Cyanotoxin Analysis Methods ASDWA Webinar November 26 2018

Cyanotoxin Analysis Methods ASDWA Webinar November 26, 2018 Heather Raymond Ohio EPA HAB Coordinator

2014 - City of Toledo “Do Not Drink” Advisory: Spotlight on Microcystins Methods 37

2014 City of Toledo Finished Drinking Water Microcystins Detections Raw = 14 ug/L Raw > 50 ug/L (ND) ND= Not Detected (Concentration <0. 30) Data Source: Toledo PWS

Analytical Method Comparison & Microcystin Variant Evaluation • 11 Sites: 4 up-ground reservoirs, 2 in-stream reservoirs, 2 Lake Erie locations, 2 canal-feeder lakes, and 1 river source • 22 samples from 2014 selected to help evaluate spatial and temporal variability within source waters • Variety of cyanobacteria genera represented • Each sample analyzed using 5 separate analytical methods

Methods Evaluated • Enzyme-Linked Immuno. Sorbent Assay (ELISA) Microcystin-ADDA Method • Liquid Chromatography (LC) – Ultraviolet (UV) • Liquid Chromatography(LC) –Tandem Mass Spectrometry (MS/MS) (individual variants) • LC-MS/MS (MMPB) • LC-UV and LC-MS (scan for variants without standards)

Microcystins Testing No “Perfect” Analytical Method for Detecting TOTAL Microcystins - Over 300 microcystin variants - Standards not available for majority of variants

ELISA Microcystins-ADDA Enzyme-Linked Immuno. Sorbent Assay (ELISA) Microcystin-ADDA Method (detection of antigen using an antibody) – Measures total microcystins (all variants/congeners, based on ADDA) – Highly selective/specific (for ADDA) – Certified by ETV Program – Moderately sensitive (RL: 0. 30 ug/L) – Suitable for raw & finished water – Quick (~four hours), useful for operational adjustments – Relatively inexpensive – Does not require high end equipment or expertise to run (can be used in water system lab) – Does not require pre-concentration solid phase extraction (SPE) step – Does not provide concentrations of specific microcystin variants – Is an indirect measure of the toxin – Non-linear curve: may require sample dilution and reanalysis if results out of range – Ohio EPA Standard Method 701. 0 & Lab Certification – U. S. EPA Method 546

Liquid Chromatography (LC) – Ultraviolet (UV) LC-UV - Liquid Chromatography separates components - Microcystins have UV absorption maxima at 238 nm - Non-selective detector; co-eluting interferents prevent accurate identification of components and quantitation - Less expensive than mass spectrometry - Less sensitive than mass spectrometry (average LOQ ~ 0. 3 µg/L) - ISO 20179 Standard Method

Liquid Chromatography(LC) –Tandem Mass Spectrometry (MS/MS) • LC/MS/MS – Highly specific identification of components (based on standards) – MS can identify a component in the presence of co-eluting interferents but quantitation may be compromised • Presence of co-eluting interferents can act to suppress or enhance response resulting in analytical bias • Sensitive (LOQ ~ 0. 02 µg/L) – “Weak” product ion abundance limits sensitivity. Requires preconcentration with SPE to augment sensitivity (LOQs < 0. 02 µg/L) • Preconcentrates NOM too – U. S. EPA Method 544 • Standard Method- includes QA/QC protocols and reduces variability in results between labs • Limited to 6 microcystin variants and finished water only – Expensive and requires highly skilled analysts – Issues with standard availability, purity, and variability
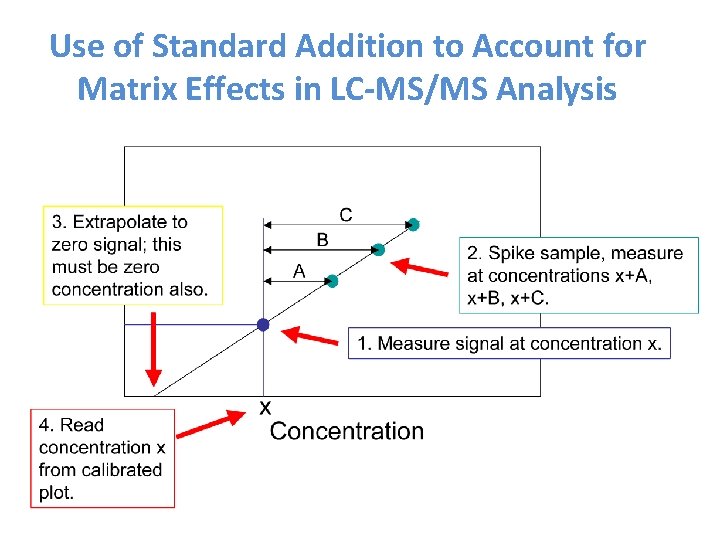
Use of Standard Addition to Account for Matrix Effects in LC-MS/MS Analysis

