CWORTHY Study grazoprevir elbasvir ribavirin for genotype 1

C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 § Design Randomisation* Open-label > 18 years HCV genotype 1, treatment naïve HCV RNA ≥ 10, 000 IU/ml No cirrhosis ± HIV infection, on 2 NRTI + RAL > 8 weeks and undetectable HIV RNA > 24 weeks and CD 4 ≥ 300/mm 3 § Dosage of study drugs Part A : HCV mono-infected patients Part B : HCV mono-infected and HCV-HIV coinfected patients * Patients and investigators masked to group assignment – Grazoprevir (GZR) 100 mg qd – Elbasvir (EBR) : 20 or 50 mg qd – RBV (bid dosing) : 800 mg/day if 51 -65 kg, 1000 mg/day if 66 -80 kg, 1200 mg/day if 81 -105 kg, 1400 mg/day if > 105 kg § Primary efficacy endpoint – SVR 12 (HCV RNA < 25 IU/ml), with 2 -sided 95% CI, comparison between groups C-WORTHY Sulkowski M. Lancet 2015; 385: 1087 -97
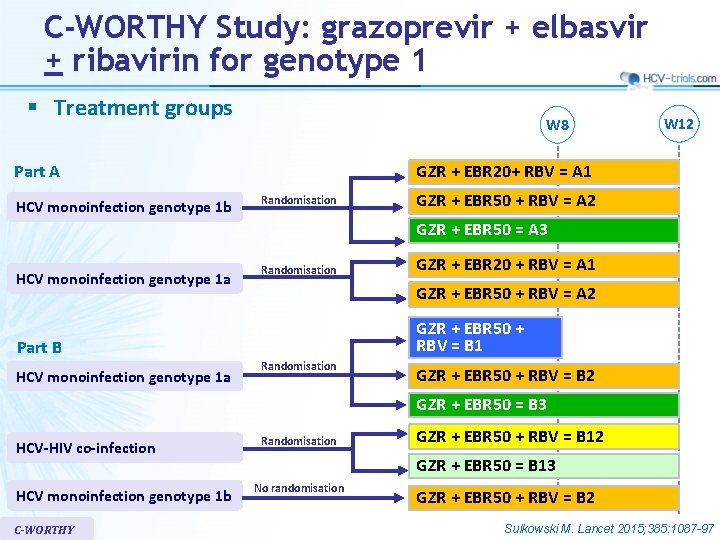
C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 § Treatment groups W 8 GZR + EBR 20+ RBV = A 1 Part A HCV monoinfection genotype 1 b W 12 Randomisation GZR + EBR 50 + RBV = A 2 GZR + EBR 50 = A 3 HCV monoinfection genotype 1 a Randomisation GZR + EBR 50 + RBV = A 2 GZR + EBR 50 + RBV = B 1 Part B HCV monoinfection genotype 1 a GZR + EBR 20 + RBV = A 1 Randomisation GZR + EBR 50 + RBV = B 2 GZR + EBR 50 = B 3 HCV-HIV co-infection HCV monoinfection genotype 1 b C-WORTHY Randomisation GZR + EBR 50 + RBV = B 12 GZR + EBR 50 = B 13 No randomisation GZR + EBR 50 + RBV = B 2 Sulkowski M. Lancet 2015; 385: 1087 -97
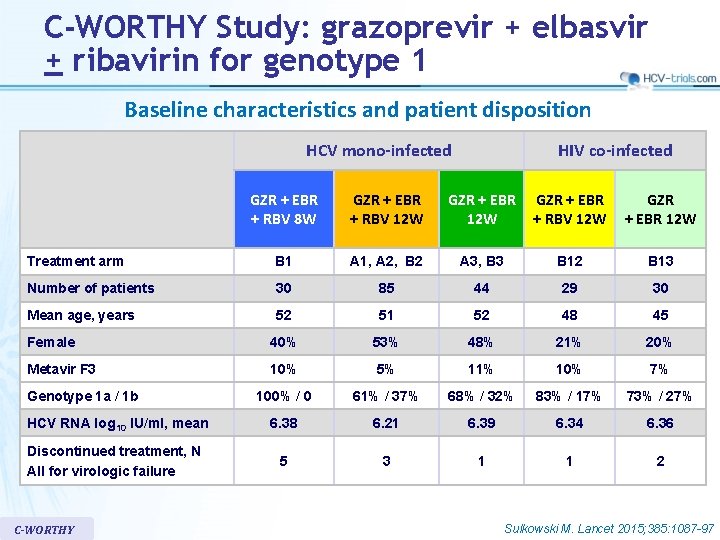
