Daily Anti Retroviral Therapy II Once Daily Regimen


![PLASMA CONCENTRATION [ng/m. L] d 4 T XR 100 mg Once Daily Comparable Drug PLASMA CONCENTRATION [ng/m. L] d 4 T XR 100 mg Once Daily Comparable Drug](https://slidetodoc.com/presentation_image/b89db0a635fca514c4f87f26f0461f99/image-4.jpg)








- Slides: 19

Daily Anti. Retroviral Therapy II Once Daily Regimen for Treatment-naïve HIV+ Patients with Stavudine XR + Lamivudine + Efavirenz 24 Week Interim Efficacy and Safety Results D. Jayaweera 1, S. Becker 2, F. Felizarta 3, M. Sands 4, L. Slater 5 S. Gothelf 6, J. Maa 6, C. Dezii 6, S. Hodder 6, J. Tudor 6 University of Miami, FL, USA. Pacific Horizon Medical Group, San Francisco, CA, USA. 3 34 th Street Community Health Center, Bakersfield, CA, USA. 4 University of Florida, Jacksonville, FL, USA. 5 University of Oklahoma, Oklahoma City, OK, USA. 6 Bristol-Myers Squibb, Plainsboro, , NJ, USA. Plainsboro 1 2 DART II

AI 455 -096 and -099 Study Outlines ARV-naive patients Randomized Study 096 Study 099 d 4 T XR 100 mg QD d 4 T IR 40 mg BID (d 4 T IR placebo BID) 3 TC 150 mg BID EFV 600 mg QD (d 4 T XR placebo QD) 3 TC 150 mg BID EFV 600 mg QD n=74 n=392 n=76 n=392 Patients <60 kg: d 4 T XR 75 mg QD; d 4 T IR 30 mg BID NFV substitution permitted if EFV-intolerant DART II Adapted from: Brett-Smith H et al. 43 rd ICAAC, September, 2003. Poster H-843

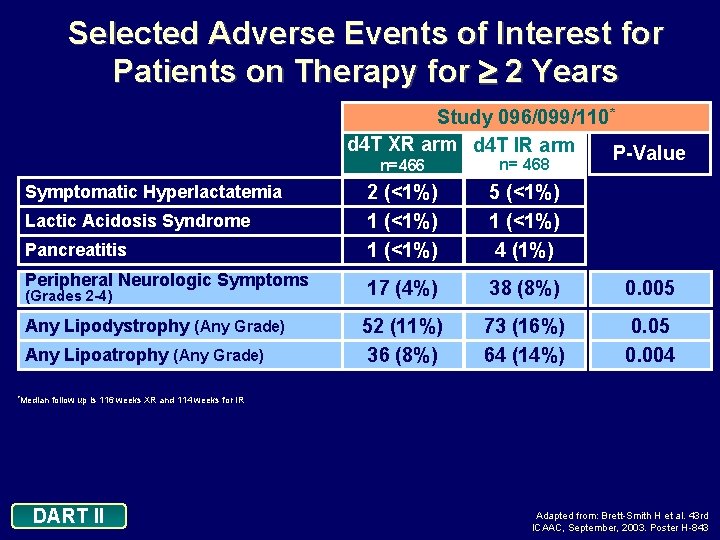
Selected Adverse Events of Interest for Patients on Therapy for 2 Years Study 096/099/110* d 4 T XR arm d 4 T IR arm P-Value Symptomatic Hyperlactatemia Lactic Acidosis Syndrome Pancreatitis Peripheral Neurologic Symptoms (Grades 2 -4) Any Lipodystrophy (Any Grade) Any Lipoatrophy (Any Grade) *Median n=466 n= 468 2 (<1%) 1 (<1%) 5 (<1%) 1 (<1%) 4 (1%) 17 (4%) 38 (8%) 0. 005 52 (11%) 36 (8%) 73 (16%) 64 (14%) 0. 05 0. 004 follow up is 116 weeks XR and 114 weeks for IR DART II Adapted from: Brett-Smith H et al. 43 rd ICAAC, September, 2003. Poster H-843
![PLASMA CONCENTRATION ngm L d 4 T XR 100 mg Once Daily Comparable Drug PLASMA CONCENTRATION [ng/m. L] d 4 T XR 100 mg Once Daily Comparable Drug](https://slidetodoc.com/presentation_image/b89db0a635fca514c4f87f26f0461f99/image-4.jpg)
PLASMA CONCENTRATION [ng/m. L] d 4 T XR 100 mg Once Daily Comparable Drug Exposure to d 4 T IR 40 mg BID Peak (Cmax) for d 4 T XR is ~50% of the IR formulation 1000 100 d 4 T XR formulation has 2 -3 times higher trough plasma levels than d 4 T IR 10 1 0 4 12 8 16 TIME [h] DART II d 4 T XR *Parallel d 4 T IR groups for XR and IR formulations in HIV patients 20 24

