CWORTHY Study Part C grazoprevir elbasvir RBV in
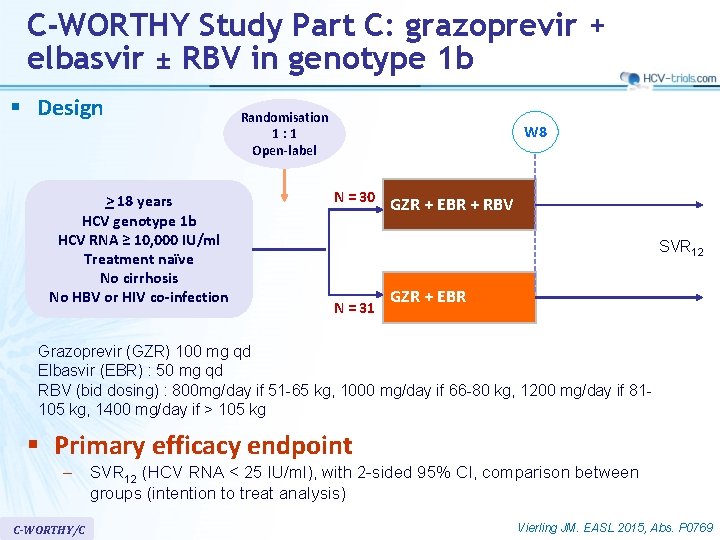
C-WORTHY Study Part C: grazoprevir + elbasvir ± RBV in genotype 1 b § Design > 18 years HCV genotype 1 b HCV RNA ≥ 10, 000 IU/ml Treatment naïve No cirrhosis No HBV or HIV co-infection Randomisation 1: 1 Open-label W 8 N = 30 GZR + EBR + RBV SVR 12 N = 31 GZR + EBR Grazoprevir (GZR) 100 mg qd Elbasvir (EBR) : 50 mg qd RBV (bid dosing) : 800 mg/day if 51 -65 kg, 1000 mg/day if 66 -80 kg, 1200 mg/day if 81105 kg, 1400 mg/day if > 105 kg § Primary efficacy endpoint – SVR 12 (HCV RNA < 25 IU/ml), with 2 -sided 95% CI, comparison between groups (intention to treat analysis) C-WORTHY/C Vierling JM. EASL 2015, Abs. P 0769
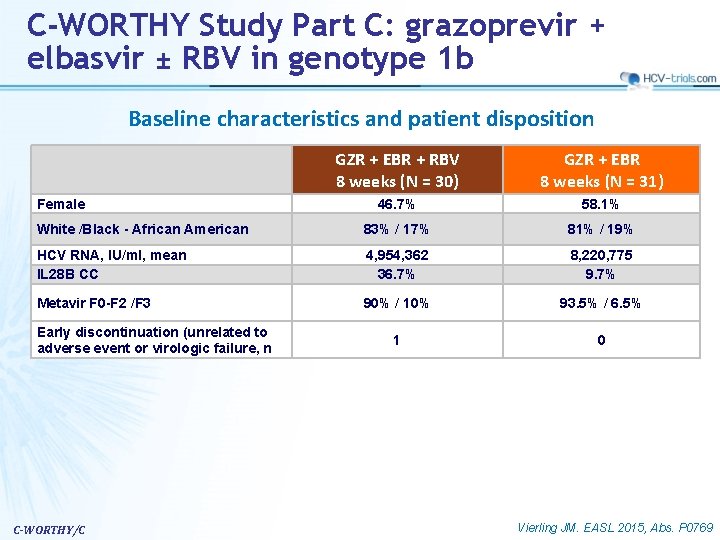
C-WORTHY Study Part C: grazoprevir + elbasvir ± RBV in genotype 1 b Baseline characteristics and patient disposition GZR + EBR + RBV 8 weeks (N = 30) GZR + EBR 8 weeks (N = 31) 46. 7% 58. 1% White /Black - African American 83% / 17% 81% / 19% HCV RNA, IU/ml, mean IL 28 B CC 4, 954, 362 36. 7% 8, 220, 775 9. 7% Metavir F 0 -F 2 /F 3 90% / 10% 93. 5% / 6. 5% 1 0 Female Early discontinuation (unrelated to adverse event or virologic failure, n C-WORTHY/C Vierling JM. EASL 2015, Abs. P 0769
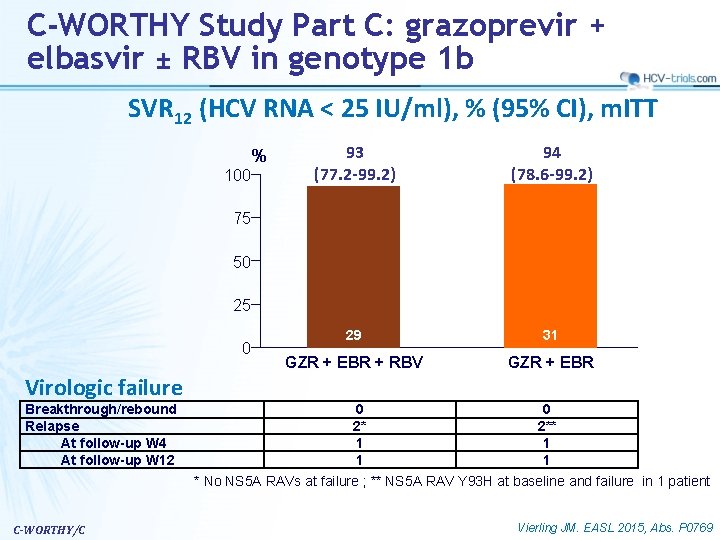