LC-MS/MS MMPB Method – MMPB (2 -methyl-3(methoxy)-4 -phenylbutyic acid) method analyzes the chemically cleaved Adda group common to all microcystin variants – Measures total microcystins (all variants, based on ADDA) – Quick (~2 hours, does not require freeze/thaw or sonication) – Sensitive (0. 05 ug/L) – Does not require standards for individual variants – Utilizes 4 PB internal standard – Suitable for raw water, some limitations with finished water – Does not provide data on individual variants – Requires oxidation step – Potential for detection of microcystins disinfection byproducts Toxicon 104 (2015) 91 -101 (Foss & Aubel): Using the MMPB technique to confirm microcystin concentrations in water measured by ELISA and HPLC (UV, MS/MS)
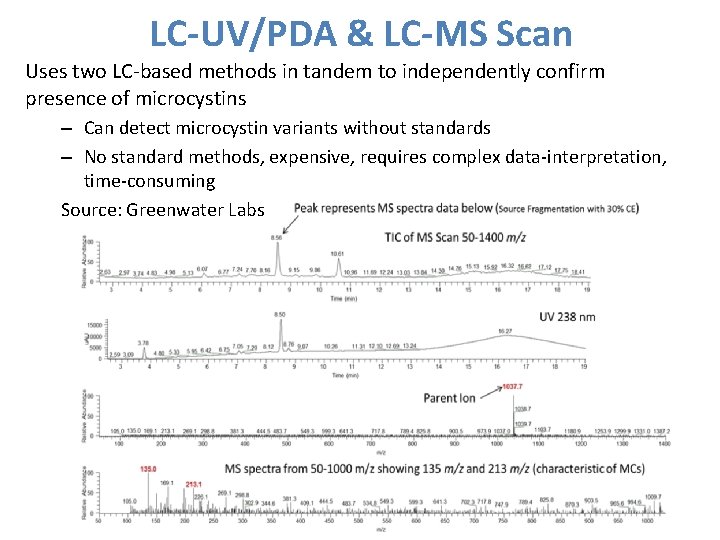
LC-UV/PDA & LC-MS Scan Uses two LC-based methods in tandem to independently confirm presence of microcystins – Can detect microcystin variants without standards – No standard methods, expensive, requires complex data-interpretation, time-consuming Source: Greenwater Labs
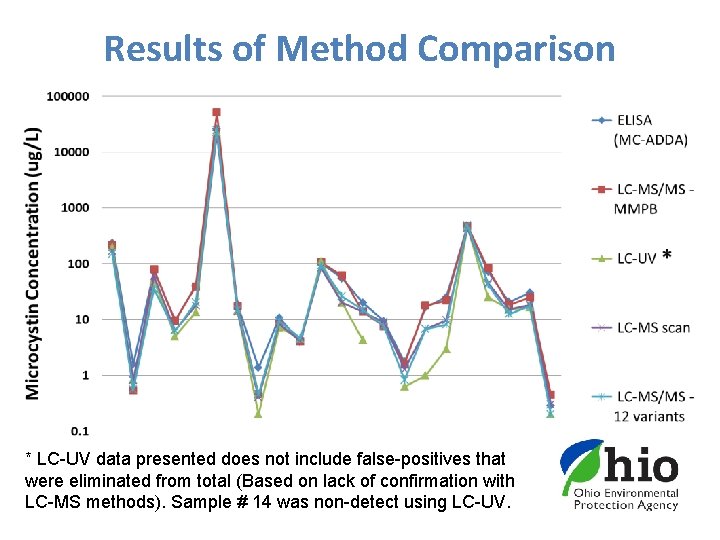
Results of Method Comparison * LC-UV data presented does not include false-positives that were eliminated from total (Based on lack of confirmation with LC-MS methods). Sample # 14 was non-detect using LC-UV.
