Cytoskeleton A network of protein fibers collectively called

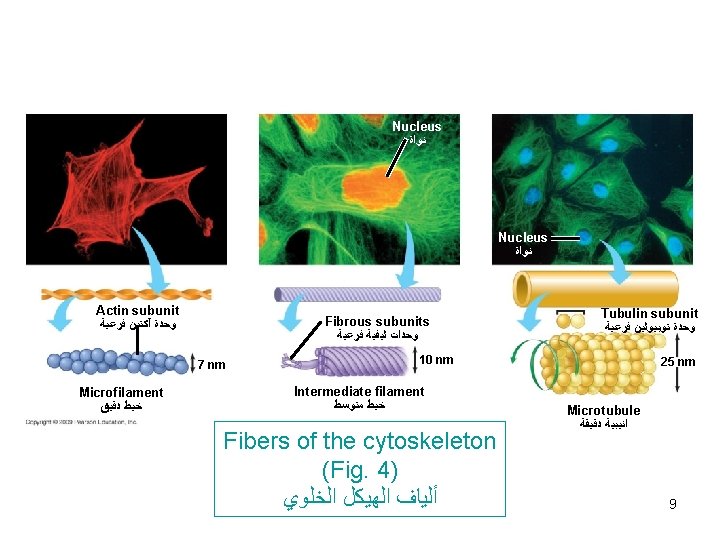
Cytoskeleton *A network of protein fibers collectively called cytoskeleton extend through cytoplasm. *According to size of filaments, they are divided into 3 classes : 1) Microfilaments (7 nm. ) composed of actin. 2) Intermediate filaments (10 nm. ) composed of 6 proteins. 3) Microtubules (25 nm. ) composed of 2 tubulin proteins. Functions of cytoskeleton : 1)Attach to cell membranes and to each other by anchoring and joining proteins to form a 3 D internal meshwork responsible for maintenance of cell morphology. 1

2) Structural support + various types of cell movements, It is an active participant in cellular motion either organelles or vesicles within the cell or the entire cell. 3) Anchors organelles. 4) Regulate activities by transmitting signals from cell’s surface to its interior. 1) Microfilaments (assemblies of actin) -Globular protein (G-actin). -polymerize to form filaments (F-actin). -Have several molecular variants isoforms. -Solid rods arranged in a twisted double chain. 2

Functions: 1) Mechanical support of cell membranes by attachment to it via membranes-anchoring proteins such as ankyrin + Spectrin present in RBCs to attach actin to RBCs membrane. (See Fig 1) 2) Present in microvilli in the form of bundles. (Fig. 2) 3) Interact with myosin to produce ms contraction. (see chapter 3). 4) contractile ring (cleavage furrow) during cytokinesis. 2) Intermediate filaments -Rope-like filaments. -According to cell types and functions are 6 types desmin, cytokeratin, vimentin, nuclear lamin, neurofilament protein and glial fibrillary acidic protein. 3

