CURRY Study SOF RBV for HCV with liver
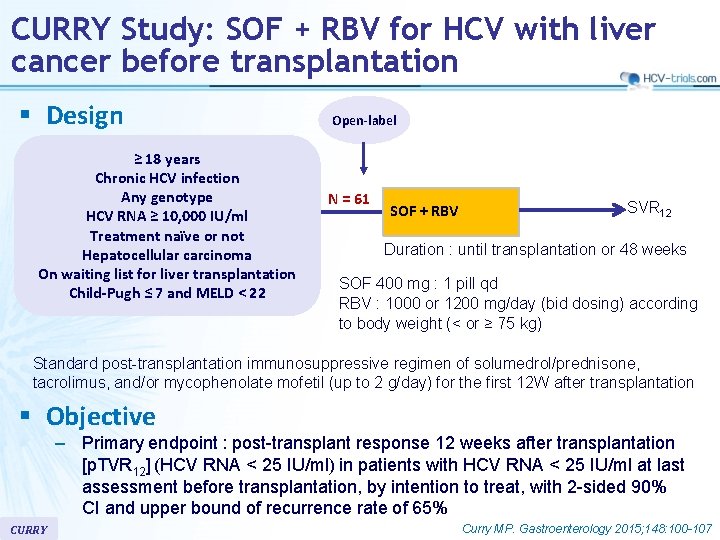
CURRY Study: SOF + RBV for HCV with liver cancer before transplantation § Design ≥ 18 years Chronic HCV infection Any genotype HCV RNA ≥ 10, 000 IU/ml Treatment naïve or not Hepatocellular carcinoma On waiting list for liver transplantation Child-Pugh ≤ 7 and MELD < 22 Open-label N = 61 SOF + RBV SVR 12 Duration : until transplantation or 48 weeks SOF 400 mg : 1 pill qd RBV : 1000 or 1200 mg/day (bid dosing) according to body weight (< or ≥ 75 kg) Standard post-transplantation immunosuppressive regimen of solumedrol/prednisone, tacrolimus, and/or mycophenolate mofetil (up to 2 g/day) for the first 12 W after transplantation § Objective – Primary endpoint : post-transplant response 12 weeks after transplantation [p. TVR 12] (HCV RNA < 25 IU/ml) in patients with HCV RNA < 25 IU/ml at last assessment before transplantation, by intention to treat, with 2 -sided 90% CI and upper bound of recurrence rate of 65% CURRY Curry MP. Gastroenterology 2015; 148: 100 -107
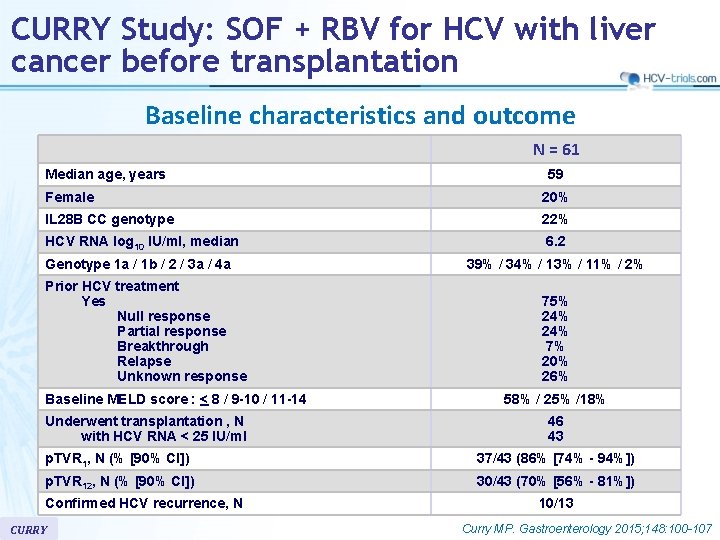
CURRY Study: SOF + RBV for HCV with liver cancer before transplantation Baseline characteristics and outcome N = 61 Median age, years 59 Female 20% IL 28 B CC genotype 22% HCV RNA log 10 IU/ml, median 6. 2 Genotype 1 a / 1 b / 2 / 3 a / 4 a 39% / 34% / 13% / 11% / 2% Prior HCV treatment Yes Null response Partial response Breakthrough Relapse Unknown response Baseline MELD score : < 8 / 9 -10 / 11 -14 Underwent transplantation , N with HCV RNA < 25 IU/ml 75% 24% 7% 20% 26% 58% / 25% /18% 46 43 p. TVR 1, N (% [90% CI]) 37/43 (86% [74% - 94%]) p. TVR 12, N (% [90% CI]) 30/43 (70% [56% - 81%]) Confirmed HCV recurrence, N CURRY 10/13 Curry MP. Gastroenterology 2015; 148: 100 -107
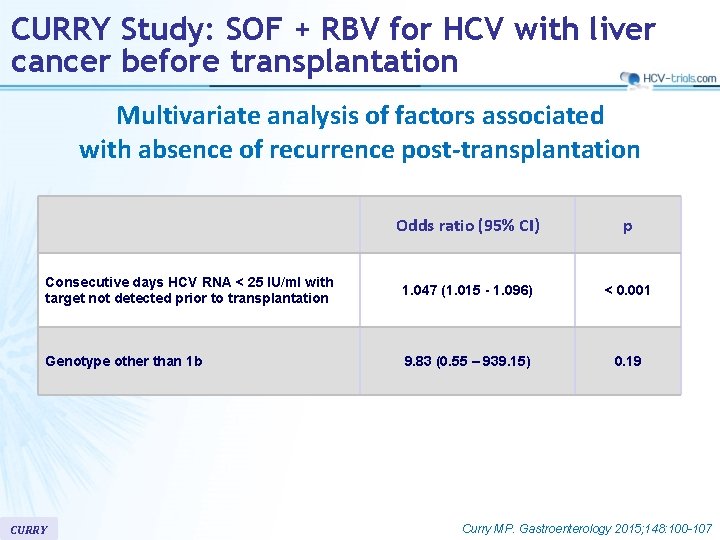
CURRY Study: SOF + RBV for HCV with liver cancer before transplantation Multivariate analysis of factors associated with absence of recurrence post-transplantation Odds ratio (95% CI) p Consecutive days HCV RNA < 25 IU/ml with target not detected prior to transplantation 1. 047 (1. 015 - 1. 096) < 0. 001 Genotype other than 1 b 9. 83 (0. 55 – 939. 15) 0. 19 CURRY Curry MP. Gastroenterology 2015; 148: 100 -107