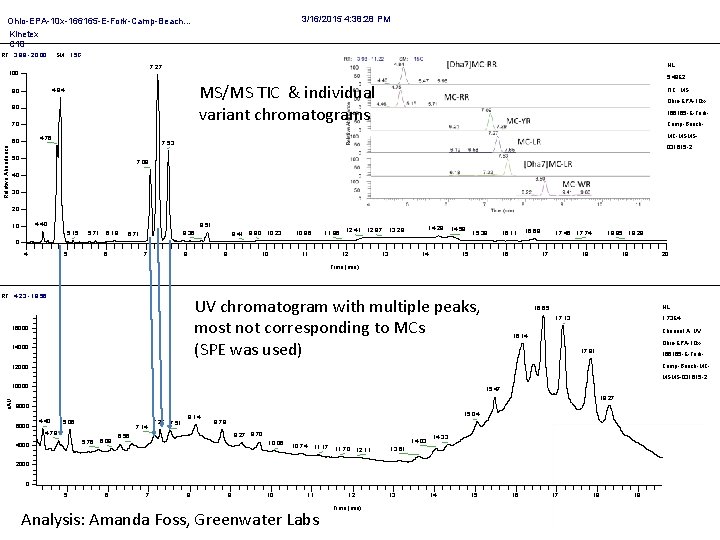
3/16/2015 4: 38: 28 PM Ohio-EPA-10 x-166165 -E-Fork-Camp-Beach. . . Kinetex C 18 RT: 3. 98 - 20. 00 SM: 15 G NL: 7. 27 100 5. 49 E 2 MS/MS TIC & individual variant chromatograms 4. 84 90 80 70 Relative Abundance Ohio-EPA-10 x 166165 -E-Fork. Camp-Beach. MC-MSMS- 4. 76 60 TIC MS 7. 53 50 031615 -2 7. 09 40 30 20 4. 40 10 8. 51 5. 15 5. 71 6. 19 8. 36 6. 71 9. 41 9. 80 10. 23 10. 96 11. 86 12. 41 12. 87 14. 28 13. 29 14. 59 15. 38 16. 11 16. 69 17. 45 17. 74 18. 95 19. 28 0 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Time (min) RT: 4. 23 - 19. 56 UV chromatogram with multiple peaks, most not corresponding to MCs (SPE was used) 16000 14000 NL: 16. 65 1. 73 E 4 17. 13 Channel A UV 16. 14 Ohio-EPA-10 x 17. 81 166165 -E-Fork. Camp-Beach-MC- 12000 MSMS-031615 -2 u. AU 10000 15. 47 18. 27 8000 6000 4. 40 5. 06 7. 14 4. 78 5. 76 4000 6. 08 7. 27 7. 51 8. 14 15. 04 8. 79 9. 27 9. 70 6. 56 10. 06 10. 74 11. 17 14. 03 11. 70 12. 11 14. 33 13. 61 2000 0 5 6 7 8 9 10 11 Analysis: Amanda Foss, Greenwater Labs 12 Time (min) 13 14 15 16 17 18 19
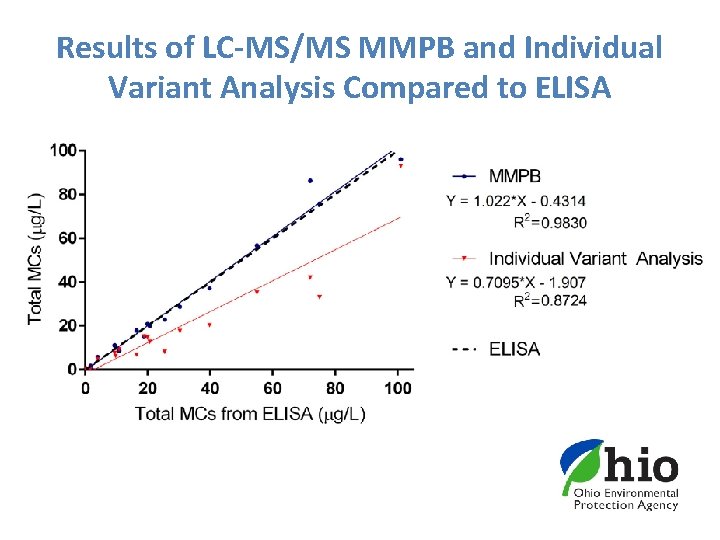
Results of LC-MS/MS MMPB and Individual Variant Analysis Compared to ELISA
