Current Statistical Issues in Dissolution Profile Comparisons Sutan















- Slides: 24

Current Statistical Issues in Dissolution Profile Comparisons Sutan Wu, Ph. D. FDA/CDER 5/20/2014 1

Outlines: • Background of Dissolution Profile Comparisons • Current Methods for Dissolution Profile Comparisons • Current Statistical Concerns • Simulation Cases • Discussions 2

Disclaimer: The presented work and views in this talk represents the presenter’s personal work and views, and do not reflect any views or policy with CDER/FDA. 3

Backgrounds: Dissolution profile comparison: why so important? ü Extensive applications throughout the product development process ü Comparison between batches of pre-change and post-change under certain post-change conditions e. g. : add a lower strength, formulation change, manufacturing site change ü Generic Drug Evaluations ü FDA Guidance: Dissolution, SUPAC-SS, SUPAC-IR, IVIV and etc. 4

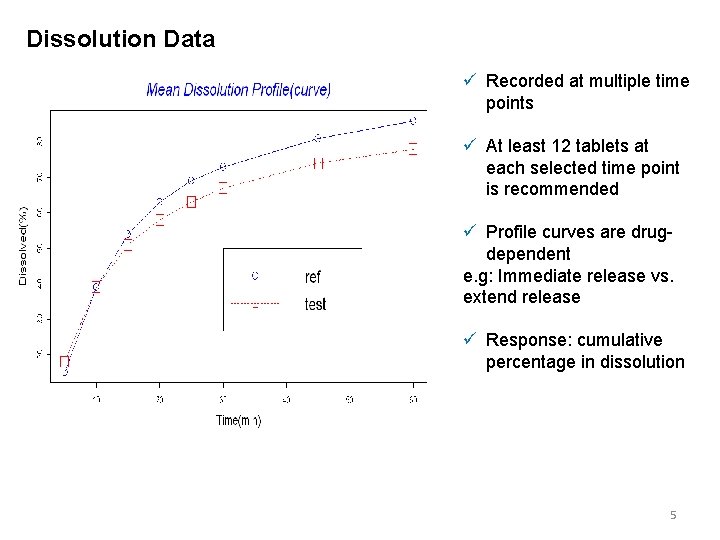
Dissolution Data ü Recorded at multiple time points ü At least 12 tablets at each selected time point is recommended ü Profile curves are drugdependent e. g: Immediate release vs. extend release ü Response: cumulative percentage in dissolution 5

Current Methods for Dissolution Profile Comparisons 6

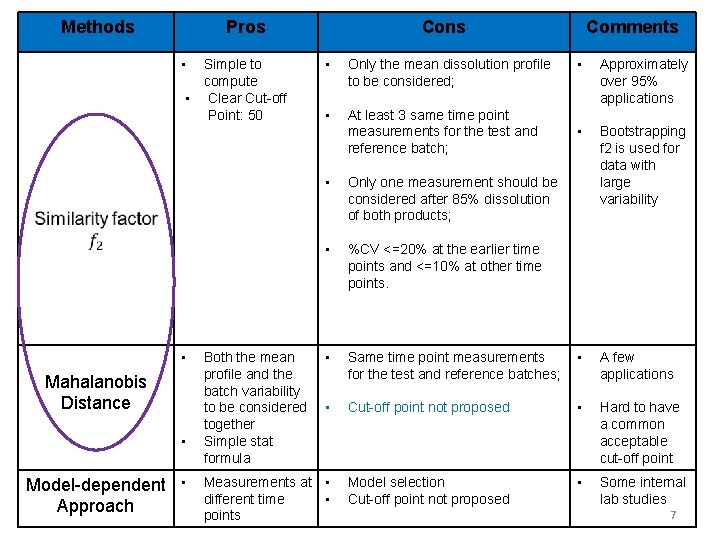
Methods Pros • Simple to compute • Clear Cut-off Point: 50 • Mahalanobis Distance • Model-dependent • Approach Cons • Only the mean dissolution profile to be considered; • At least 3 same time point measurements for the test and reference batch; Comments • Approximately over 95% applications • Bootstrapping f 2 is used for data with large variability • Only one measurement should be considered after 85% dissolution of both products; • %CV <=20% at the earlier time points and <=10% at other time points. • Same time point measurements for the test and reference batches; • A few applications • Cut-off point not proposed • Hard to have a common acceptable cut-off point Measurements at • different time • points Model selection Cut-off point not proposed • Some internal lab studies Both the mean profile and the batch variability to be considered together Simple stat formula 7