DART II: Study Design Prospective, once-daily, open-label, ARV-Naïve patients, VL > 1000 c/m. L, CD 4 > 100 Planned N= 70 d 4 T XR + 100 mg QD 3 TC 300 mg QD + EFV 600 mg QD Patients <60 kg: d 4 T XR 75 mg QD DART II Epivir® (lamivudine, 3 TC) is a registered trademark and manufactured by Glaxo. Smith. Kline

DART II: OBJECTIVES Primary Objective: • Efficacy of once daily d 4 T XR + 3 TC + EFV as determined by proportion of patients with plasma HIV-RNA <400 copies/m. L at 48 -weeks Secondary Objective: • Proportion of patients with plasma HIV-RNA <400 and <50 copies/m. L at Weeks 24, 48, 72, and 96 • Patient adherence using pill counts and Antiviral Medication Adherence Form (AMAF) • Safety and tolerability DART II

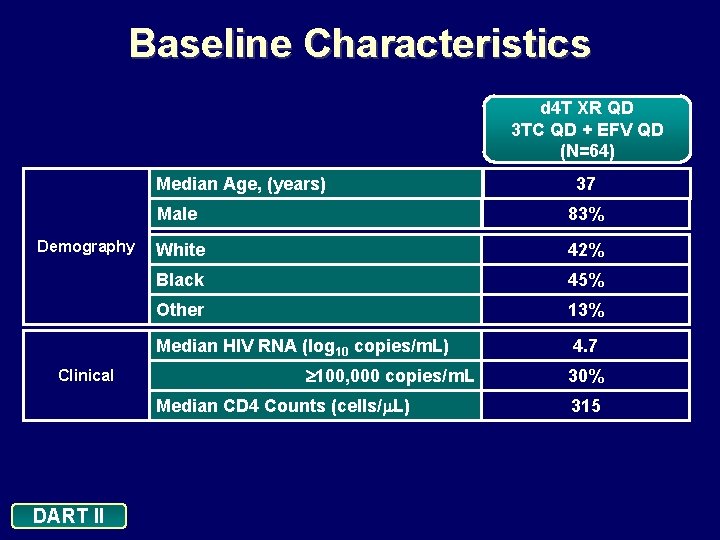
Baseline Characteristics d 4 T XR QD 3 TC QD + EFV QD (N=64) Median Age, (years) Demography Male 83% White 42% Black 45% Other 13% Median HIV RNA (log 10 copies/m. L) Clinical 100, 000 copies/m. L Median CD 4 Counts (cells/ L) DART II 37 4. 7 30% 315