Functions -Reinforce cell shape and to anchor certain organelles e. g. 1) in nucleus : nuclear lamins form a lattice on inner side of nuclear membrane which hold it in place. 2) In epithelial cells of skin (keratin) form a tough outer layer and makes an impermeable barrier as well as the main protein of hairs + nails. 4
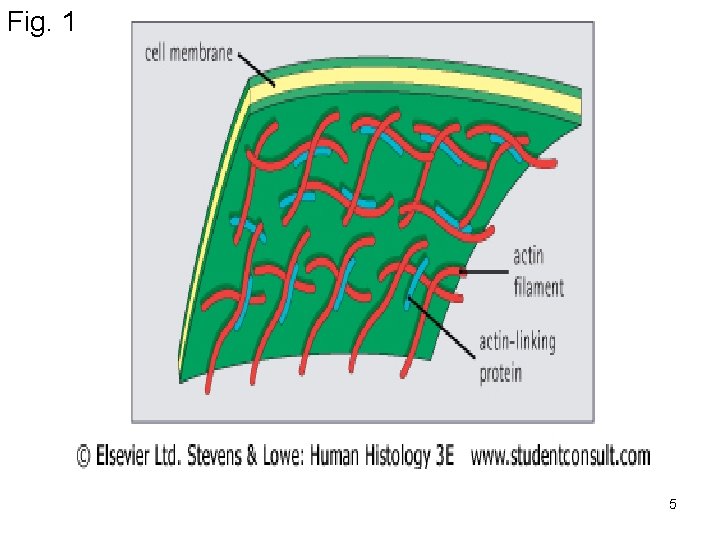
Fig. 1 5
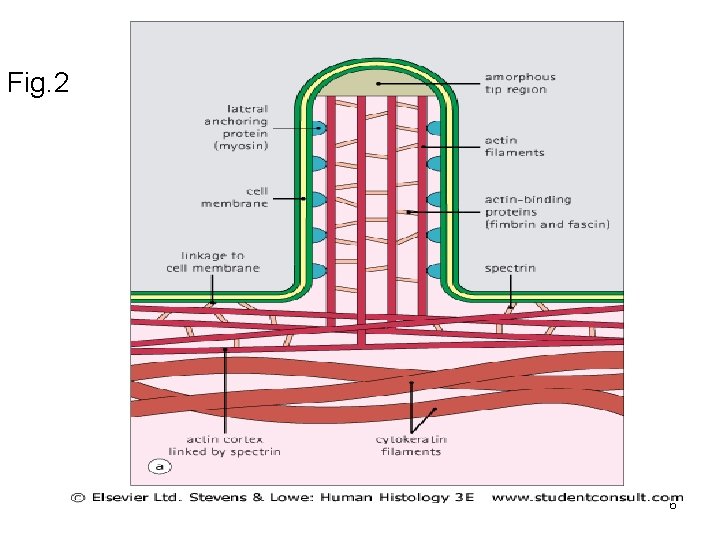
Fig. 2 6

3) Microtubules 1) Are straight hollow tubes composed of globular proteins α-tubulin and β-tubulin form a dimer which polymerize to form 13 protofilaments. (Fig. 3) 2) A microtubule elongated by addition of tubulin dimer and dissassembled by removal of dimers to be recycled and form microtubules in other parts of cells. Assembly needs (GTP + Ca+), drugs as colchicine prevent assembly of microtubules. 3) Has 2 ends plus and minus. 4)They grow from a region called ‘’mirotubule organizing center called’’ Centrosome’’ which contains 2 centrioles. (MTOC) 7
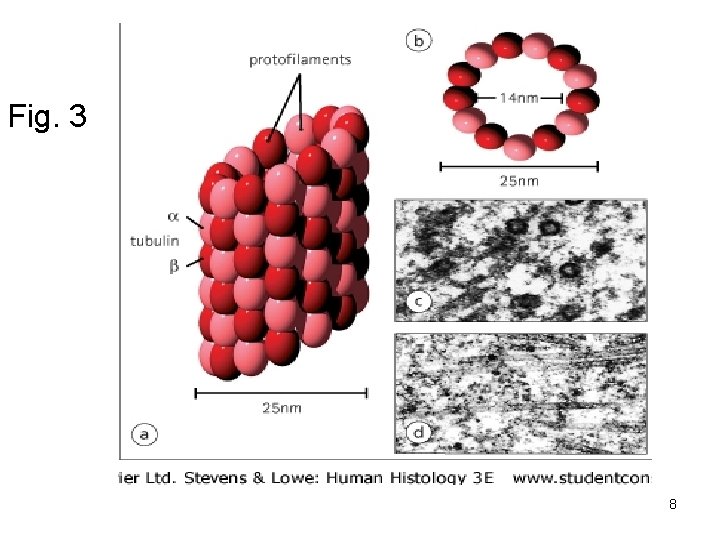
Fig. 3 8
CENTRIOLES - Are formed of 9 bundles, each bundle formed of 3 microtubules ( 9+3). - Duplicated before cell division. - Plant cells+ Fungal cells have (MTOC) but lack centrioles. Microtubule-associated proteins (Fig. 5) Structural MAPs Motor MAPs Cross- link microtubules Use energy to produce to other cytoskeleton polymers movement -Move of vesicles or organeles within the cell need 2 motor proteins: Kinesin Move towards plus end. Dynein move towards minus end 10
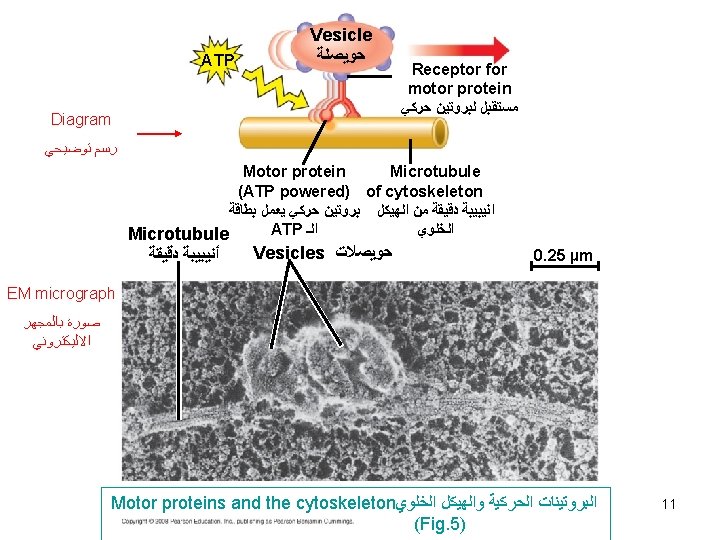