CURRY Study: SOF + RBV for HCV with liver cancer before transplantation § HCV Recurrence and resistance analysis – 10 confirmed recurrence after liver transplantation • Genotype 1 a, N = 2; 1 b, N = 7; 3 a, N = 1 • ILB 28 non-CC, N = 10 • Recurrence at W 1 (N = 3), W 2 (N = 2), W 4 (N = 4), or W 12 (N = 1) posttransplantation • Duration of SOF+ RBV pre-transplantation < 12 weeks , N = 4 – NS 5 B sequencing • At baseline : – 4 patients with L 159 F variant : 4/4 relapsed – 1 patient with N 142 T : achieved SVR 12 • 29 patients with failure before transplantation or recurrence after transplantation – no S 282 T mutant – 12 patients with other variants as minor subpopulations (< 10%) in 11/12 : N 142 T (N = 2), L 159 F (N = 5), S 282 G (N = 1), L 230 F (N = 3); L 159 F + S 282 R + L 230 F + V 321 A (N = 1) CURRY Curry MP. Gastroenterology 2015; 148: 100 -107

CURRY Study: SOF + RBV for HCV with liver cancer before transplantation § Adverse events – – Median duration of exposure to study regimen : 21 weeks Serious adverse events, N = 11 ≥ Grade 3 adverse event, N = 11 Discontinuation due to adverse event, N = 2 (pneumonia, sepsis/acute renal failure) – Most common adverse events : • • • Fatigue (38%) Headache (23%) Anemia (21%) Nausea (16%) Rash (15%) Dyspnea (11%) Cough (11%) Insomnia (11%) Constipation (10%) Pruritus (10%) – Most common grade 3 -4 laboratory abnormalities : grade 3 decrease in hemoglobin level, grade 3 hyperglycemia, grade 3 -4 bilirubin elevation, lymphopenia < 500/mm 3 • 12 patients with RBV dose reduction, but no transfusion, no epoetin needed CURRY Curry MP. Gastroenterology 2015; 148: 100 -107

CURRY Study: SOF + RBV for HCV with liver cancer before transplantation § Summary – In this pilot study, SOF + RBV before liver transplantation prevented recurrence of HCV infection in 70% of patients with chronic HCV infection and liver cancer who achieved an HCV RNA level < 25 IU/ml before transplantation and in almost half of the total patients in the study – The rate of discontinuation owing to adverse events was low, and most adverse events were those associated with RBV therapy— fatigue, anemia, headache, and nausea—as were the laboratory abnormalities of decreased hemoglobin and increased bilirubin – Enrichment in minor resistance-associated variants, although rare, may encode for marginal reductions in susceptibility to SOF – Limitations • Low sample size • Exclusion of patients with decompensated liver disease CURRY Curry MP. Gastroenterology 2015; 148: 100 -107
- Slides: 6