DAILY QUESTION September 22 2008 1 Name the

DAILY QUESTION September 22, 2008 1. Name the four states of matter. 2. Which state of matter is the most common in the universe?

Agenda 9/22/08 • Daily Question • Hand back Papers • Chapter 3 Section 1 Notes Assignments: 1. Ch 3 Section 1 Review Wksht due 9/23

Kinetic Theory page 70 • All matter is made of particles (atoms and molecules) • The particles are always in motion (↑ temp. = ↑ speed) • At the same temp. , smaller = faster

States of Matter page 71 - 72 Definite shape Solid Liquid Gas Plasma Shape changes Definite volume Volume changes Most common in Universe Broken particles

Energy page 73 Capacity to do work Kinetic energy = energy of motion Thermal energy = total kinetic energy of the particles ↑speed = ↑KE =↑thermal energy
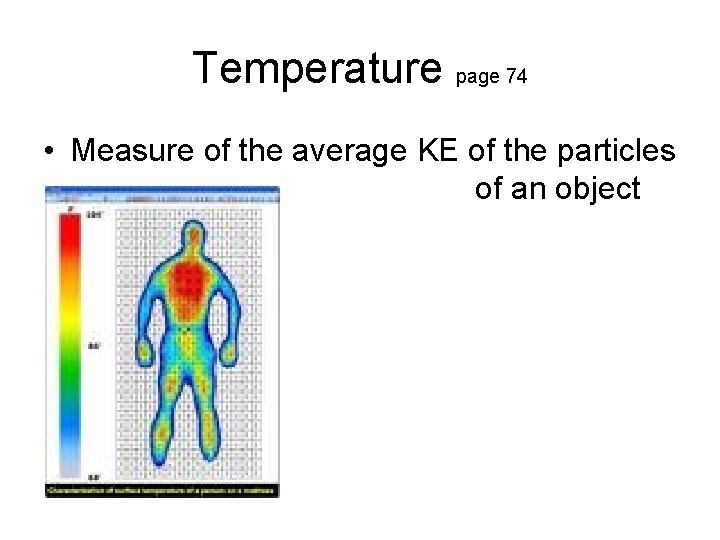
Temperature page 74 • Measure of the average KE of the particles of an object

Changes of State page 74 - 76 • Physical change • Energy of objects change • Transfer of energy is known as heat

Changes that require energy (Endothermic changes) page 75 • Evaporation – Liquid to gas – Usually occurs at the boiling point • Examples: water boiling, sweat on skin • Sublimation – Solid to gas • Examples: ice left in freezer, dry ice (solid CO 2)

Changes that release energy (Exothermic changes) page 76 • Condensation – Gas to liquid – Condensation point = temp at which gas becomes a liquid • Examples: water droplets on side of glass • Freezing – Liquid to solid – Freezing point = temp at which liquid becomes a solid • Examples: water as ice

Temp Change Vs. State Change page 76 • A temperature of a substance does not change during a change of state… – Example: If you add heat to ice at 0°C, the temperature will not rise until all the ice has melted.

Conservation of Mass and Energy page 77 • Mass cannot be created or destroyed • Reactants = Products – Example: Burning a match in closed system (Match + Oxygen = ash + smoke + gases) • Energy cannot be created or destroyed • Reactants = Products – Example: Moving Car (Gasoline = Energy to move car + Heat)
- Slides: 11