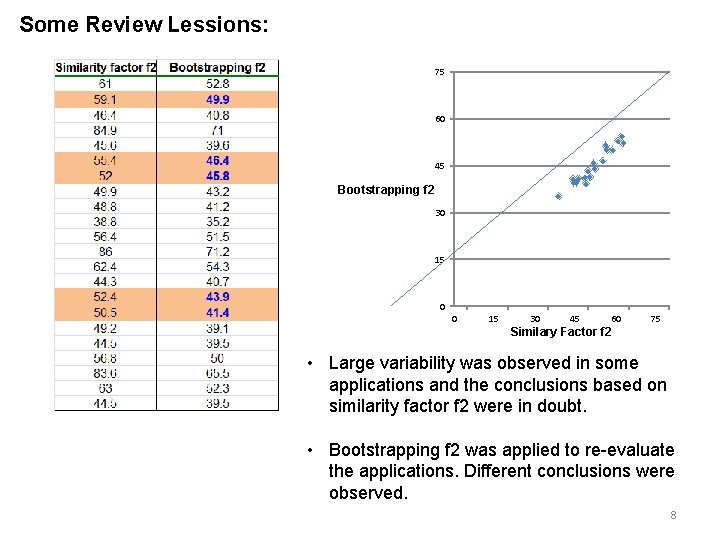
Some Review Lessions: 75 60 45 Bootstrapping f 2 30 15 0 0 15 30 45 60 75 Similary Factor f 2 • Large variability was observed in some applications and the conclusions based on similarity factor f 2 were in doubt. • Bootstrapping f 2 was applied to re-evaluate the applications. Different conclusions were observed. 8

Motivations: How to cooperate the variability consideration into dissolution profile comparison in a feasible and practical way? ü Bootstrapping f 2: § Lower bound of the non-parametric bootstrapping confidence interval (90%) for f 2 index § 50 could be the cut-off point § Subsequent Concerns: The validity of bootstrapping f 2? ü Mahalanobis-Distance (M-Distance): § A classical multivariate analysis tool for describing the distance between two vectors and widely used for outlier detection § Upper Bound of the 90% 2 -sided confidence interval (Tsong et. al. 1996) § Subsequent Concerns: The validity of M-Distance? The cut-off point? 9

Objectives: ü Thoroughly examine the performance of bootstrapping f 2 and f 2 index: can bootstrapping f 2 save the situations that f 2 is not applicable? ü Gain empirical knowledge of the values of M-distance: does Mdistance is a good substitute? What would be the “appropriate” cut-off point(s)? 10

Simulation Cases: q Scenarios 1: similarity factor f 2 “safe” cases For both batches 1) %CV at earlier time points (within 15 mins) <= 20% and %CV <= 10% at other time points; 2) Only one measurement after 85% dissolution q Scenarios 2: large batch variability cases (f 2 is not recommended generally) %CV > 20% (<= 15 mins) or/and %CV > 10% (> 15 mins) Ø Different mean dissolution profile but same variability for both batches Ø Same mean dissolution profile but testing batch has large variability q Scenarios 3: multiple measurements after 85% dissolution Ø “Safe” Variability cases: Dissolution Guidance recommendations Ø Large Variability cases 11

