Current Results of Drug Coated Balloons William A

Current Results of Drug Coated Balloons William A. Gray MD Director of Endovascular Services Associate Professor of Clinical Medicine Columbia University Medical Center The Cardiovascular Research Foundation

William A. Gray, MD I have no real or apparent conflicts of interest to report.

The paradox and challenge of the ideal DCB Simply stated: To fully maintain an antiproliferative agent on a balloon until it is positioned at the lesion and then have all of the intended (metered? ) dose leave the balloon and reside completely within the targeted tissue with little if any systemic loss

Transfer and maintenance of drug in vessel wall: an order of magnitude POBA Day 7 • Following 60 -sec dilatation approximately 10%-15% of the drug is in the vessel wall 40– 60 min later. Drug-balloon Day 7 • Microtubule 24 hours later ~10% of drug delivered still resides in staining confirms vessel wall (1/100 ofof original dose) sustained presence paclitaxel at 1 week JACC Vol. 35, No. 7, June 2000: 1969– 76 J Am Coll Cardiol 2003; 42: 1415– 1420.
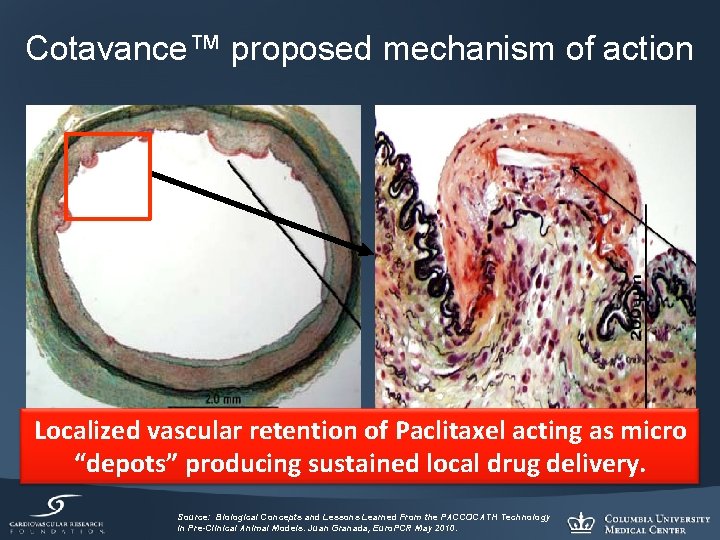
Cotavance™ proposed mechanism of action Localized vascular retention of Paclitaxel acting as micro “depots” producing sustained local drug delivery. Source: Biological Concepts and Lessons Learned From the PACCOCATH Technology in Pre-Clinical Animal Models. Juan Granada, Euro. PCR May 2010.
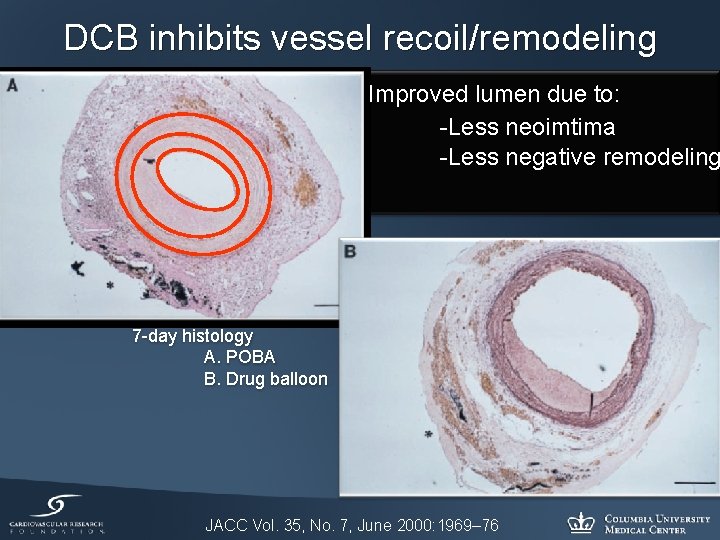
DCB inhibits vessel recoil/remodeling Improved lumen due to: -Less neoimtima -Less negative remodeling 7 -day histology A. POBA B. Drug balloon JACC Vol. 35, No. 7, June 2000: 1969– 76

