Cystic Fibrosis A Review for Healthcare Providers Provided

Cystic Fibrosis: A Review for Healthcare Providers Provided by the Indiana State Department of Health

Outline • • Fast facts about CF Diagnosis CFTR gene Newborn screening Symptoms Medical management / prognosis Inheritance / recurrence risks

Fast facts about CF • Currently, there approximately 30, 000 people in the US with CF* • ISDH estimates that each year in Indiana, approximately 300 children with at least one CFTR mutation will be identified through newborn screening^ *Cystic Fibrosis Foundation Patient Registry Annual Data Report, 2005 ^Based on annual live birth rate of 87, 000 children

Fast facts (cont. ) Frequency of CFTR mutations in ethnic groups* Caucasians^ 1 in 25 is the most common autosomal recessive condition in Caucasians carries at least one CFTR mutation ^CF Hispanics African-Americans 1 in 46 carries at least one CFTR mutation 1 in 65 carries at least one CFTR mutation *Genzyme Genetics, 2006

Newborn screening for CF • All infants receive IRT screen as part of newborn screening (heelstick) – IRT = immunoreactive trypsin • Infants with IRT above cutoff identified by the IU Newborn Screening Laboratory will receive DNA testing – IU Newborn Screening Laboratory tests for a panel of 44 common CFTR mutations – Note: CF newborn screening does NOT test for all known CFTR mutations

Newborn screening (cont. ) • If an infant is found to have an elevated IRT and 1+ CFTR mutations: – IU Newborn Screening Laboratory will notify primary care physician • Phone call & letter – IU Newborn Screening Laboratory will notify ISDH – ISDH will contact PCPs to assist with: • Referring patients for sweat chloride testing • Referring patients to CF centers • Counseling / educating families

Timeline for referrals • Primary care physicians should refer infants with 2 identified mutations to a Cystic Fibrosis Center within 2 weeks for evaluation and treatment • Primary care physicians should refer infants with 1 identified mutation for sweat chloride testing within 2 weeks of receiving newborn screening result – ISDH recommends referring patients to Cystic Fibrosis Foundation (CFF) accredited laboratories

Why refer to CFF-accredited labs? • Indiana has chosen to follow the national precedent by referring patients to CFFaccredited centers for sweat chloride testing and management • CFF-accredited laboratories meet nationally-accepted standards selected by the CFF in conjunction with the Clinical and Laboratory Standards Institute (CLSI, formerly NCCLS)

Why refer to CFF-accredited labs? (cont. ) • National standards are imperative to ensure that sweat chloride test results are consistently accurate and reliable – Several different sweat chloride test procedures exist – Procedures not approved by CFF & CLSI have demonstrated high false-positive and false-negative rates • Collection procedure is highly sensitive • Other procedures are erroneous

What about children with one CFTR mutation? • All children with at least one identified CFTR mutation should be referred for sweat chloride testing

What about children with one CFTR mutation? • Children with one identified mutation are typically asymptomatic carriers. . . • . . . however, some children with a single mutation will have CF • Again, newborn screening does not test for all known CFTR mutations – Some children could have a common mutation (identified through newborn screening) and a rare mutation that is not included in newborn screening panel