C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 Baseline characteristics and patient disposition HCV mono-infected HIV co-infected GZR + EBR + RBV 8 W GZR + EBR + RBV 12 W GZR + EBR 12 W Treatment arm B 1 A 1, A 2, B 2 A 3, B 3 B 12 B 13 Number of patients 30 85 44 29 30 Mean age, years 52 51 52 48 45 Female 40% 53% 48% 21% 20% Metavir F 3 10% 5% 11% 10% 7% 100% / 0 61% / 37% 68% / 32% 83% / 17% 73% / 27% HCV RNA log 10 IU/ml, mean 6. 38 6. 21 6. 39 6. 34 6. 36 Discontinued treatment, N All for virologic failure 5 3 1 1 2 Genotype 1 a / 1 b C-WORTHY Sulkowski M. Lancet 2015; 385: 1087 -97
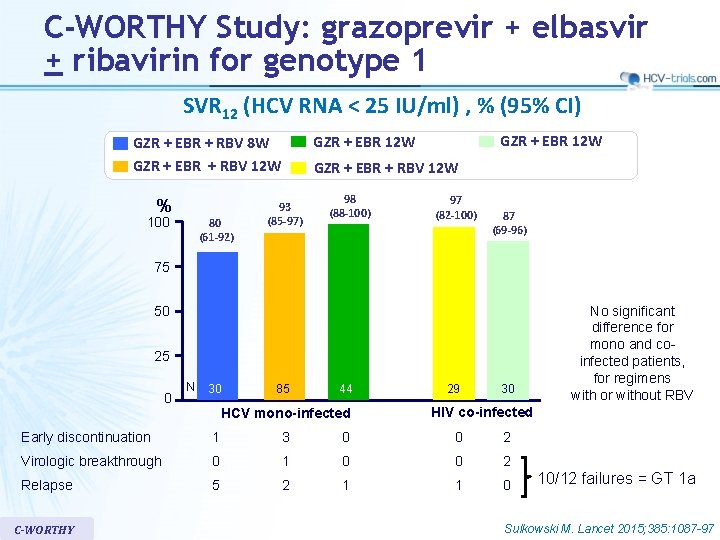
C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 SVR 12 (HCV RNA < 25 IU/ml) , % (95% CI) % 100 80 (61 -92) GZR + EBR 12 W GZR + EBR + RBV 8 W GZR + EBR + RBV 12 W 93 (85 -97) 98 (88 -100) 97 (82 -100) 87 (69 -96) 75 50 25 0 N 30 85 44 HCV mono-infected 29 30 HIV co-infected Early discontinuation 1 3 0 0 2 Virologic breakthrough 0 1 0 0 2 Relapse 5 2 1 1 0 C-WORTHY No significant difference for mono and coinfected patients, for regimens with or without RBV 10/12 failures = GT 1 a Sulkowski M. Lancet 2015; 385: 1087 -97
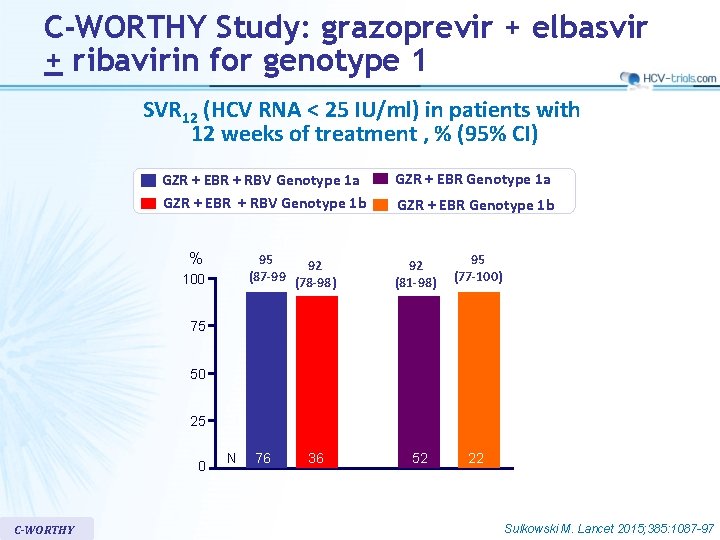
C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 SVR 12 (HCV RNA < 25 IU/ml) in patients with 12 weeks of treatment , % (95% CI) GZR + EBR + RBV Genotype 1 a GZR + EBR + RBV Genotype 1 b % 95 92 (87 -99 (78 -98) 100 GZR + EBR Genotype 1 a GZR + EBR Genotype 1 b 92 (81 -98) 95 (77 -100) 75 50 25 0 C-WORTHY N 76 36 52 22 Sulkowski M. Lancet 2015; 385: 1087 -97
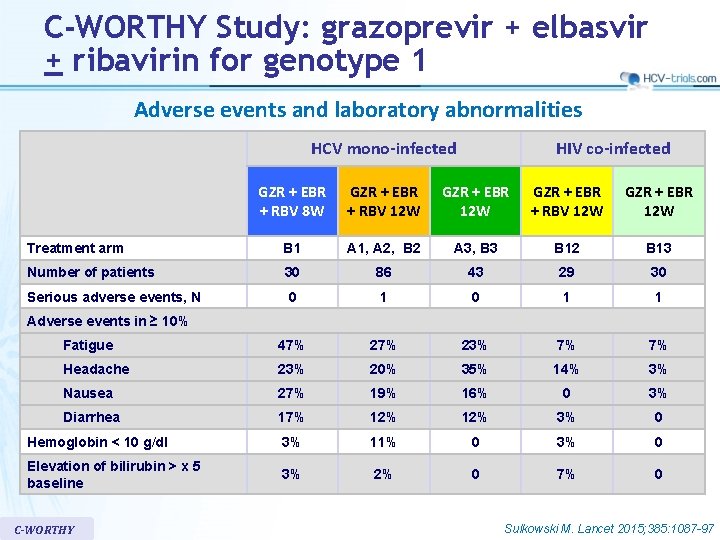
C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 Adverse events and laboratory abnormalities HCV mono-infected HIV co-infected GZR + EBR + RBV 8 W GZR + EBR + RBV 12 W GZR + EBR 12 W Treatment arm B 1 A 1, A 2, B 2 A 3, B 3 B 12 B 13 Number of patients 30 86 43 29 30 Serious adverse events, N 0 1 1 Fatigue 47% 23% 7% 7% Headache 23% 20% 35% 14% 3% Nausea 27% 19% 16% 0 3% Diarrhea 17% 12% 3% 0 Hemoglobin < 10 g/dl 3% 11% 0 3% 0 Elevation of bilirubin > x 5 baseline 3% 2% 0 7% 0 Adverse events in ≥ 10% C-WORTHY Sulkowski M. Lancet 2015; 385: 1087 -97