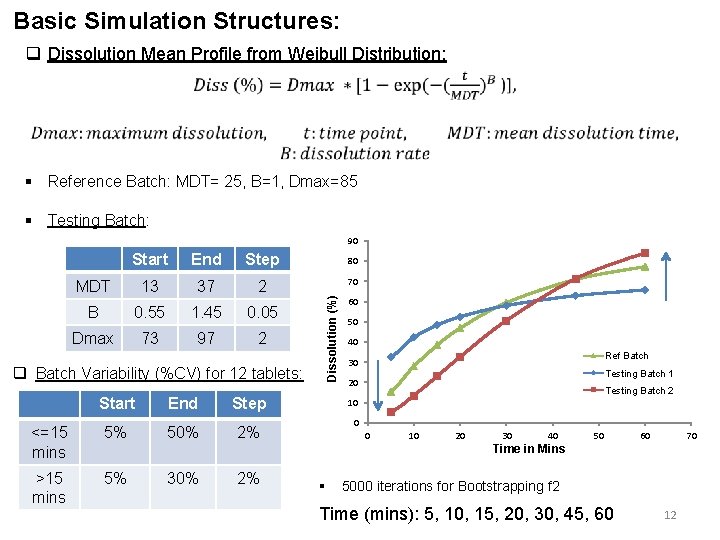
Basic Simulation Structures: q Dissolution Mean Profile from Weibull Distribution: § Reference Batch: MDT= 25, B=1, Dmax=85 § Testing Batch: 90 End Step 80 MDT 13 37 2 70 B 0. 55 1. 45 0. 05 Dmax 73 97 2 q Batch Variability (%CV) for 12 tablets: Start End Step <=15 mins 5% 50% 2% >15 mins 5% Dissolution (%) Start 60 50 40 Ref Batch 30 Testing Batch 1 20 Testing Batch 2 10 0 0 10 20 30 40 50 60 70 Time in Mins 30% 2% § 5000 iterations for Bootstrapping f 2 Time (mins): 5, 10, 15, 20, 30, 45, 60 12

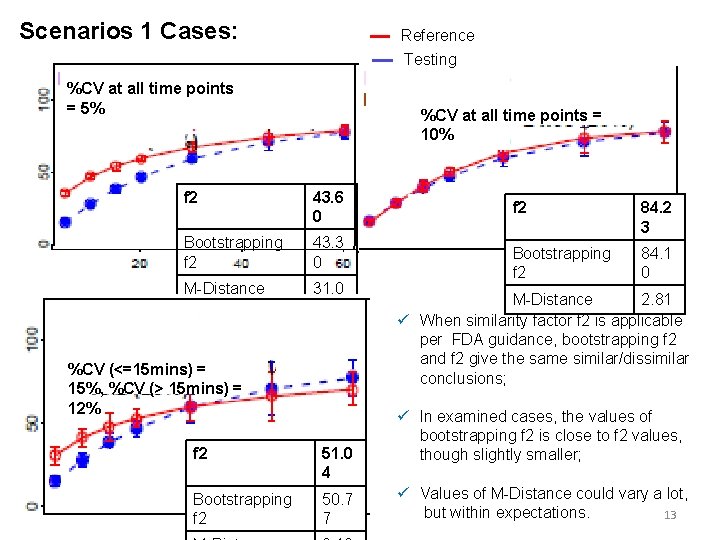
Scenarios 1 Cases: Reference Testing %CV at all time points = 5% %CV at all time points = 10% f 2 43. 6 0 Bootstrapping f 2 43. 3 0 M-Distance 31. 0 7 %CV (<=15 mins) = 15%, %CV (> 15 mins) = 12% f 2 51. 0 4 Bootstrapping f 2 50. 7 7 f 2 84. 2 3 Bootstrapping f 2 84. 1 0 M-Distance 2. 81 ü When similarity factor f 2 is applicable per FDA guidance, bootstrapping f 2 and f 2 give the same similar/dissimilar conclusions; ü In examined cases, the values of bootstrapping f 2 is close to f 2 values, though slightly smaller; ü Values of M-Distance could vary a lot, but within expectations. 13

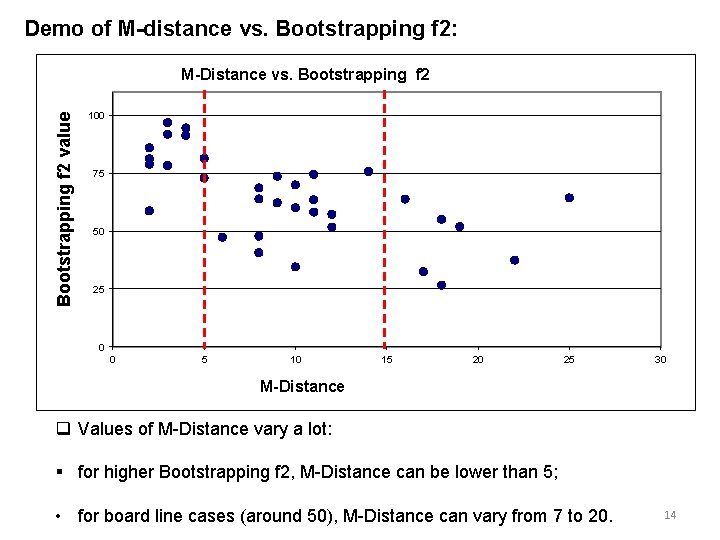
Demo of M-distance vs. Bootstrapping f 2: Bootstrapping f 2 value M-Distance vs. Bootstrapping f 2 100 75 50 25 0 0 5 10 15 20 25 30 M-Distance q Values of M-Distance vary a lot: § for higher Bootstrapping f 2, M-Distance can be lower than 5; • for board line cases (around 50), M-Distance can vary from 7 to 20. 14