![Inland Lake Microcystin Variants (Planktothrix) MC-Variant Site 1 Site 2 6/16/14 9/2/14 [DAsp 3] Inland Lake Microcystin Variants (Planktothrix) MC-Variant Site 1 Site 2 6/16/14 9/2/14 [DAsp 3]](http://slidetodoc.com/presentation_image_h/a9d131ca7a4797b934c84d8c4e75455b/image-16.jpg)
Inland Lake Microcystin Variants (Planktothrix) MC-Variant Site 1 Site 2 6/16/14 9/2/14 [DAsp 3] MC-RR 5. 3 6. 1 17. 5 [Dha 7] MC-LR 1. 1 1. 4 1. 5 MC-YR 0. 2 -0. 6 1. 2 MC-RR 0. 1 -0. 3 Inland Lake Microcystin Variants (Mixed Bloom) MC-Variant Site 1 Site 2 Site 3 6/18/14 7/9/14 6/30/14 [Dha 7] MC-RR 2. 9 3 -9 1. 0 0. 08 MC-RR 1. 4 39 1. 0 0. 01 -0. 03 MC-YR 1. 1 15 1. 0 MC-LR 4. 0 67 2. 4 0. 55 [DAsp 3] MC-LR 0. 6 18 0. 4 0. 03 [Dha 7] MC-LR 3. 6 1. 0 0. 05 MC-WR 0. 2 -0. 6 MC-LA 0. 2 -0. 6 MC-LY 0. 2 -0. 6 6 0. 2 -0. 6 0. 10
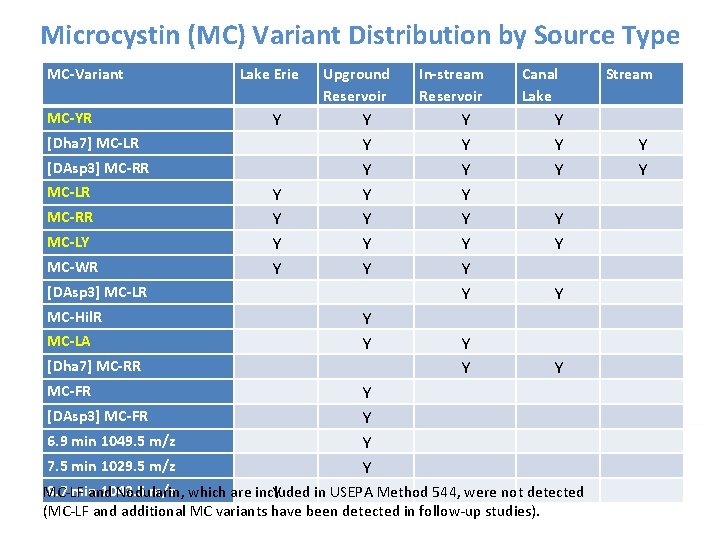
Microcystin (MC) Variant Distribution by Source Type MC-Variant Lake Erie Upground Reservoir In-stream Reservoir Canal Lake MC-YR Y Y [Dha 7] MC-LR Y Y Y [DAsp 3] MC-RR Y Y Y MC-LR Y Y Y MC-RR Y Y MC-LY Y Y MC-WR Y Y Y [DAsp 3] MC-LR Y Y MC-Hil. R Y MC-LA Y Y [Dha 7] MC-RR Y Y MC-FR Y [DAsp 3] MC-FR Y 6. 9 min 1049. 5 m/z Y 7. 5 min 1029. 5 m/z Y 8. 7 min 1043. 5 m/z MC-LF and Nodularin, which are included in USEPA Method 544, were not detected Y (MC-LF and additional MC variants have been detected in follow-up studies). Stream Y Y