Elements of drug-coated balloon systems • Balloon ¡ • Anti-proliferative Standard • Wrapped configuration ¡ Paclitaxil ¡ ‘limus family ¡ Others? ¡ Surface modified • Excipient § iopromide § urea § polymers § nanoparticles

Choice of antiproliferative agent for DCB • Ideal properties include: ¡ High lipophilicity resulting in rapid/high absorption ¡ Long-term antiproliferative effects ¡ No local or systemic toxicity ¡ Easily applied to balloon surface • Uniform distribution • Minimal loss with packing, sterilization, shipping, and handling prior to use • Minimal loss on transition through catheter/body and on initial inflation • While most of the data for DEB currently exists with paclitaxel, the ‘limus family of agents possess many of these qualities and may also be suitable
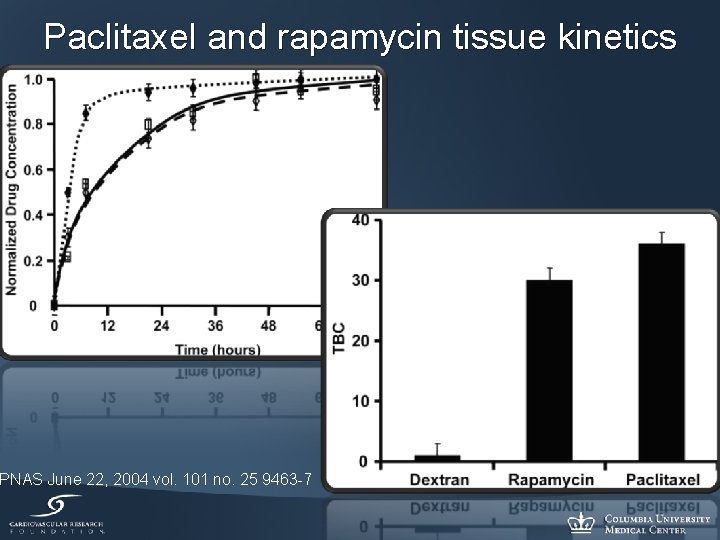
Paclitaxel and rapamycin tissue kinetics PNAS June 22, 2004 vol. 101 no. 25 9463 -7

Current DCB paclitaxel dosing DCBs Balloon Surface Dosing Product Paclitaxel (μg/mm 2) Dose on Balloon Surface Lutonix 2 Paccocath® (Medrad Interventional) 3 IN. PACT ™ (Invatec/Medtronic) 3 Dior® -Gen II (Euro. Cor) 3