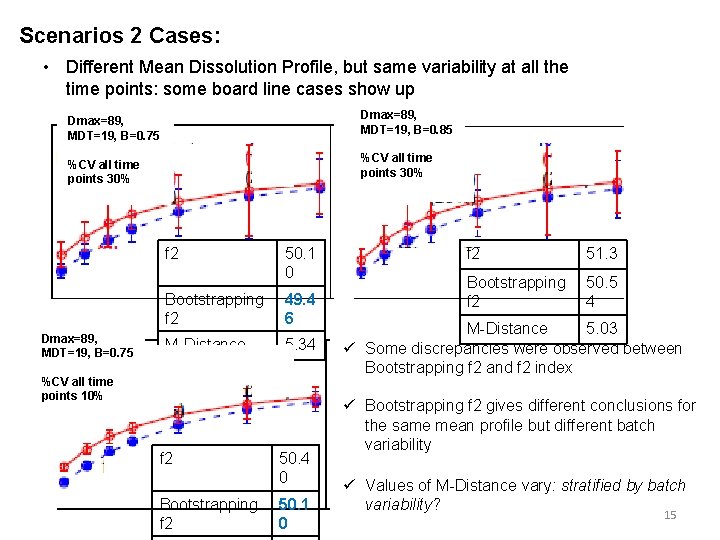
Scenarios 2 Cases: • Different Mean Dissolution Profile, but same variability at all the time points: some board line cases show up Dmax=89, MDT=19, B=0. 85 Dmax=89, MDT=19, B=0. 75 %CV all time points 30% f 2 Dmax=89, MDT=19, B=0. 75 50. 1 0 Bootstrapping f 2 49. 4 6 M-Distance 5. 34 %CV all time points 10% f 2 Bootstrapping f 2 50. 4 0 50. 1 0 f 2 51. 3 Bootstrapping f 2 50. 5 4 M-Distance 5. 03 ü Some discrepancies were observed between Bootstrapping f 2 and f 2 index ü Bootstrapping f 2 gives different conclusions for the same mean profile but different batch variability ü Values of M-Distance vary: stratified by batch variability? 15

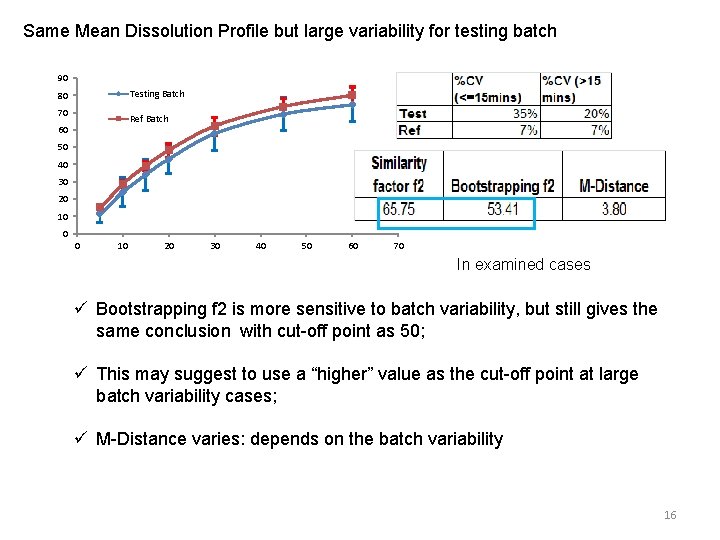
Same Mean Dissolution Profile but large variability for testing batch 90 Testing Batch 80 70 Ref Batch 60 50 40 30 20 10 0 0 10 20 30 40 50 60 70 In examined cases ü Bootstrapping f 2 is more sensitive to batch variability, but still gives the same conclusion with cut-off point as 50; ü This may suggest to use a “higher” value as the cut-off point at large batch variability cases; ü M-Distance varies: depends on the batch variability 16

