CZECH TECHNICAL UNIVERSITY IN PRAGUE FACULTY OF BIOMEDICAL

CZECH TECHNICAL UNIVERSITY IN PRAGUE FACULTY OF BIOMEDICAL ENGINEERING Fluorescence spectroscopy and microscopy for biology and medicine Martin Hof, Radek Macháň

CZECH TECHNICAL UNIVERSITY IN PRAGUE FACULTY OF BIOMEDICAL ENGINEERING Fluorescence spectroscopy and microscopy for biology and medicine Martin Hof, Radek Macháň Absorption of light and electronic transitions Basic principles of fluorescence, fluorescence spectra Lifetime of fluorescence and its measurement Quenching of fluorescence and its biological applications Anisotropy of fluorescence and its biological applications Influence of solvent on fluorescence spectra Foerster resonance energy transfer and excimer fluorescence Fluorescent proteins Fluorescence microscopy, confocal and 2 -photon microscopy Resolution of fluorescence microscope and its enhancement Fluorescence correlation spectroscopy Photodynamic Therapy

Basic literature: 1. Lakowicz J. R. : Principles of Fluorescence Spectroscopy, 3 rd edn. Springer 2006 cfs. umbi. umd. edu/ 2. Hof M. , Hutterer R. , Fidler V. : Fluorescence Spectroscopy in Biology. Springer Verlag 3. Gauglitz G. , Vo-Dinh T. : Handbook of Spectroscopy. Wiley VCH Verlag, Weinheim 2003 4. Prosser V. a kol. : Experimentální metody biofyziky. Academia, Praha 1989 5. Invitrogen Tutorials www. invitrogen. com/site/us/en/home/support/Tutorials. html 6. Becker W. : The bh TCSPC Handbook http: //www. becker-hickl. com/literature. htm