Functions of microtubules 1) Tracks for organelles equipped with motor proteins can move. e. g a lysosome can move along a microtubule to reach a vacuole. 2) Movement of cilia + flagella. 3) Formation of mitotic spindle during nuclear division 4) Maintain the extended tubular arrangement of ER. 12

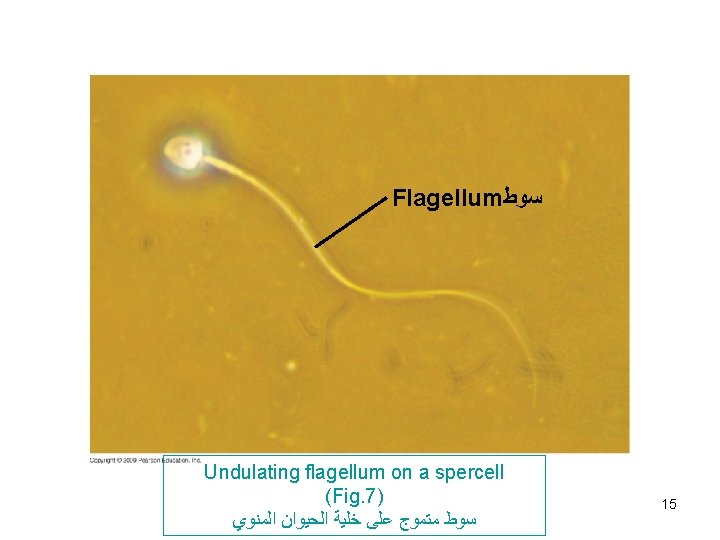
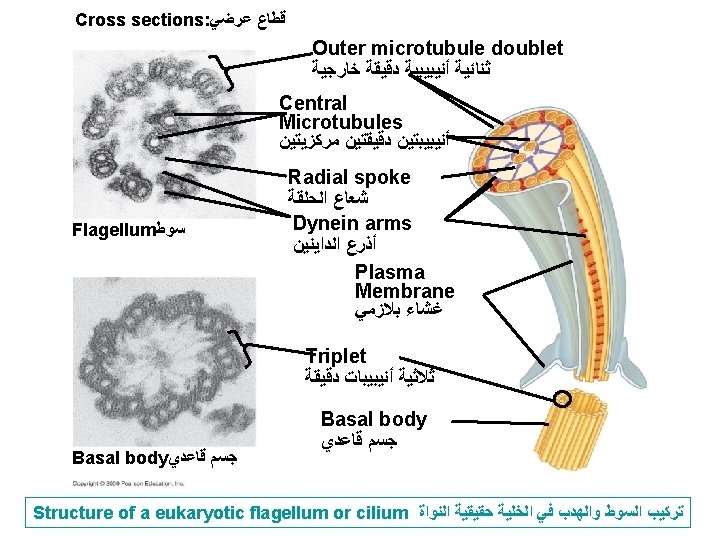
Cilia and flagella (Fig. 6 -7) -Both are composed of microtubules wrapped in an extension of cell membrane. -Composed of 9 bundles of microtubules each consist of 2. This characteristic arrangement is (9+2) called doublet tubules -In eukaryotic cilia and flagella, there are in addition 2 unpaired central microtubules. -They are anchored in a structure called ‘’basal body’’ similar to centrioles. (9 + 3) called triplet tubules. -Cilia move like coordinated oars e. g in human respiratory system to sweep mucus containing trapped debris. Flagella move like an undulating whipelike motion e. g. flagellated sperm 13
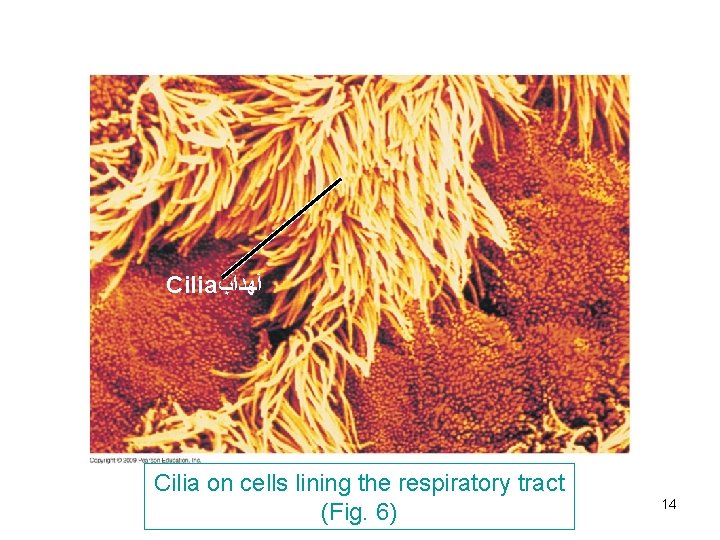
Cilia ﺃﻬﺪﺍﺏ Cilia on cells lining the respiratory tract (Fig. 6) 14