Key Findings • 16 different MC-variants were detected • MC-LR was only detected at 5 of 11 sites (45%) • Most common variants were: MC-YR, [Dha 7] MC-LR and [DAsp 3] MC-RR • LC-PDA methods prone to interference, potential for false positives and false negatives • LC-MS/MS MMPB method helped confirm ELISA results • 91% of samples had MC-variants not detectable by U. S. EPA Method 544 (including dominant MC-variant in some samples) • LC-MS/MS individual variant analysis under-reported total microcystins, based on MMPB and LC-UV/MS scan data
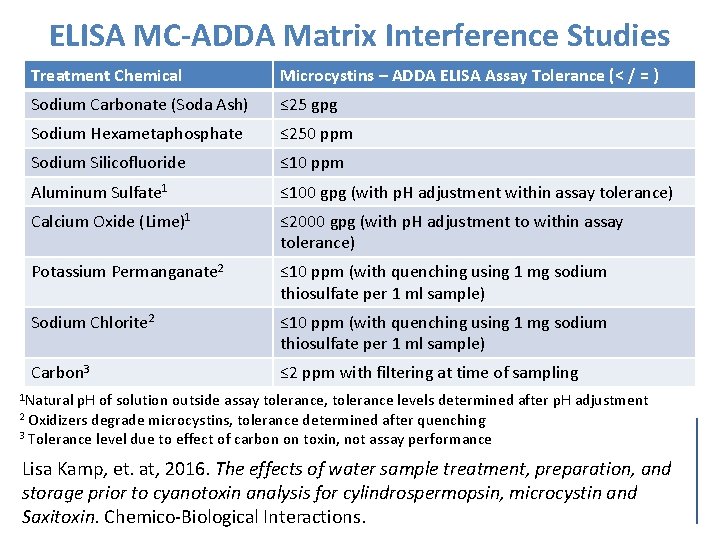
ELISA MC-ADDA Matrix Interference Studies Treatment Chemical Microcystins – ADDA ELISA Assay Tolerance (< / = ) Sodium Carbonate (Soda Ash) ≤ 25 gpg Sodium Hexametaphosphate ≤ 250 ppm Sodium Silicofluoride ≤ 10 ppm Aluminum Sulfate 1 ≤ 100 gpg (with p. H adjustment within assay tolerance) Calcium Oxide (Lime)1 ≤ 2000 gpg (with p. H adjustment to within assay tolerance) Potassium Permanganate 2 ≤ 10 ppm (with quenching using 1 mg sodium thiosulfate per 1 ml sample) Sodium Chlorite 2 ≤ 10 ppm (with quenching using 1 mg sodium thiosulfate per 1 ml sample) Carbon 3 ≤ 2 ppm with filtering at time of sampling 1 Natural p. H of solution outside assay tolerance, tolerance levels determined after p. H adjustment 2 Oxidizers degrade microcystins, tolerance determined after quenching 3 Tolerance level due to effect of carbon on toxin, not assay performance Lisa Kamp, et. at, 2016. The effects of water sample treatment, preparation, and storage prior to cyanotoxin analysis for cylindrospermopsin, microcystin and Saxitoxin. Chemico-Biological Interactions.