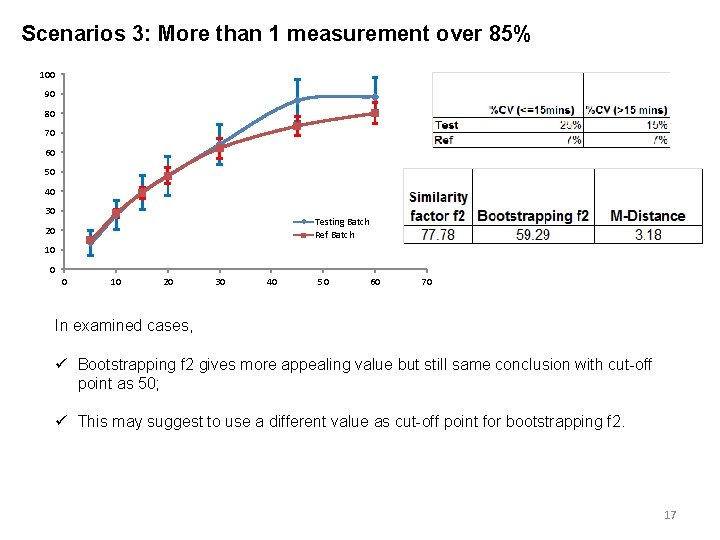
Scenarios 3: More than 1 measurement over 85% 100 90 80 70 60 50 40 30 Testing Batch Ref Batch 20 10 0 0 10 20 30 40 50 60 70 In examined cases, ü Bootstrapping f 2 gives more appealing value but still same conclusion with cut-off point as 50; ü This may suggest to use a different value as cut-off point for bootstrapping f 2. 17

Findings: ü When similarity factor f 2 is applicable per FDA Dissolution guidance, bootstrapping f 2 and f 2 give the same similar/dissimilar conclusions; In the examined cases, ü Bootstrapping f 2 is more sensitive to batch variability or multiple >85% measurements; However, with 50 as the cut-off points, bootstrapping f 2 still gives the same conclusion as similarity factor f 2; ü Values of M-Distance varies a lot and appears that <=3 could be a similar case, and over 30 could be a different case. Conclusions: ü Based on current review experiences and examined cases, bootstrapping f 2 is recommended when the similarity factor f 2 is around 50 or large batch variability is observed; ü At the large batch variability cases, new cut-off points may be proposed. Testing batches would be penalized by larger batch variability. ü M-Distance is another alternative approach for dissolution profile comparisons. Its values also depends on the batch variability. The cut-off point is required for further deep examinations, particularly, M-Distance values at different batch variability and bootstrapping f 2 around 50. 18

Proposal: To compute the increment M-Distance The proposed increment M-Distance can help us solve the convergence problem caused by highly correlated data (cumulative measurements); The interpretation of increment M-Distance: the distance between the increment vectors from the testing and reference batches. 19

References: • FDA Guidance: Dissolution Testing of Immediate Release Solid Oral Dosage Forms, 1997 • FDA Guidance: SUPAC for Immediate Release Solid Oral Dosage Forms, 1995 • FDA Guidance: Extended Release Oral Dosage Forms: Development, Evaluation, and Application of In Vitro/In Vivo Correlation, 1997 • In Vitro Dissolution Profile Comparison, Tsong et. al, 2003 • Assessment of Similarity Between Dissolution Profiles, Ma et. al, 2000 • In Vitro Dissolution Profile Comparison – Statistics and Analysis of the Similarity Factor f 2, V. Shah et. al, 1998 • Statistical Assessment of Mean Differences Between Dissolution Data Sets, Tsong et al, 1996 20

Acknowledgement: FDA Collaborators and Co-workers: • ONDQA: Dr. John Duan, Dr. Tien-Mien Chen • OGD: Dr. Pradeep M. Sathe • OB: Dr. Yi Tsong 21

THANK YOU! 22

Back Up 23

90% Confidence Region of M-Distance: , where By Langrage Multiplier Method 24