Why fluorescence? electric fields ions • it provides information on the molecular environment Also fluorescence is very, Fluorescent very sensitive! viscosity temperature • it provides information on dynamic processes on the nanosecond timescale Work with subnanomolar Probe concentrations is routine while femtomolar polarity p. H and even SINGLE MOLECULE studies are possible with some effort Fluorescence Probes are essentially molecular stopwatches which monitor dynamic events which occur during the excited state lifetime – such as movements of proteins or protein domains
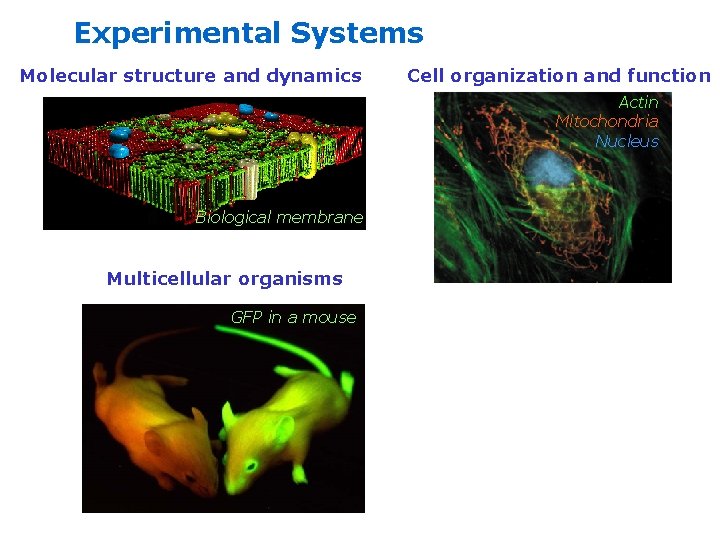
Experimental Systems Molecular structure and dynamics Cell organization and function Actin Mitochondria Nucleus Biological membrane Multicellular organisms GFP in a mouse
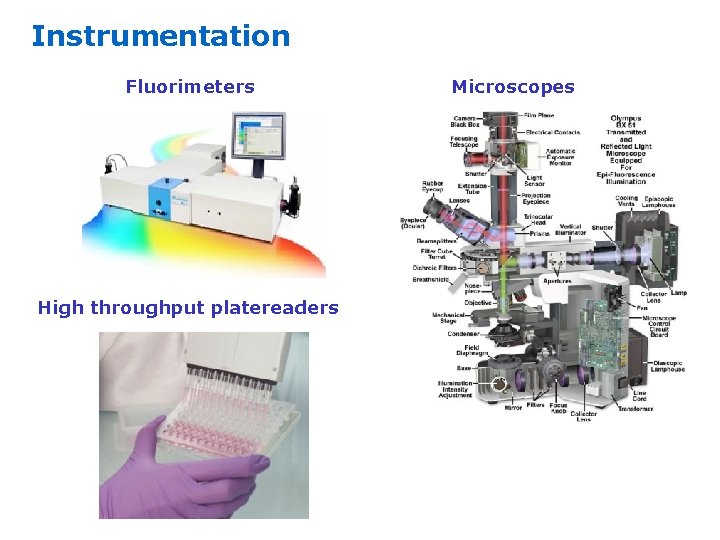
Instrumentation Fluorimeters High throughput platereaders Microscopes
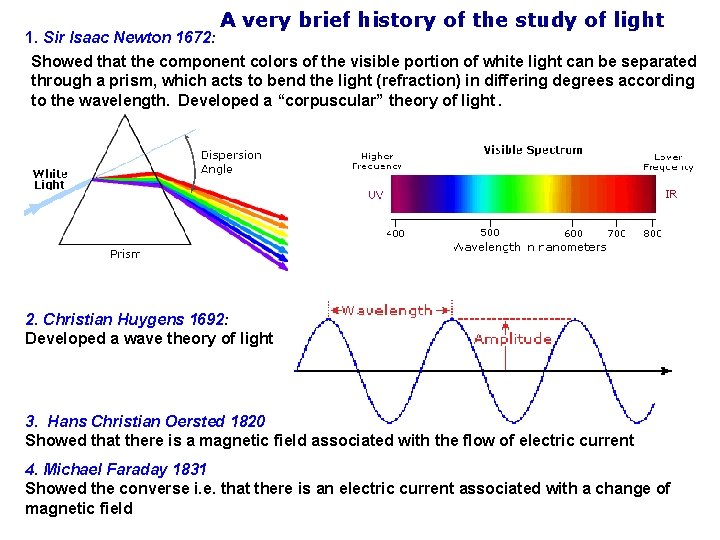
A very brief history of the study of light 1. Sir Isaac Newton 1672: Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light (refraction) in differing degrees according to the wavelength. Developed a “corpuscular” theory of light. 2. Christian Huygens 1692: Developed a wave theory of light 3. Hans Christian Oersted 1820 Showed that there is a magnetic field associated with the flow of electric current 4. Michael Faraday 1831 Showed the converse i. e. that there is an electric current associated with a change of magnetic field
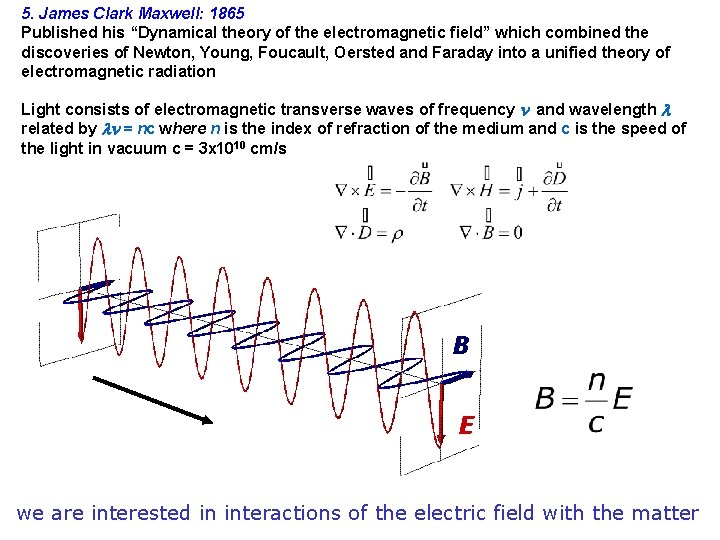
5. James Clark Maxwell: 1865 Published his “Dynamical theory of the electromagnetic field” which combined the discoveries of Newton, Young, Foucault, Oersted and Faraday into a unified theory of electromagnetic radiation Light consists of electromagnetic transverse waves of frequency and wavelength related by = nc where n is the index of refraction of the medium and c is the speed of the light in vacuum c = 3 x 1010 cm/s B E we are interested in interactions of the electric field with the matter