C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 SVR 12 according to detection of resistance-associated variants at baseline NS 3 variants at baseline NS 5 A variants at baseline NO YES N 141 75 191 25 SVR 12 (HCV RNA < 25 IU/ml) 92% 91% 95%* 68% * Mutations detected Most prevalent NS 3 variants at failure : Y 56 H A 156 T D 168 A/N Most prevalent NS 5 A variants at failure : Q 30 R/H L 31 M Y 93 H/N * p < 0. 001 C-WORTHY Sulkowski M. Lancet 2015; 385: 1087 -97

C-WORTHY Study: grazoprevir + elbasvir + ribavirin for genotype 1 § Summary – In this phase II study, 12 weeks of oral treatment with grazoprevir and elbasvir, with or without RBV, was well tolerated and led to high rates of SVR 12 (87– 98%) in previously untreated patients without cirrhosis with HCV genotype 1 monoinfection or HIV/HCV co-infection • • • C-WORTHY The addition of RBV was not associated with increased rates of SVR 12 Similar efficacy was seen in patients with genotype 1 a and 1 b SVR 12 was similar with 12 weeks of treatment in mono and co-infected patients 8 weeks of GZR + EBR + RBV led to suboptimal SVR 12, mainly because of relapse Patients with NS 5 A baseline resistance-associated variants had lower SVR 12 Virological relapse was associated with emergence of resistance-associated variants to one or both of the drugs (GZR, EBR) in the regimen Treatment-emergent, clinically significant, adverse events were infrequent No discontinuation for adverse event In HIV co-infected patients, adverse events were less frequently reported, and HIV suppression was maintained Sulkowski M. Lancet 2015; 385: 1087 -97
- Slides: 8