Cycling of Matter in Ecosystems Recycling Matter All








- Slides: 18

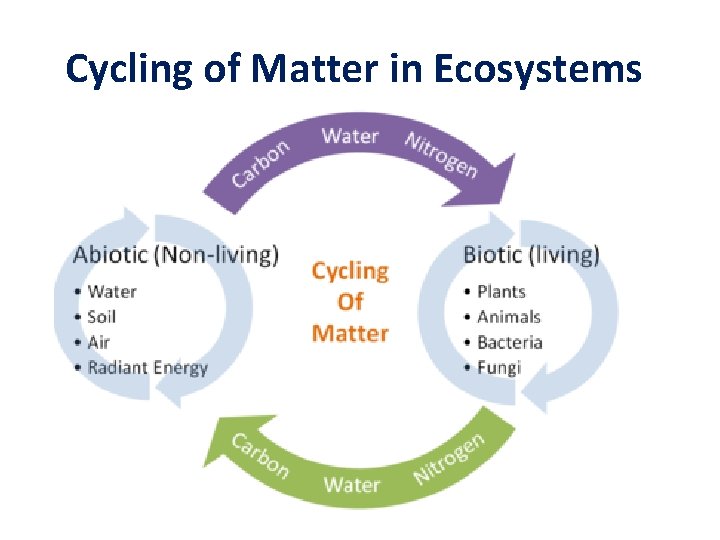
Cycling of Matter in Ecosystems

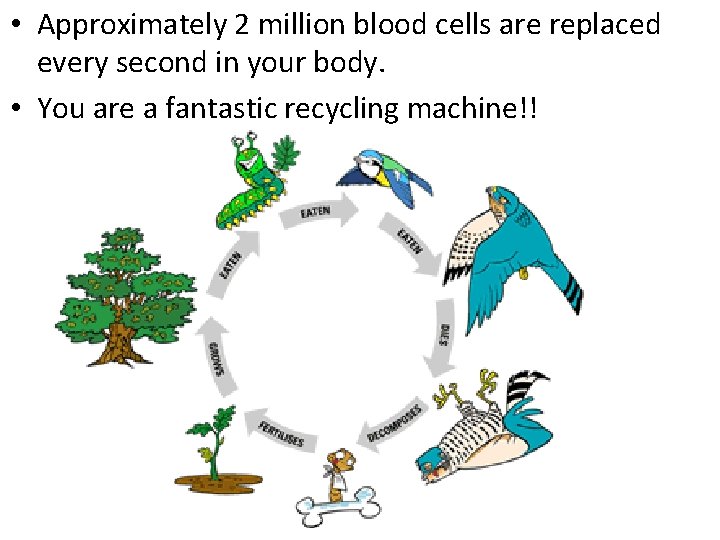
Recycling Matter • All life on Earth requires water and nutrients • These particles of matter don’t remain in your body forever • Every part of every cell in your body is replaced over time • We are like living recycling machines • Nutrients from food are used to repair and renew all the cells in our body

• Approximately 2 million blood cells are replaced every second in your body. • You are a fantastic recycling machine!!

Biogeochemical Cycles • As matter cannot be created or destroyed, it must be produced or obtained from chemicals that already exist in the environment. • Therefore biogeochemical cycles involve movement of matter (cycles) through the biotic and abiotic environment on earth. • These cycles are: 1. Water Cycle 2. Carbon Cycle 3. Nitrogen Cycle