6. Max Karl Ernst Ludwig Planck: 1900 Explained the laws of black body radiation by postulating that electromagnetic radiation is emitted at discrete energetic quanta E = h , where Planck constant h = 6. 6256 *10 -34 Js. 7. Albert Einstein: 1905 Explained the explained the photoelectric effect by assuming that light is adsorbed at discrete energetic quanta E = h , photons. 8. Louis de Broglie: 1924 Introduced properties of electromagnetic waves to all particles – the wave-corpuscular dualism of quantum physics. A freely moving particle of momentum p has wavelength =h/p.
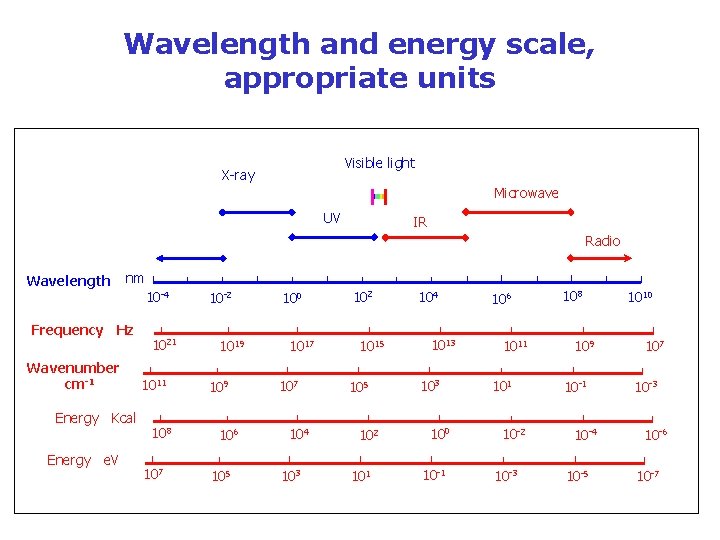
Wavelength and energy scale, appropriate units Visible light X-ray Microwave UV IR Radio Wavelength nm Frequency Hz Wavenumber cm-1 Energy Kcal Energy e. V 10 -4 1021 1011 108 107 10 -2 1019 106 105 100 1017 104 103 102 1015 102 101 104 1013 100 10 -1 106 1011 10 -2 10 -3 108 109 10 -1 10 -4 10 -5 1010 107 10 -3 10 -6 10 -7
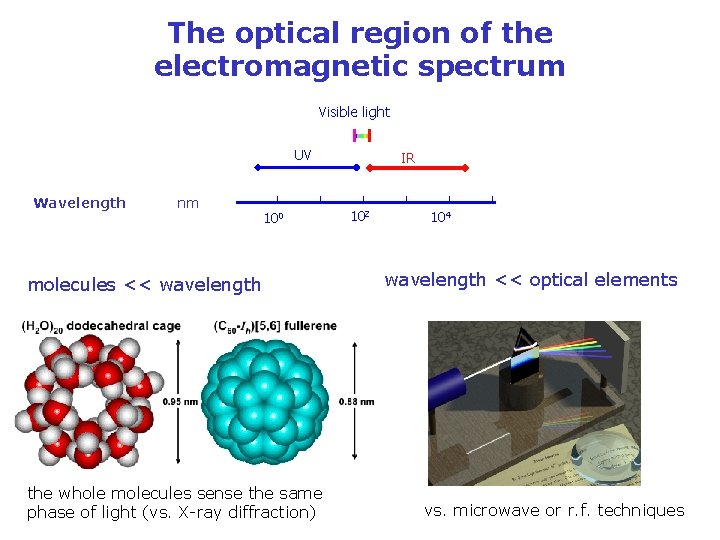
The optical region of the electromagnetic spectrum Visible light UV nm Wavelength 10 -4 nm 10 -2 100 molecules << wavelength the whole molecules sense the same phase of light (vs. X-ray diffraction) IR 102 104 106 108 wavelength << optical elements vs. microwave or r. f. techniques 1010
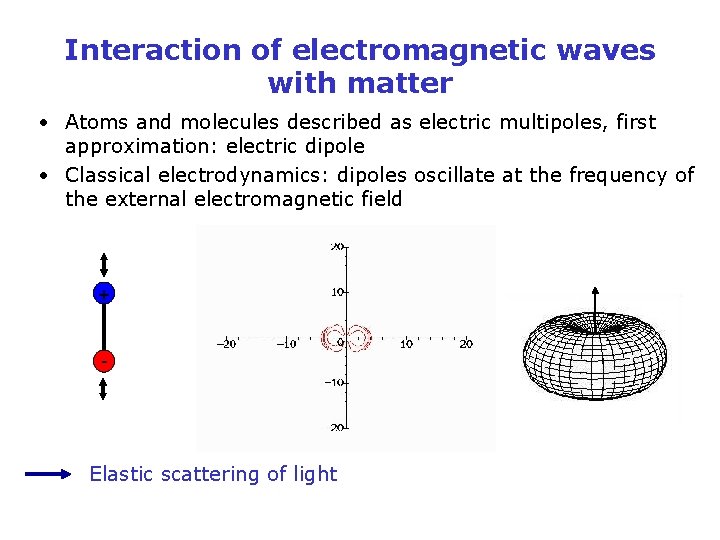
Interaction of electromagnetic waves with matter • Atoms and molecules described as electric multipoles, first approximation: electric dipole • Classical electrodynamics: dipoles oscillate at the frequency of the external electromagnetic field + - Elastic scattering of light