C-WORTHY Study Part C: grazoprevir + elbasvir ± RBV in genotype 1 b SVR 12 (HCV RNA < 25 IU/ml), % (95% CI), m. ITT % 100 93 (77. 2 -99. 2) 94 (78. 6 -99. 2) 29 31 GZR + EBR + RBV GZR + EBR 75 50 25 0 Virologic failure Breakthrough/rebound Relapse At follow-up W 4 At follow-up W 12 C-WORTHY/C 0 0 2* 2** 1 1 * No NS 5 A RAVs at failure ; ** NS 5 A RAV Y 93 H at baseline and failure in 1 patient Vierling JM. EASL 2015, Abs. P 0769
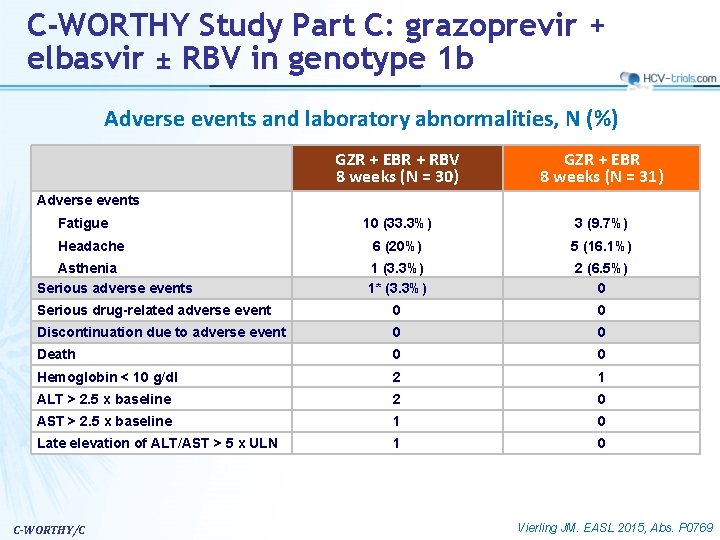
C-WORTHY Study Part C: grazoprevir + elbasvir ± RBV in genotype 1 b Adverse events and laboratory abnormalities, N (%) GZR + EBR + RBV 8 weeks (N = 30) GZR + EBR 8 weeks (N = 31) 10 (33. 3%) 3 (9. 7%) 6 (20%) 5 (16. 1%) 1 (3. 3%) 1* (3. 3%) 2 (6. 5%) 0 Serious drug-related adverse event 0 0 Discontinuation due to adverse event 0 0 Death 0 0 Hemoglobin < 10 g/dl 2 1 ALT > 2. 5 x baseline 2 0 AST > 2. 5 x baseline 1 0 Late elevation of ALT/AST > 5 x ULN 1 0 Adverse events Fatigue Headache Asthenia Serious adverse events C-WORTHY/C Vierling JM. EASL 2015, Abs. P 0769

C-WORTHY Study Part C: grazoprevir + elbasvir ± RBV in genotype 1 b § Summary – GZR + EBR ± RBV for 8 weeks in treatment-naïve, non-cirrhotic patients with HCV genotype 1 b infection was • highly efficacious • safe and well-tolerated • rarely associated with resistance-associated variants at the time of failure C-WORTHY/C Vierling JM. EASL 2015, Abs. P 0769
- Slides: 5