Extracellular Matrix -Materials that are external to the plasma membrane composed of 2 major materials : 1) Glycosoaminoglycans (GAG) 2) Fbrillar proteins. Glycosoaminoglycans and proteoglycans - Large unbranched, polysaccharides chains composed of repeated disaccharide units. The disaccharide units contain either of two modified sugars, N-acetylgalactosamine (Gal. NAc) or N-acetylglucosamine (Glc. NAc), and a uronic acid such as glucuronate. These polysaccharides link to backbone of proteins forming proteoglycans. -The exception is the GAG hyaluronanic acid, which is uniquely synthesized without a protein core and does not contain any 17 sulfate.

Properties of GAG 1) A high negative charge because one of the repeated units is N-acetylglucosamine or N-cetyl galactosamine is sulfated (SO 3 -), and most GAG has a second sugar uronic acid with a COO- group. with extended conformation that imparts high viscosity to the solution. Along with the high viscosity of GAGs comes low compressibility, which makes these molecules ideal for a lubricating fluid in the joints. 2) Strongly hydrophilic behavior. 3) Retension of +ve ions (e. g. Na+) and water. This property makes them excellent lubricators and shock absorbers. 4) Covalent attachment with proteins to form proteoglycans. 18

Classification They are 4 groups according to structure : hyaluronic acid, chondroitin sulfate, keratan sulfate, heparin and heparin sulfate, dermatan sulfate, 19
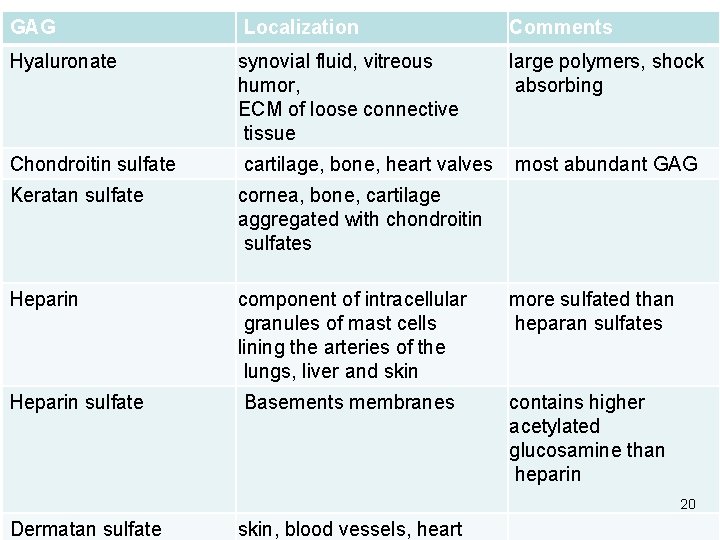
GAG Localization Comments Hyaluronate synovial fluid, vitreous humor, ECM of loose connective tissue large polymers, shock absorbing Chondroitin sulfate cartilage, bone, heart valves most abundant GAG Keratan sulfate cornea, bone, cartilage aggregated with chondroitin sulfates Heparin component of intracellular granules of mast cells lining the arteries of the lungs, liver and skin more sulfated than heparan sulfates Heparin sulfate Basements membranes contains higher acetylated glucosamine than heparin 20 Dermatan sulfate skin, blood vessels, heart