ELISA MC-ADDA Matrix Interference Studies by U. S. EPA as part of ELISA MC-ADDA Method Development for UCMR 4: • Storage Stability – Holding Times • Sample Preservation and Container Studies • Matrix Interference Studies -Microcystins Variant Fortified Sample Studies (finished water, raw water, reagent water with chemical addition, etc. ) -Dilution Experiments (real world raw/finished water samples) • U. S. EPA Method Validation & Interlab Validation LC-MS/MS MMPB Method Finished Water Matrix Evaluation: • Concern regarding detection of microcystins disinfection byproducts

Analytical Methods Utilized by Ohio EPA Microcystins (μg/L) Cylindrospermopsin (μg/L) Saxitoxins (μg/L) Anatoxin-a (μg/L) Surveillance sampling ELISA (MC-ADDA) ELISA LC-MS/MS Repeat sampling in response to a finished water detection ELISA (MC-ADDA) LC-MS/MS ELISA: Enzyme-Linked Immunosorbent Assay LC-MS/MS: Liquid Chromatography followed by tandem Mass Spectrometry

Ohio HAB Response Policy 2010: Ohio EPA began incident-response sampling for cyanotoxins 2011: Ohio EPA/ODNR/ODH created Ohio HAB Response Strategy 2013 & 2014: Drinking Water Advisories due to Microcystins 2015: USEPA Established Health Advisories for Microcystins and Cylindrospermopsin & Ohio Senate Bill 1 passed § NSF HAB Workshop (Bowling Green) & Akron HAB Conference- April, 2015 2016: HAB Monitoring and Treatment Rules Effective June, 2016: § OAC Chapter 3745 -90: epa. ohio. gov/ddagw/rules. aspx § Microcystins and Cyanobacteria Screening Monitoring Requirements § Lab Certification (ELISA and q. PCR) § Microcystins Method: epa. ohio. gov/Portals/28/documents/labcert/Total. Microcystins. pdf § Cyanobacteria Screening q. PCR Method: https: //epa. ohio. gov/Portals/28/documents/habs/705. 0 -q. PCR. pdf § Treatment technique requirements

Ohio HAB Monitoring Requirements • Total Microcystins (ELISA) May – October – Weekly raw and finished water – Raw water detections >5 ug/L and any finished water detections trigger additional sampling November – April – Raw water only every other week – Detections trigger additional monitoring • Cyanobacteria Screening (q. PCR) All year – Biweekly raw water – Triggers follow up sampling by Ohio EPA for saxitoxins and cylindrospermopsin Rules provide flexibility for reduced monitoring

Cyanobacteria Screening: Molecular Methods (Multiplex q. PCR) • Multiplex quantitative polymerase chain reaction (q. PCR) assay – identifies and quantifies the presence of genes unique to: – – – • Cyanobacteria: 16 S r. DNA • Microcystins and Nodularin production: mcy. E gene • Cylindrospermopsin production: cyr. A gene • Saxitoxins production: sxt. A gene Fast: 2 -3 hours Scalable Cost-effective Utilizes certified reference material Specific • Ohio EPA DES 705. 0 Method and lab certification • www. phytoxigene. com (current assay in use) • Ohio EPA method evaluation study