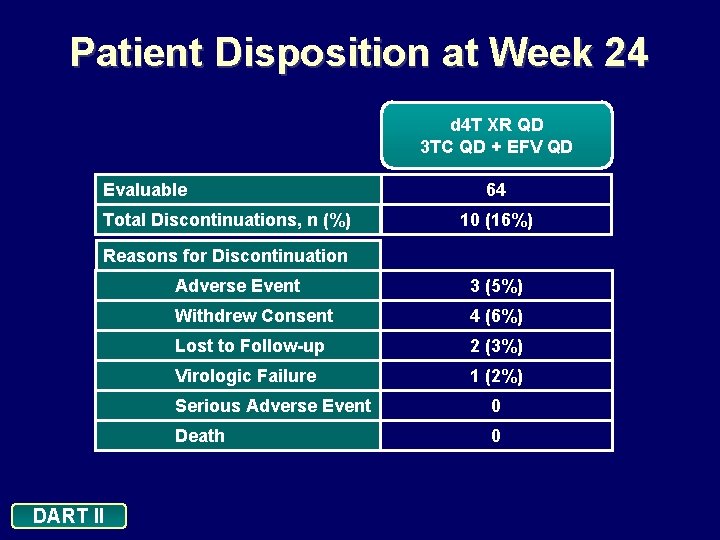
Patient Disposition at Week 24 d 4 T XR QD 3 TC QD + EFV QD Evaluable Total Discontinuations, n (%) 64 10 (16%) Reasons for Discontinuation DART II Adverse Event 3 (5%) Withdrew Consent 4 (6%) Lost to Follow-up 2 (3%) Virologic Failure 1 (2%) Serious Adverse Event 0 Death 0

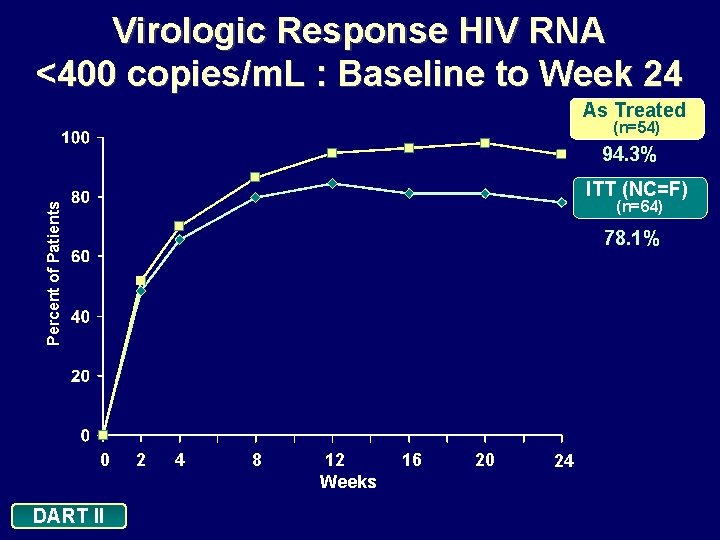
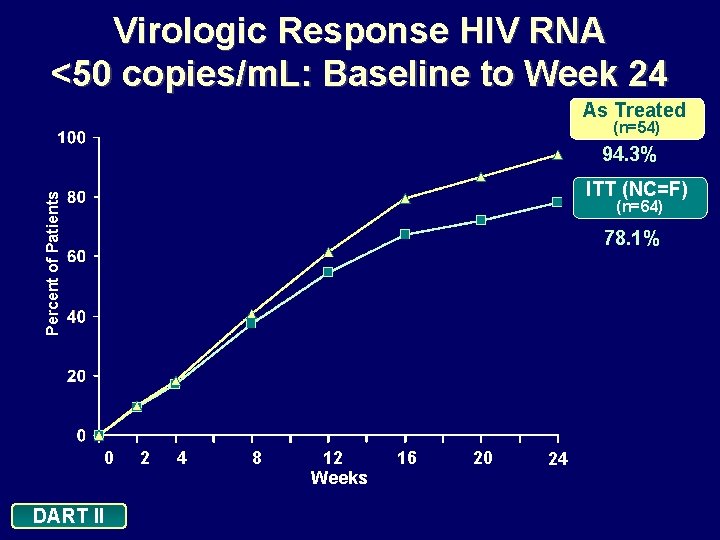
Virologic Response HIV RNA <400 copies/m. L : Baseline to Week 24 As Treated (n=54) 94. 3% ITT (NC=F) Percent of Patients (n=64) 78. 1% 0 DART II 2 4 8 12 Weeks 16 20 24

Virologic Response HIV RNA <50 copies/m. L: Baseline to Week 24 As Treated (n=54) 94. 3% Percent of Patients ITT (NC=F) (n=64) 78. 1% 0 DART II 2 4 8 12 Weeks 16 20 24