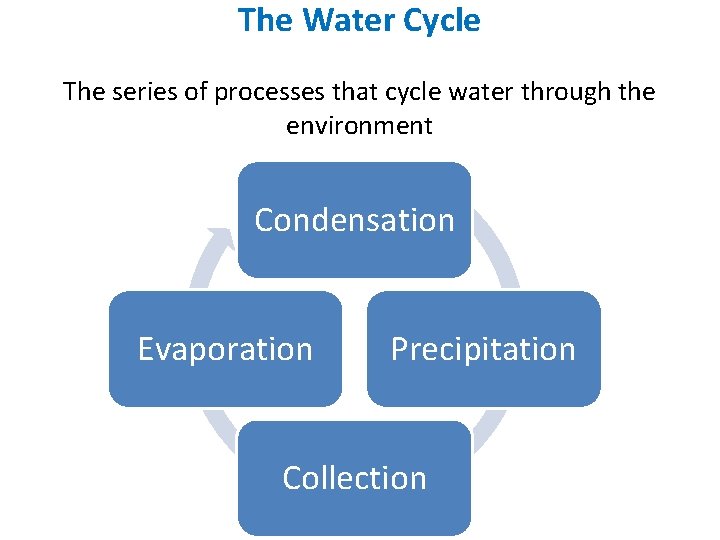
The Water Cycle The series of processes that cycle water through the environment Condensation Evaporation Precipitation Collection


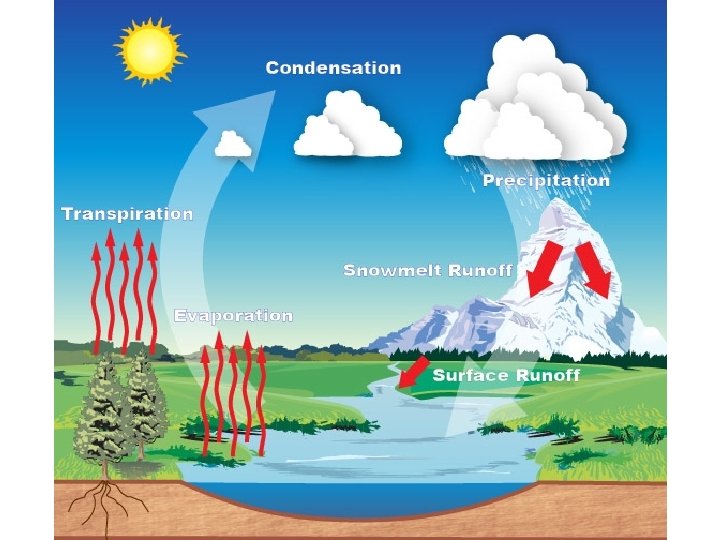
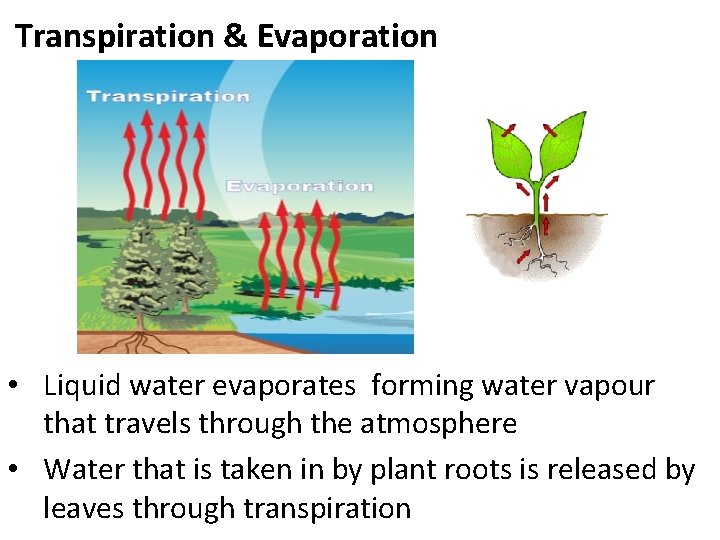
Transpiration & Evaporation • Liquid water evaporates forming water vapour that travels through the atmosphere • Water that is taken in by plant roots is released by leaves through transpiration

Condensation • The vapour eventually condenses • It forms liquid water or ice crystals in the atmosphere

Precipitation and Runoff • Vapour returns to the Earth as rain, hail or snow • Water falling on land melting snow move across the surface (runoff) and enter bodies of water • If this doesn’t occur it will enter the soil and groundwater instead