Fibrillar proteins They are collagen, fibrillin, elastin, fibronectin. Role : 1) Provide different tensile properties to support tissues 2) Anchoring for other cellular elements in tissues. 1) Collagen *Collagen are a family of closely related proteins which can aggregrate to produce either filaments, fibrils or meshwork, they interact with other proteins to provide support for ECM. * The tropocollagen or "collagen molecule" is a subunit of larger collagen aggregates. This fibril is approximately 300 nm long and 1. 5 nm in diameter, made up of three polypeptide strands (called alpha 21 chains).

§ These three left-handed helices are twisted together into a larger triple helix or "super helix", a cooperative quaternary structure stabilized by numerous hydrogen bonds. § It is produced by fibroblasts. § After proteolytic cleavage, the triple helical portions are assembled into long filaments and bundles. 22

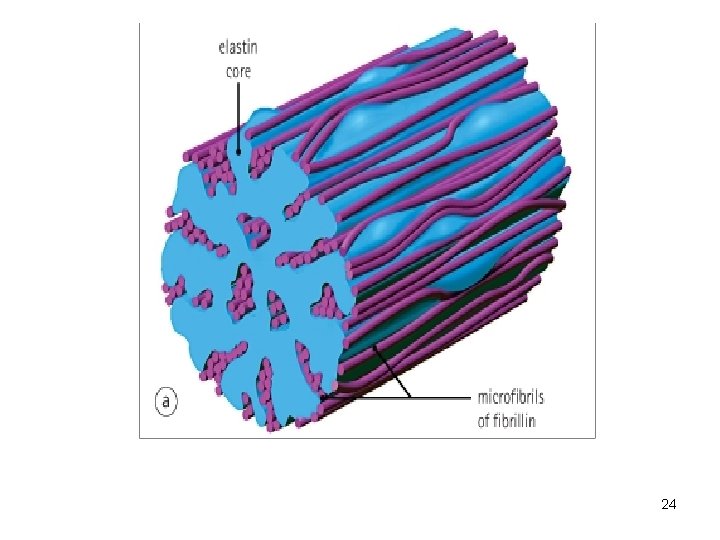
2) Elastin (Fig. 8) -Is a protein assembled into filaments and sheets. -Elastic fibers are formed by interaction between elastin and fibrillin (the latter organize secreted elastin to form elastic fibers. 3) Fibrillin (Fig. 8) -Fibrils forming glycoproteins (microfibrils). -Main component of ECM. -Microfibrils are one constituent of elastic fibers + suspensory fibers of lens. -Microfibrils are prominent in elastic containing 23
24

extracellular matrix particullary in lung, skin and blood vessels. 4) Fibronectin (Fig. 9 + Fig. 10) -Is a multifunctional glycoprotein an has the ability to adhere to different tissue components because it possesses binding sites that bind collagen as well as cell adhesion molecules such as integrin family of receptors (Integrins). Cell adhesion molecules are molecules that allow cells to adhere to ECM. 5) Integrins (Fig. 9+ Fig. 10) -Transmembrane proteins similar to cell membrane receptors in that they form bonds with ligands. Ligands are not signaling molecules but structural members of 25