CFTR gene • Over 1, 400 disease-causing mutations currently known – Most are rare • Found in a single family – Almost all are small deletions or “point mutations” • Note: CF newborn screening does NOT test for all known mutations • Most common mutation: F 508 – Represents approximately 30 – 80% of all identified mutations in patients with CF – Results in “classic” features of CF if two copies are present
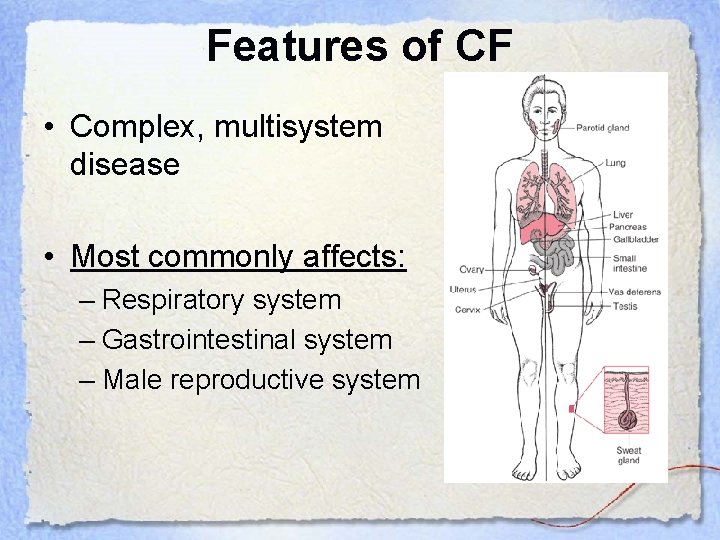
Features of CF • Complex, multisystem disease • Most commonly affects: – Respiratory system – Gastrointestinal system – Male reproductive system
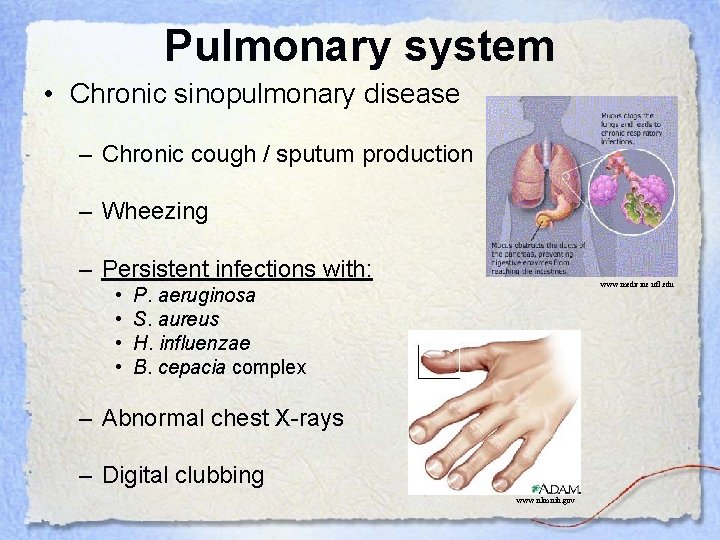
Pulmonary system • Chronic sinopulmonary disease – Chronic cough / sputum production – Wheezing – Persistent infections with: • • www. medicine. ufl. edu P. aeruginosa S. aureus H. influenzae B. cepacia complex – Abnormal chest X-rays – Digital clubbing www. nlm. nih. gov

Gastrointestinal system • Pancreatic insufficiency (> 90% of patients) • Malabsorption – Especially of fats / fat-soluble vitamins – May lead to failure to thrive, skin rashes, anemia • • • Meconium ileus (15 – 20% of patients) Pancreatitis Hepatobiliary disease Hypoproteinemia Distal intestinal obstructions

Male reproductive system • > 95% males with CF are infertile – Abnormal development of Wolffian ducts leads to azoospermia – Testicular development / function and spermatogenesis are usually normal

Male reproductive system (cont. ) • Congenital bilateral absence of vas deferens (CBAVD) can occur in males with specific CFTR mutations – Regardless of whether these males have other symptoms of CF • For more information, or with questions about specific gene changes, please contact ISDH – Contact information is provided at the end of this review

Medical management • Recommend referral to Cystic Fibrosis Foundation (CFF) accredited center if two mutations present or with a positive sweat test – Centers offer a multidisciplinary approach that includes: • • • MDs RNs Respiratory therapists Dieticians Social workers Genetic counselors • A list of CFF-accredited centers in Indiana is included in this review – For a list of CFF-accredited centers in other states, please contact Constance Burrus at ISDH