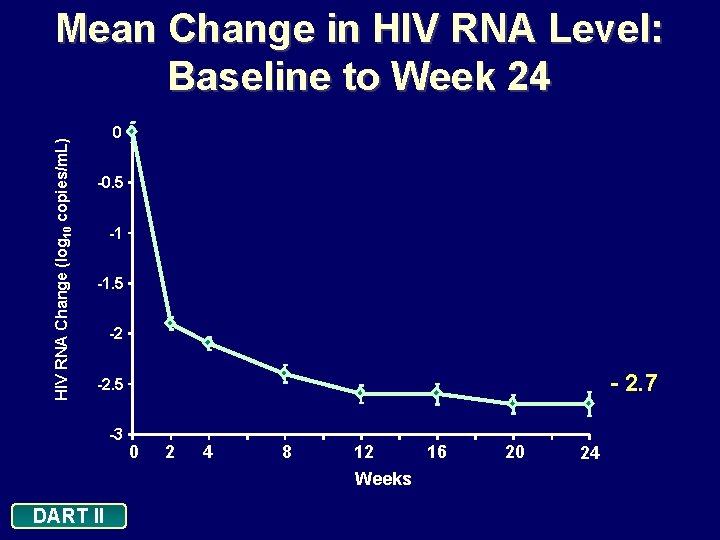
HIV RNA Change (log 10 copies/m. L) Mean Change in HIV RNA Level: Baseline to Week 24 0 -0. 5 -1 -1. 5 -2 - 2. 7 -2. 5 -3 DART II 0 2 4 8 12 16 Weeks 20 24

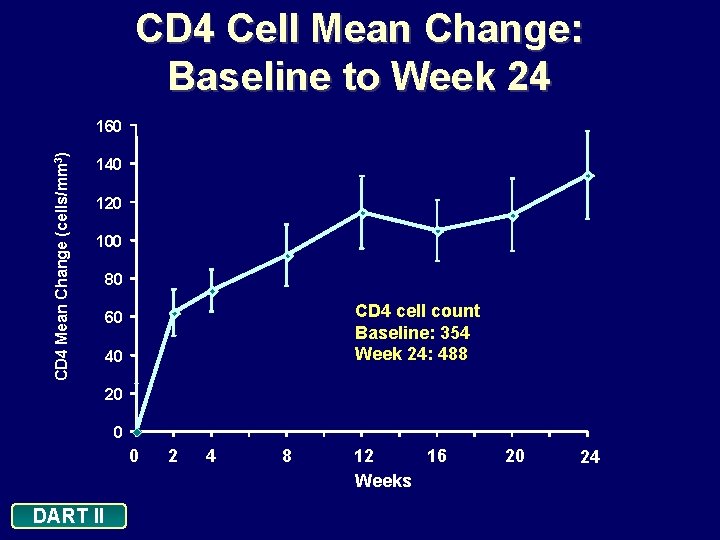
CD 4 Cell Mean Change: Baseline to Week 24 CD 4 Mean Change (cells/mm 3) 160 140 120 100 80 CD 4 cell count Baseline: 354 Week 24: 488 60 40 20 0 0 DART II 2 4 8 12 16 Weeks 20 24

Grade 2 - 4 Treatment Related Adverse Events (>2%) Grade 2 -4 d 4 T XR + 3 TC + EFV n=64 Nausea 4 (6. 3%) Hypoaesthesia 3 (4. 7%) Peripheral Neuropathy 2 (3. 1%) Diarrhea 2 (3. 1%) Somnolence 2 (3. 1%) Vomiting 2 (3. 1%) DART II

Treatment Related Adverse Events of Interest All Grades Peripheral Neuropathy d 4 T XR + 3 TC + EFV n=64 2 (3. 1%) Lipodystrophy 0 Symptomatic Hyperlactatemia 0 Lactic Acidosis Syndrome 0 Pancreatitis 0 DART II

Grade 3 -4 New Onset Laboratory Abnormalities Grade 3 -4 d 4 T XR + 3 TC + EFV n=64 Lipase 3 (4. 7%) Creatine Kinase 2 (3. 1%) ALT 2 (3. 1%) ANC 1 (1. 6%) *No DART II report of Grade 3 -4 triglyceride levels