First-generation DCB vary by formulation of paclitaxil and excipient • Excipient affects: ¡ Total drug load possible/required on balloon for efficacy ¡ Peak drug dose and drug half-life in arterial wall ¡ Downstream drug loss during balloon transit and inflation ¡ Coating durability and dose uniformity • Paclitaxel is generally preferred in a crystalline form
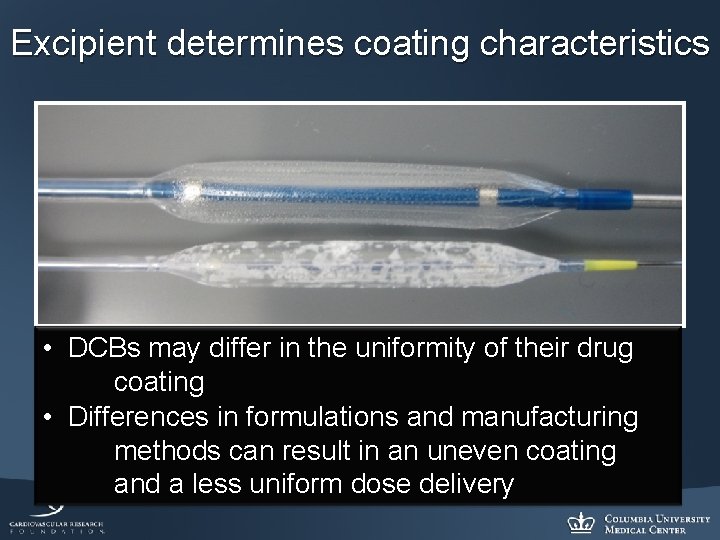
Excipient determines coating characteristics • DCBs may differ in the uniformity of their drug coating • Differences in formulations and manufacturing methods can result in an uneven coating and a less uniform dose delivery
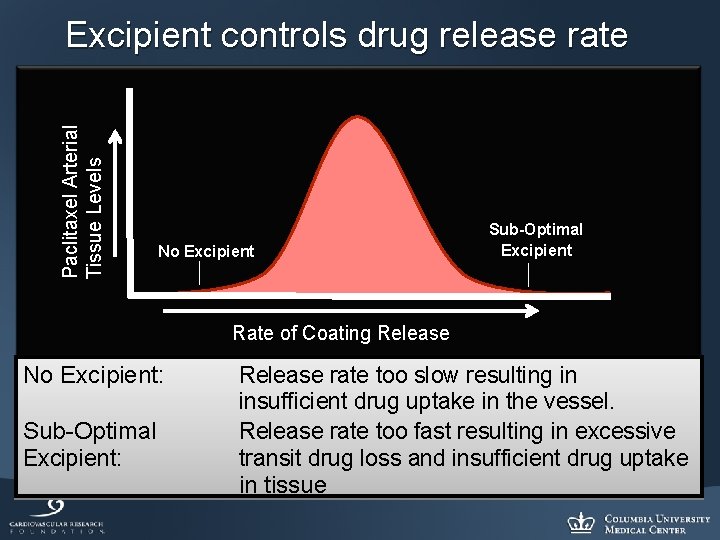
Paclitaxel Arterial Tissue Levels Excipient controls drug release rate No Excipient Sub-Optimal Excipient Rate of Coating Release No Excipient: Sub-Optimal Excipient: Release rate too slow resulting in insufficient drug uptake in the vessel. Release rate too fast resulting in excessive transit drug loss and insufficient drug uptake in tissue
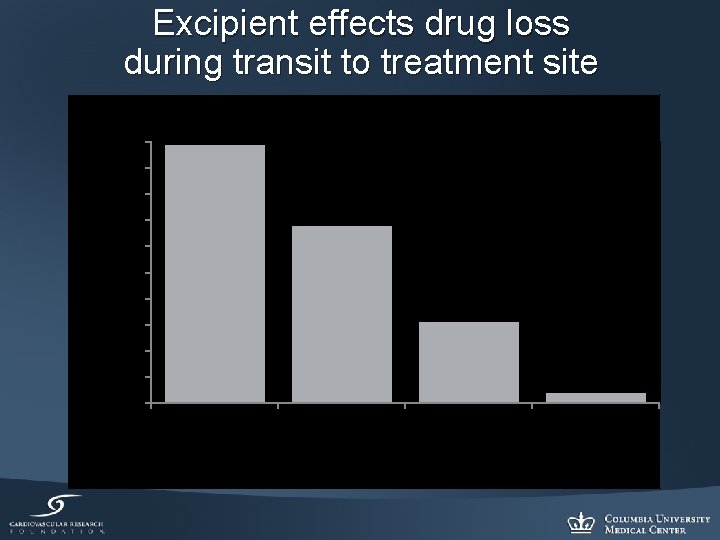
Percent of Paclitaxel Load Lost (%) Excipient effects drug loss during transit to treatment site 100 90 80 70 60 50 40 30 20 10 0 Additive A Additive B Additive C Excipient Formulations NO Additive
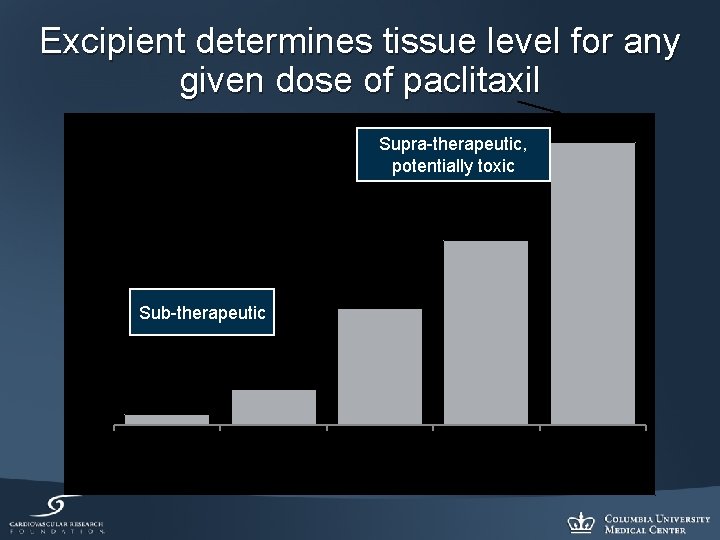
Peak Paclitaxel Level in Treated Tissue Segment Excipient determines tissue level for any given dose of paclitaxil Supra-therapeutic, potentially toxic Sub-therapeutic NO Additive E Additive F Additive G Different Excipients Additive H
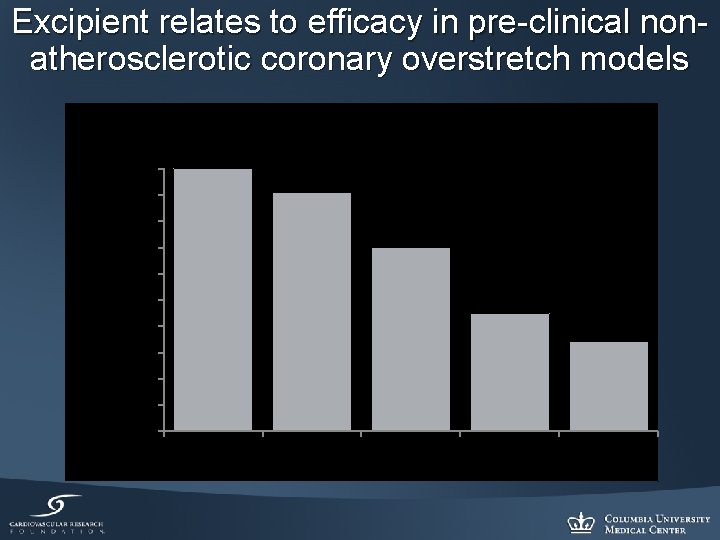
Excipient relates to efficacy in pre-clinical nonatherosclerotic coronary overstretch models Restenosis at 4 Weeks 100% Percent of Control Late Lumen Loss (%) 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% POBA NO Additive J Additive K Additive L