Prognosis • Average lifespan into the upper 30 s • Progressive pulmonary disease is most common cause of death • Available therapies include nutritional, pulmonary, insulin, etc. – Gene therapy is not currently available clinically
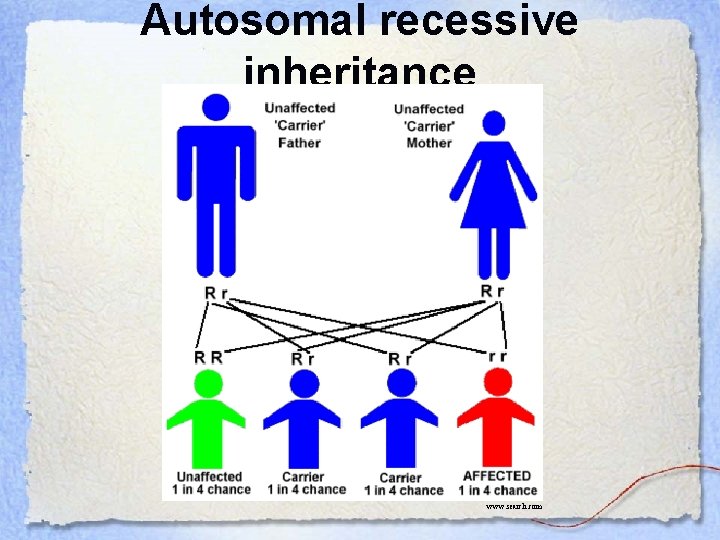
Autosomal recessive inheritance www. search. com
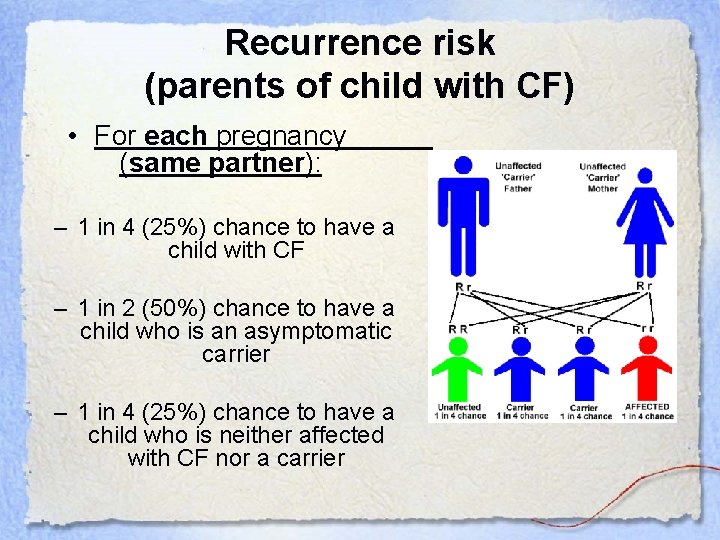
Recurrence risk (parents of child with CF) • For each pregnancy (same partner): – 1 in 4 (25%) chance to have a child with CF – 1 in 2 (50%) chance to have a child who is an asymptomatic carrier – 1 in 4 (25%) chance to have a child who is neither affected with CF nor a carrier

Recurrence risk (cont. ) • Each unaffected sibling of a child with CF has a 2/3 chance of being an asymptomatic carrier of a single CFTR mutation • Other family members are also at increased risk of being asymptomatic carriers – Carriers would have increased risk of having children with CF – Recommend genetic counseling / testing for at

Future pregnancies (parents of a child with CF) • Prenatal genetic counseling* recommended for any couple concerned about the risk of having a child with CF • Prenatal screening for CF routinely offered to: – All Caucasian couples – Couples with family history of CF / CBAVD *Contact ISDH for a list of prenatal genetic counselors & clinics in your area

For more information • If you would like to receive an electronic copy of this review, please contact: Malorie Hensley, MS Cystic Fibrosis Program Director Indiana State Department of Health 317. 233. 7019 MHensley@isdh. IN. gov

For more information (cont. ) • Malorie Hensley, MS Cystic Fibrosis & Genomics Programs Director Indiana State Department of Health 317. 233. 7019 Mhensley@isdh. IN. gov • Courtney Eddy, MS, CGC, LGC, MT(ASCP) INSTEP Director Indiana State Department of Health 317. 233. 9260 CEddy@isdh. IN. gov
- Slides: 25