q. PCR as Screening Tool for Microcystins § Microcystins detected in raw water at 45 PWSs (38%) and mcy. E detected at 57 PWSs (48%) § Out of 1850 paired PWS samples: § 100% of microcystins >1. 6 µg/L had paired mcy. E gene detections. § 100% of microcystins >5 µg/L had mcy. E detections > 5 gc/µL § 90% of microcystins detections >1. 6 ug/L had mcy. E detections >5 gc/µL § Less than 2% of samples (22 sites, 32 samples) had microcystins detections without mcy. E detections: § 19 of the 22 sites had gene detections in either prior or post sampling events § The remaining three PWSs had only one low level (0. 35 – 0. 44 µg/L) microcystins detection in 2016; all had trace mcy. E gene copies
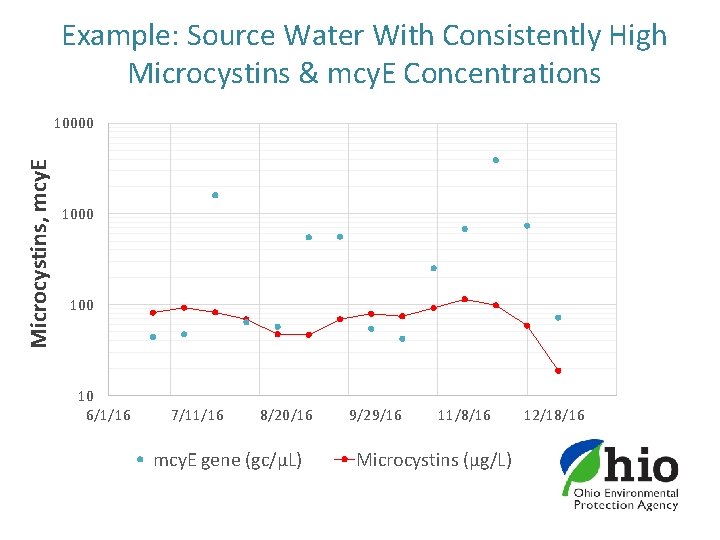
Example: Source Water With Consistently High Microcystins & mcy. E Concentrations Microcystins, mcy. E 10000 100 10 6/1/16 7/11/16 8/20/16 mcy. E gene (gc/µL) 9/29/16 11/8/16 Microcystins (µg/L) 12/18/16
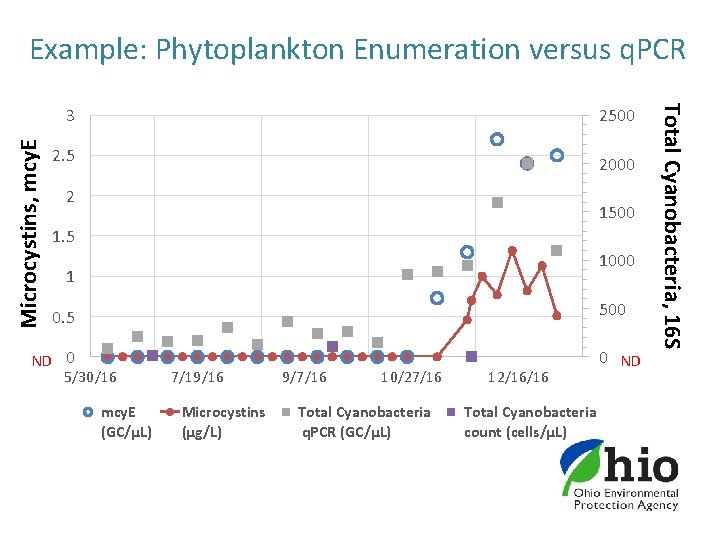
3 2500 2. 5 2000 2 1500 1. 5 1000 1 500 0. 5 ND 0 5/30/16 mcy. E (GC/µL) 7/19/16 Microcystins (µg/L) 9/7/16 10/27/16 Total Cyanobacteria q. PCR (GC/µL) 12/16/16 Total Cyanobacteria count (cells/µL) 0 ND Total Cyanobacteria, 16 S Microcystins, mcy. E Example: Phytoplankton Enumeration versus q. PCR
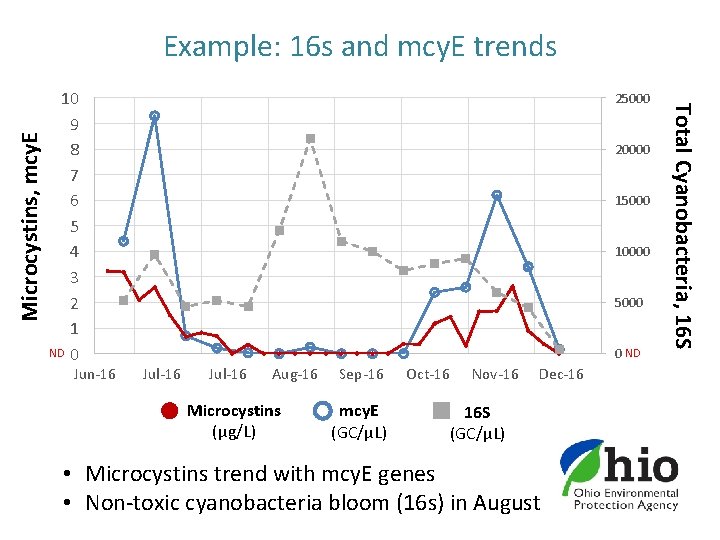
10 9 8 7 6 5 4 3 2 1 ND 0 Jun-16 25000 20000 15000 10000 5000 0 ND Jul-16 Aug-16 Microcystins (µg/L) Sep-16 mcy. E (GC/µL) Oct-16 Nov-16 Dec-16 16 S (GC/µL) • Microcystins trend with mcy. E genes • Non-toxic cyanobacteria bloom (16 s) in August Total Cyanobacteria, 16 S Microcystins, mcy. E Example: 16 s and mcy. E trends