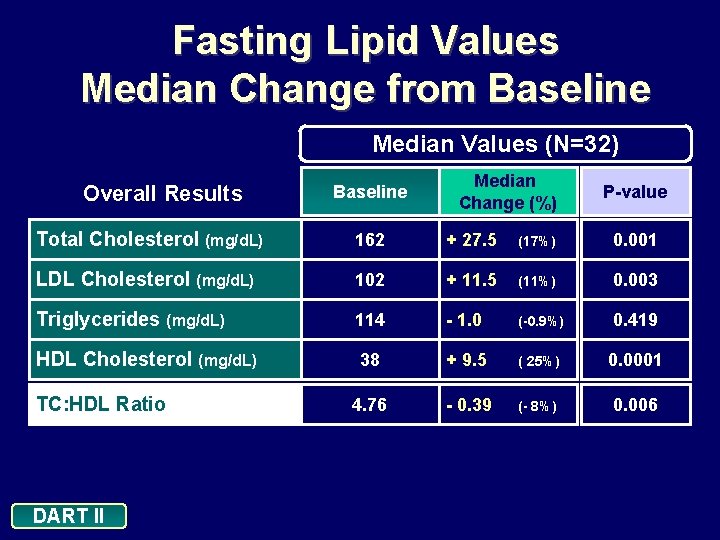
Fasting Lipid Values Median Change from Baseline Median Values (N=32) Overall Results Baseline Median Change (%) P-value Total Cholesterol (mg/d. L) 162 + 27. 5 (17%) 0. 001 LDL Cholesterol (mg/d. L) 102 + 11. 5 (11%) 0. 003 Triglycerides (mg/d. L) 114 - 1. 0 (-0. 9%) 0. 419 HDL Cholesterol (mg/d. L) 38 + 9. 5 ( 25%) 0. 0001 4. 76 - 0. 39 (- 8%) 0. 006 TC: HDL Ratio DART II

Summary of Adherence and Compliance at 24 Weeks Adherence (by AMAF* questionnaire) d 4 T XR + 3 TC +EFV Did not miss any dose during the past 4 days. 82. 3% Any time took fewer pills per dose 5. 6% Follow specific schedule 80. 8% Compliance (by pill count) (51/62) (3/54) (42/52) d 4 T XR 3 TC EFV n=53 80% Compliance at Week 24 89% 91% 89% 90% Compliance at Week 24 70% 72% *Antiviral Medication Adherence Form DART II

Conclusions: 24 Weeks In this treatment-naive population, d 4 T XR + 3 TC + EFV administered once daily: • Demonstrated a significant reduction in viral load and excellent efficacy: • 78. 1% of patients achieved viral loads <400 copies/m. L and <50 copies/m. L (ITT: NC=F) • 94. 3% of patients achieved viral loads <400 copies/m. L and <50 copies/m. L (As Treated analysis) • Is well tolerated • Minimal discontinuations due to adverse events (n=3) • Increase in HDL cholesterol, and decrease in TC: HDL ratio, with minimal effects on triglyceride levels DART II

Acknowledgments TO ALL THE PATIENTS AND STUDY CENTER PARTICIPANTS Stephen Becker, MD. San Francisco, CA Coordinator: Sunita Lundy Gary Richmond, MD. Ft. Lauderdale, FL Coordinator: Vernon Appleby, RN Nicholaos Bellos, MD. Dallas, TX Coordinators: Christopher Hayes Dawn Chaney Michael Sands, MD. Jacksonville, FL Coordinator: Debbie Aragon, RN Paul Cook, MD. Greenville, NC Coordinator: Grace Wilkins, RN Kunthavi Sathasivam, MD. Washington, DC Coordinator: Takada Harris Franco Felizarta, MD. Bakersfield, CA Coordinator: Jennifer Kuhach Leonard Slater, MD. Oklahoma City, OK Coordinator: Brenda Verel, LPN Dushyantha Jayaweera, MD. Miami, FL Coordinator: Rose Lalanne, RN DART II