Interaction of molecules with photons - quantum description • Light exists in form of discrete quanta – photons E = hn • Atoms and molecules occupy discrete energetic states, which can be found as the solution of Schroedinger’s equation. • Exchange of energy with photons is accompanied by transitions between those states. rotational states DJ = 1 microwave region vibrational states DN = 1 IR – VIS region E electronic states UV – VIS region

Interaction of light with matter – overview of processes • elastic scattering – no exchange of energy between the molecule and the photon • inelastic (Raman) scattering – the photon either gives a part of its energy to the molecule or vice versa • absorption or emission of photons by the molecule 2 absorption spontaneous emission induced emission 1 • induced emission is coherent with incident light • spontaneous emission by individual molecules is incoherent • scattering is coherent and instantaneous

Elastic scattering of light • Rayleigh scattering – small molecules (x<0. 3) as a “point dipole”, Isc ≈ n 4 blue sky, red sunset x = 0. 07 • Larger scatterers – macromolecules, cells, Mie theory for spherically symmetrical scatterers x=7 http: //omlc. ogi. edu/calc/mie_calc. html
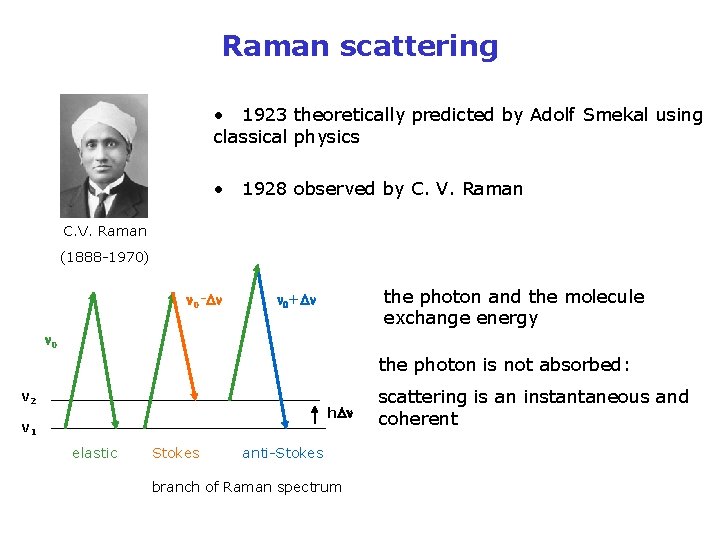
Raman scattering • 1923 theoretically predicted by Adolf Smekal using classical physics • 1928 observed by C. V. Raman (1888 -1970) n 0 -Dn the photon and the molecule exchange energy n 0+Dn n 0 the photon is not absorbed: v 2 h. Dn v 1 elastic Stokes anti-Stokes branch of Raman spectrum scattering is an instantaneous and coherent
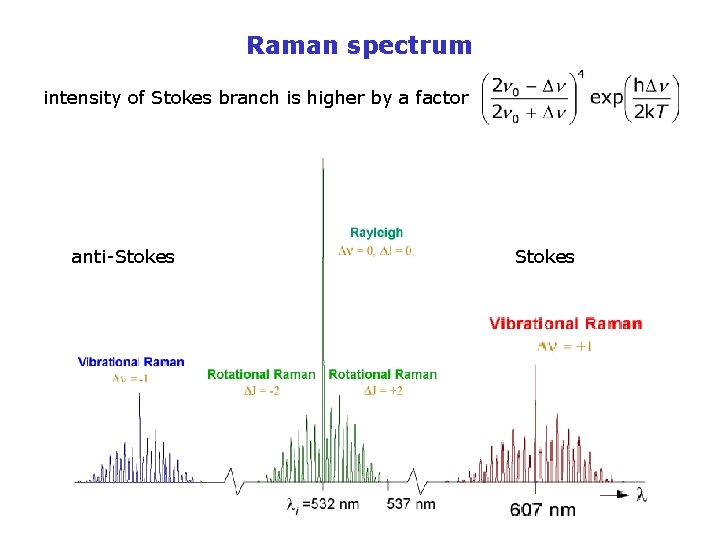
Raman spectrum intensity of Stokes branch is higher by a factor anti-Stokes