q. PCR as Screening Tool for Saxitoxins and Cylindrospermopsin § sxt. A detections triggered response sampling at 33 PWSs (22%), including many with no historic saxitoxins occurrence and three Lake Erie intakes. § 15 of those PWSs detected saxitoxins in raw water (12%) § 6 PWSs had finished water detections (none above thresholds) § Less than 1% percent of samples had saxitoxins detections without corresponding gene detections (includes 168 paired inland lakes samples) § Only one cyr. A detection, no cylindrospermopsin detections in 2016 § cyr. A detections at 2 PWSs and cylindrospermopsin detections at one of those PWSs in 2017
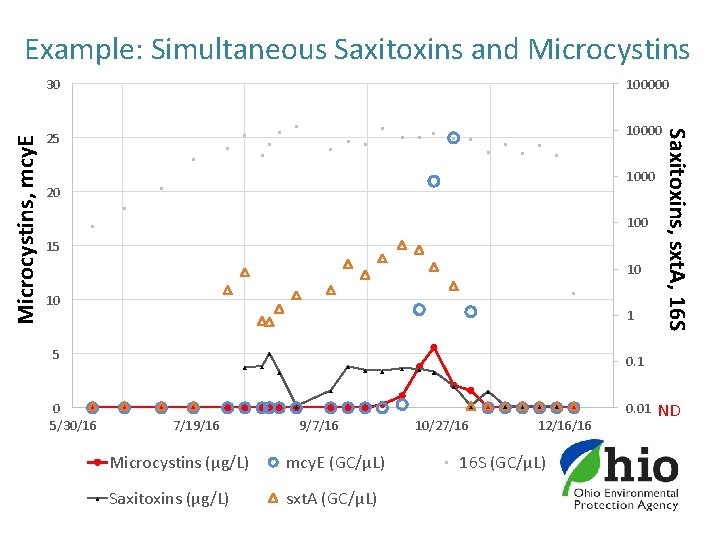
Example: Simultaneous Saxitoxins and Microcystins 100000 10000 25 1000 20 100 15 10 10 1 5 0 5/30/16 Saxitoxins, sxt. A, 16 S Microcystins, mcy. E 30 0. 1 0. 01 7/19/16 9/7/16 Microcystins (µg/L) mcy. E (GC/µL) Saxitoxins (µg/L) sxt. A (GC/µL) 10/27/16 12/16/16 16 S (GC/µL) ND
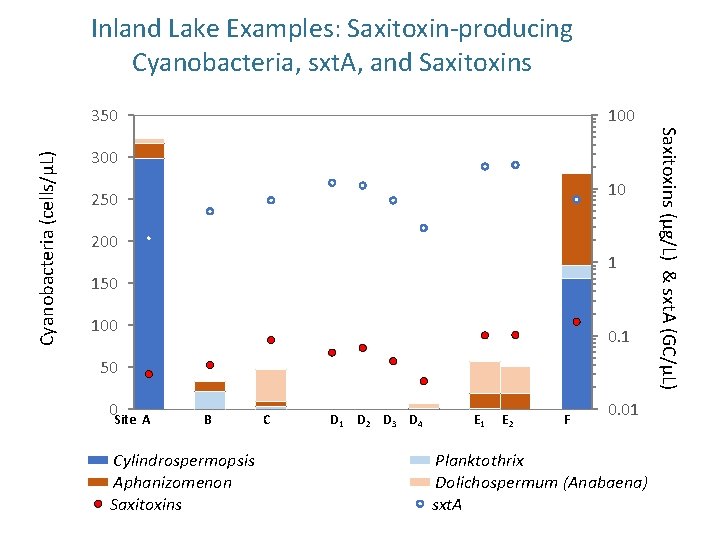
Inland Lake Examples: Saxitoxin-producing Cyanobacteria, sxt. A, and Saxitoxins 100 300 10 250 200 1 150 100 0. 1 50 0 Site A B Cylindrospermopsis Aphanizomenon Saxitoxins C D 1 D 2 D 3 D 4 E 1 E 2 F 0. 01 Planktothrix Dolichospermum (Anabaena) sxt. A Saxitoxins (µg/L) & sxt. A (GC/µL) Cyanobacteria (cells/µL) 350

Example: Use of q. PCR to Help Identify Source of Saxitoxins and Target Reservoir Management • Low concentrations of saxitoxins detected in drinking water (below Ohio EPA threshold) from late July – September, 2015. • Extracellular saxitoxins predominated all samples. • 10 different potential saxitoxin-producing genera found in multiple habitat zones (pelagic, benthic, periphyton, etc. ) in multiple (all sampling) locations. • Follow-up q. PCR analysis revealed highest sxt. A gene copies in benthic samples. • q. PCR data used to target algaecide application.

q. PCR Summary § mcy. E is an effective, specific, screen for microcystins and may be useful as an early warning tool. § sxt. A is an effective, specific, screen for saxitoxins. § Multi-plex functionality of assay works. § 16 S is not an effective screen for cyanotoxins, but potentially useful for assessing susceptibility § q. PCR is superior to phytoplankton enumeration as a screen for saxitoxins and provides a more targeted screen for microcystins. § q. PCR data can help inform reservoir management strategies.

Questions? www. epa. ohio. gov/ddagw/HAB. aspx Heather. Raymond@epa. ohio. gov (614) 644 -2752
- Slides: 34