ECM such as collagen, laminin and fibronectin. - Integrins have 3 domains: a binding domain to actin, a transmembrane domain and an extracellular domain binding to fibronectin. -Transmit information between extracellular and cytoskeleton, thus integrating changes occuring inside and outside cell. 26
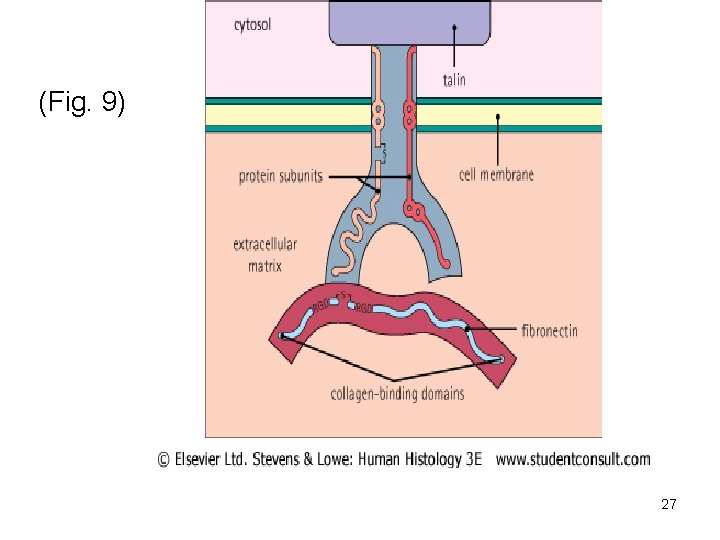
(Fig. 9) 27
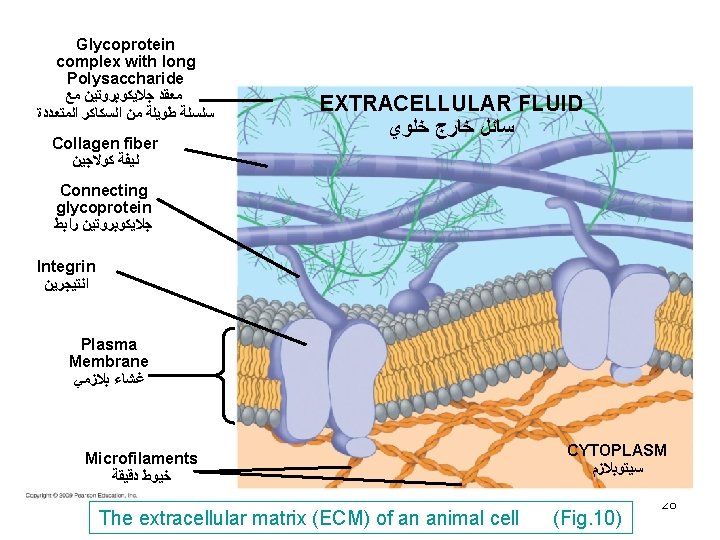
Glycoprotein complex with long Polysaccharide ﻣﻌﻘﺪ ﺟﻼﻳﻜﻮﺑﺮﻭﺗﻴﻦ ﻣﻊ ﺳﻠﺴﻠﺔ ﻃﻮﻳﻠﺔ ﻣﻦ ﺍﻟﺴﻜﺎﻛﺮ ﺍﻟﻤﺘﻌﺪﺩﺓ Collagen fiber ﻟﻴﻔﺔ ﻛﻮﻻﺟﻴﻦ EXTRACELLULAR FLUID ﺳﺎﺋﻞ ﺧﺎﺭﺝ ﺧﻠﻮﻱ Connecting glycoprotein ﺟﻼﻳﻜﻮﺑﺮﻭﺗﻴﻦ ﺭﺍﺑﻂ Integrin ﺍﻧﺘﻴﺠﺮﻳﻦ Plasma Membrane ﻏﺸﺎﺀ ﺑﻼﺯﻣﻲ Microfilaments ﺧﻴﻮﻁ ﺩﻗﻴﻘﺔ The extracellular matrix (ECM) of an animal cell CYTOPLASM ﺳﻴﺘﻮﺑﻼﺯﻡ (Fig. 10) 28

Cell adhesions in plants - In plant cells, bacteria and fungi they are surrounded by cell walls that contain multiple layers of polysaccharides ‘’Cellulose’’. - A growing plant cell secretes a primary cell wall which can stretch when it stops growing, a secondary cell wall is formed between 1 ry cell wall and plasma membrane. -Between the primary cell walls of adjacent cells, middle lamella of polysaccharides called Pectins adhere cells tightly to one another. 29
- Slides: 29