Absorption of light S 2 Nf 2 molecules DE = hv 0 1 I 0 dx Nf 1 molecules I § electromagnetic energy density § M – number of photons § N – number of molecules N = c NAS dx § angle between polarization and D 12, for random orientation of molecules § F shape of the spectral line – conservation of energy § small energy approximation – assumes that absorption does not change f 2/f 1=exp(DE/k. T)=f(T)

Absorption of light S 2 Nf 2 molecules DE = hv 0 1 I 0 dx Nf 1 molecules I the Lambert Beer law the molar extinction coefficient (molar absorptivity)
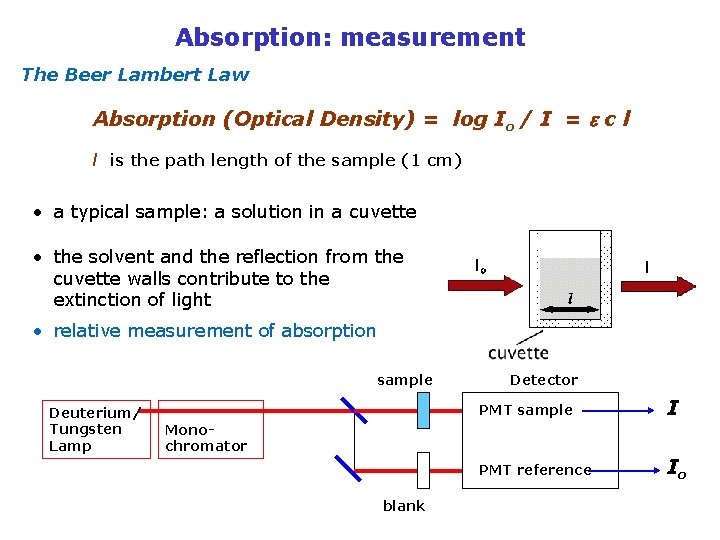
Absorption: measurement The Beer Lambert Law Absorption (Optical Density) = log Io / I = c l l is the path length of the sample (1 cm) • a typical sample: a solution in a cuvette • the solvent and the reflection from the cuvette walls contribute to the extinction of light • relative measurement of absorption sample Deuterium/ Tungsten Lamp Detector PMT sample I PMT reference Io Monochromator blank
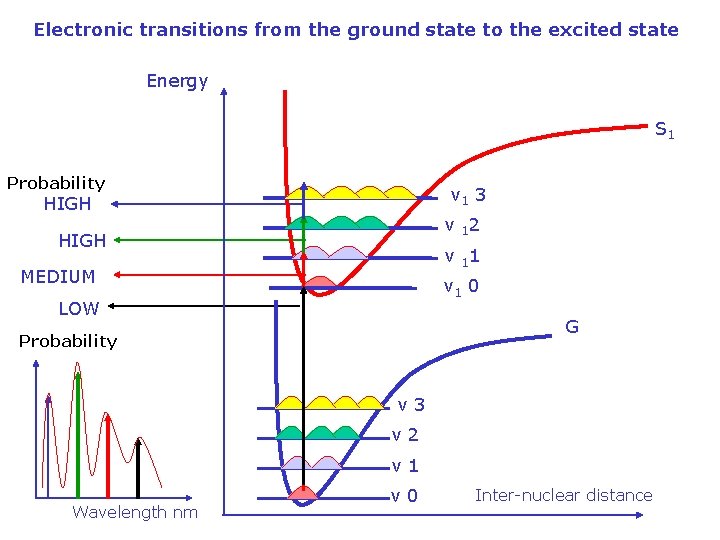
Electronic transitions from the ground state to the excited state Energy S 1 Probability v 1 3 HIGH v 12 HIGH v 11 MEDIUM v 1 0 LOW G Probability v 3 v 2 v 1 Wavelength nm v 0 Inter-nuclear distance
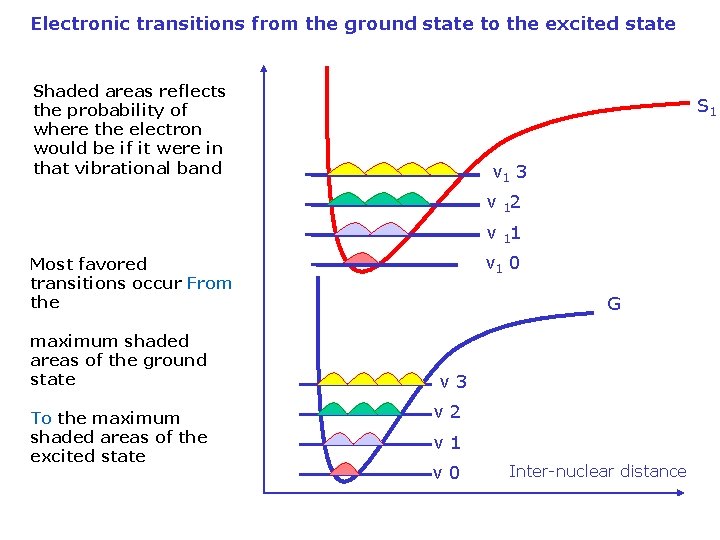