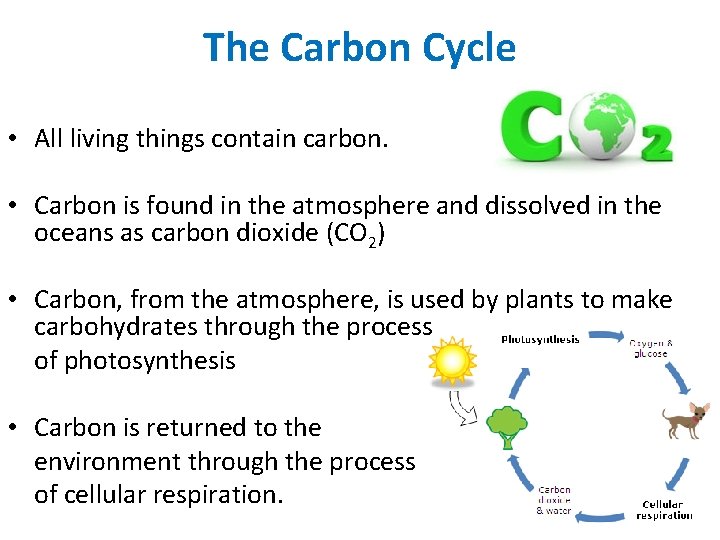
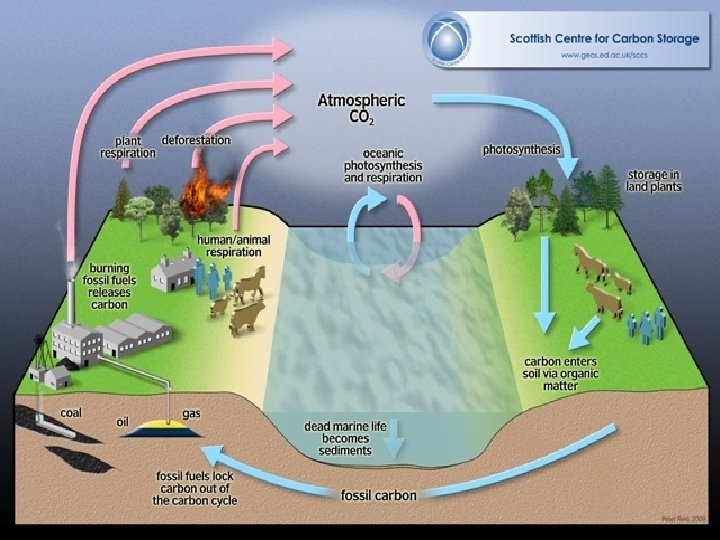
The Carbon Cycle • All living things contain carbon. • Carbon is found in the atmosphere and dissolved in the oceans as carbon dioxide (CO 2) • Carbon, from the atmosphere, is used by plants to make carbohydrates through the process of photosynthesis • Carbon is returned to the environment through the process of cellular respiration.

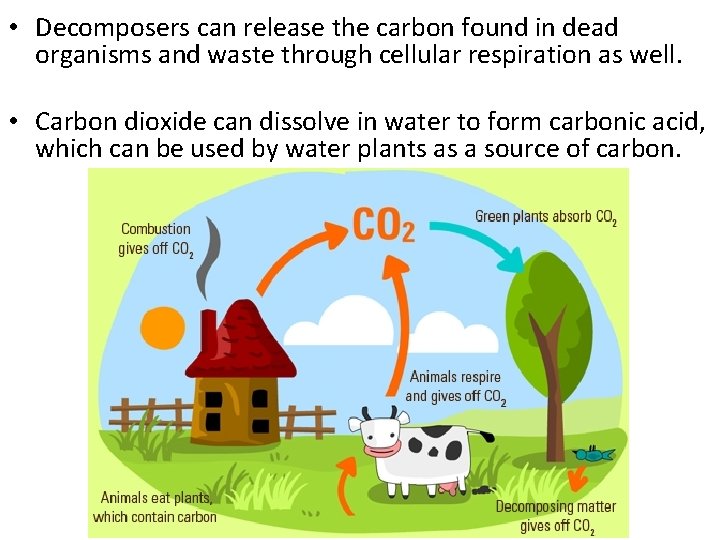
• Decomposers can release the carbon found in dead organisms and waste through cellular respiration as well. • Carbon dioxide can dissolve in water to form carbonic acid, which can be used by water plants as a source of carbon.


