ Tbnet members. WHO EURO and other regional Offic ECDC S D’Amato Ministero della Salute L Fattorini ISS SMIRA L. Codecasa M Ferrarese E Mazzola Lombardia Regional TB center GM Rossolini and coll, Careggi , Florence S Niemann and coll, F. Drobniewsky, V. Nikolayesky D. Van Soolingen, A, ERand Anthony Partners experts from ERLN TB Network Re. Seq TB P. Supply University of Lille Cryptic Consortium members E-detect TBD. Crook consortium A. Gibertoni Cruz T. Walker

RINGRAZIAMENTI

Phenotypic DST: the reference standard • DST on solid medium: Löwenstein-Jensen; Middlebrook 7 H 10, 7 H 11 • DST on liquid medium: BACTEC MGIT 960; Versa. TREK; Middlebrook 7 H 9 • Noncommercial DST: nitrate reductase assay; microscopic observation DST; colorimetric redox indicator methods Adequate infrastructures and biosafety levels MDR-TB : 3 -6 weeks; XDR-TB : 6 -9 weeks
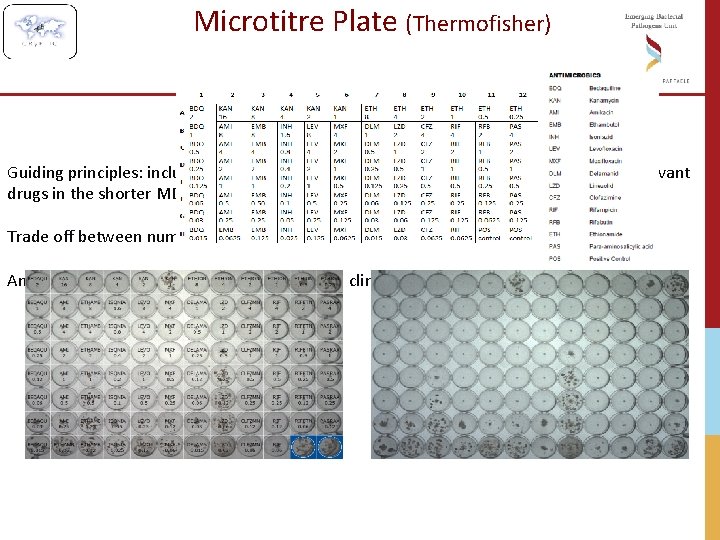
Microtitre Plate (Thermofisher) Guiding principles: inclusion of first-, second- and third-line drugs, repurposed drugs, relevant drugs in the shorter MDR-TB regimen; Trade off between number of drugs and concentration range; An attempt to accommodate needs from both clinical and research communities;
- Slides: 26