Electronic transitions from the ground state to the excited state Shaded areas reflects the probability of where the electron would be if it were in that vibrational band S 1 v 1 3 v 12 v 11 v 1 0 Most favored transitions occur From the maximum shaded areas of the ground state To the maximum shaded areas of the excited state G v 3 v 2 v 1 v 0 Inter-nuclear distance
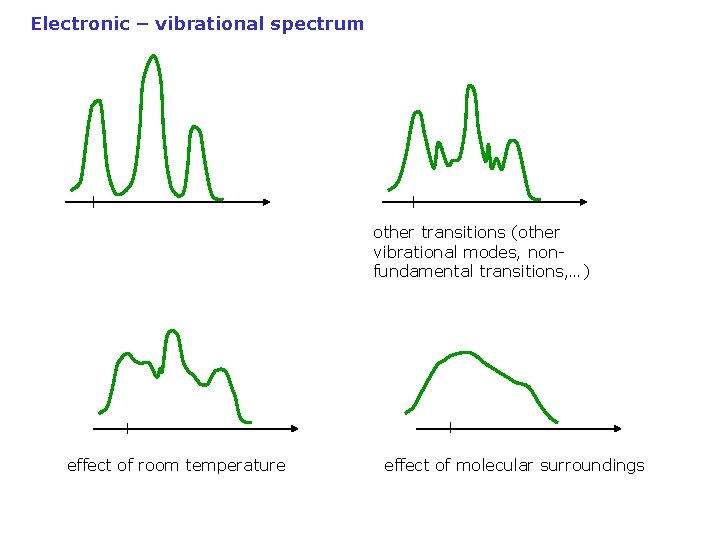
Electronic – vibrational spectrum other transitions (other vibrational modes, nonfundamental transitions, …) effect of room temperature effect of molecular surroundings
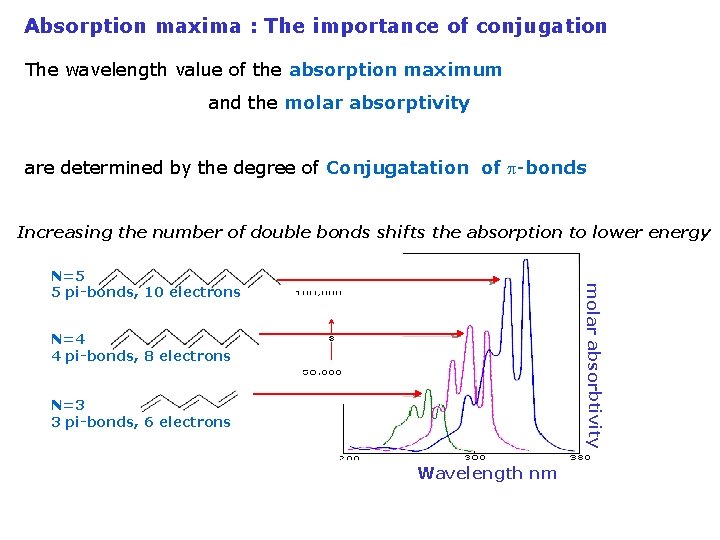
Absorption maxima : The importance of conjugation The wavelength value of the absorption maximum and the molar absorptivity are determined by the degree of Conjugatation of p-bonds Increasing the number of double bonds shifts the absorption to lower energy molar absorbtivity N=5 5 pi-bonds, 10 electrons N=4 4 pi-bonds, 8 electrons N=3 3 pi-bonds, 6 electrons Wavelength nm
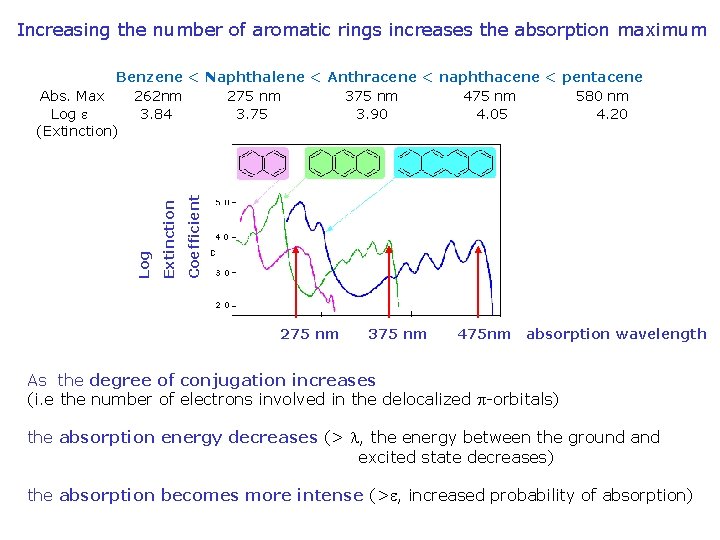
Increasing the number of aromatic rings increases the absorption maximum Coefficient Extinction Log Benzene < Naphthalene < Anthracene < naphthacene < pentacene Abs. Max 262 nm 275 nm 375 nm 475 nm 580 nm Log e 3. 84 3. 75 3. 90 4. 05 4. 20 (Extinction) 275 nm 375 nm 475 nm absorption wavelength As the degree of conjugation increases (i. e the number of electrons involved in the delocalized p-orbitals) the absorption energy decreases (> l, the energy between the ground and excited state decreases) the absorption becomes more intense (>e, increased probability of absorption)

Emission of light - Luminescence – the excess of light emitted above thermal radiation. The emission follows after the molecule has resided for some time in the excited state. according to excitation mechanism: photoluminescence – absorption of light chemiluminescence – chemical reaction thermoluminescence – heat electroluminescence – electric current … fluorescence phosphorescence

Typical sources of luminescence • organic molecules (usually with conjugated p-bonds) – synthetic fluorophores (fluorescein, rhodamine, …), biological molecules (aromatic amino acids – Trp, Tyr, chlorophyll, …) • small inorganic molecules – noble gases (in discharge lamps), N 2 (in lasers, responsible for bluish colour of spark discharges), … • inorganic crystals (diamond, Si, Ga. As, … ) – the spectra depend on the bandgap size, which depends on the size of the crystal (nanocrystals emit in VIS – quantum dots), extreme photostability quantum dots – same material, different sizes

Acknowledgement The course was inspired by courses of: Prof. David M. Jameson, Ph. D. Prof. RNDr. Jaromír Plášek, Csc. Prof. William Reusch Financial support from the grant: FRVŠ 33/119970
- Slides: 28