Carbon Deposits • Most of the earth’s carbon is stored in carbon rich deposits • Fossil fuels are the most valuable carbon deposits • It is also stored in limestone from millions of years of dead marine organisms • Carbon sinks are contained in plant tissues and dissolved in oceans

Human Activities and the Carbon Cycle • Humans increase the concentration of carbon dioxide in the atmosphere through burning fossil fuels • This causes climate change and alters average temperatures disrupting ecosystems • Deforestation reduces the amount of photosynthesis needed for absorption of carbon dioxide

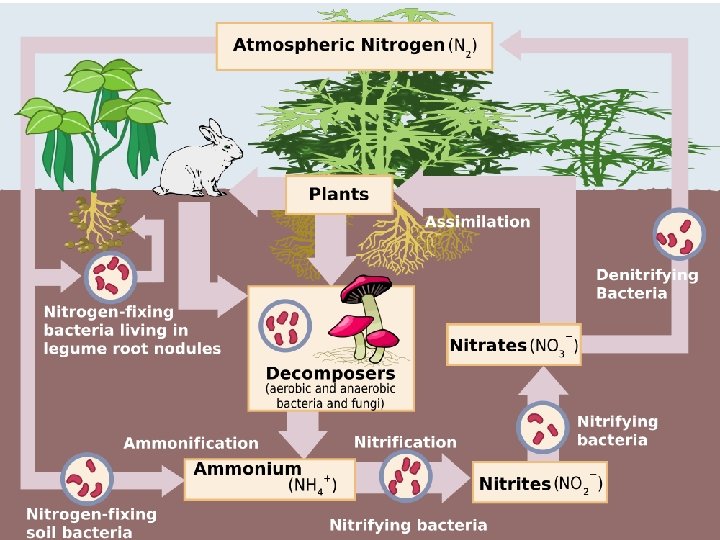
The Nitrogen Cycle • The atmosphere is composed of 78% nitrogen gas, N 2 but plants cannot use nitrogen in this form, it must be supplied in other forms: • ammonium ion (NH 4+) • nitrate ion (NO 3 -)

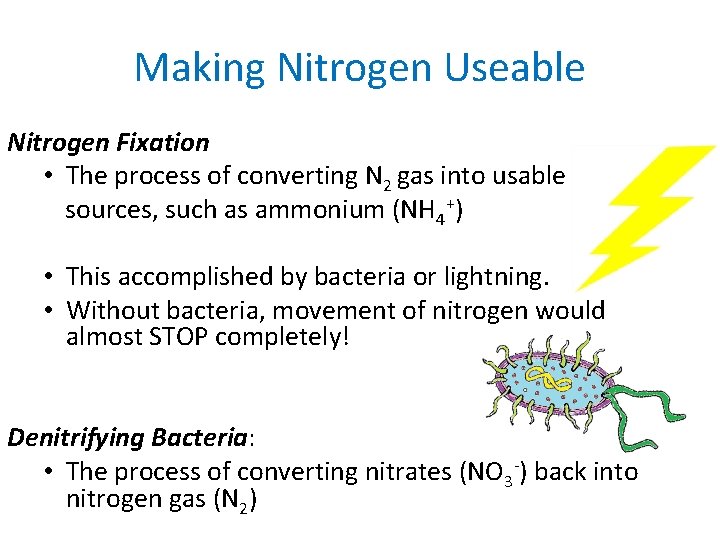
Making Nitrogen Useable Nitrogen Fixation • The process of converting N 2 gas into usable sources, such as ammonium (NH 4+) • This accomplished by bacteria or lightning. • Without bacteria, movement of nitrogen would almost STOP completely! Denitrifying Bacteria: • The process of converting nitrates (NO 3 -) back into nitrogen gas (N 2)


Class/Homework • WS “Cycling of Matter” • Pg. 51 #5, 7 - 10