Sample of testing required for DCB’s In Vitro Studies • Coating performance • Formulation stability • Manufacturability In Vivo Studies • • • Peak drug levels in tissue Drug half-life in tissue Transit/Inflation time: dose impact Particulate analysis Serum, myocardial and organ drug levels • Histopathology 41 10 634 Pre-Clinical Studies GLP Studies Animals Potential Excipients > 225 analyzed Down-Select > 250 Formulations Testing > 120 Coatings > 3000 Devices Final Optimized Formulation Courtesy of Lutonix, Inc. Investigational - CE Mark Pending

The following involved with PAD clinical trials using DCB technology will be discussed • Medrad Interventional/Possis • Invatec/Medtronic • Lutonix • Euro. Cor • Cook Aachen Resonance Elutax Biotronik Passeo-18 Lux/Pheron Caliber Theraputics TADD
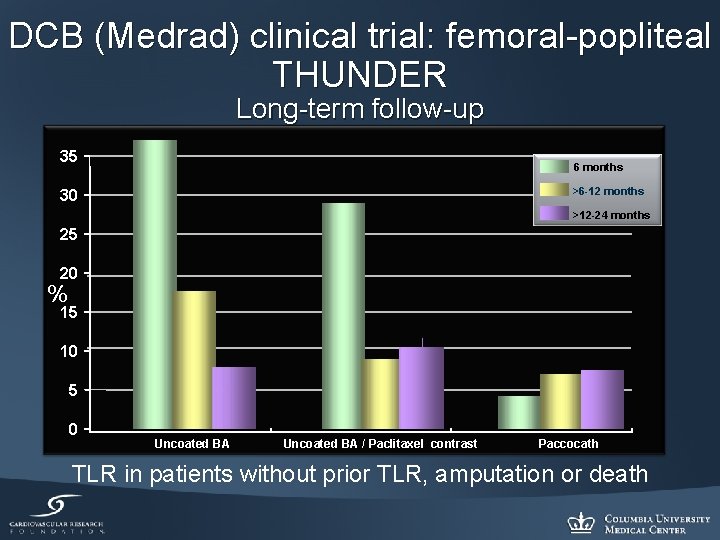
DCB (Medrad) clinical trial: femoral-popliteal THUNDER Long-term follow-up 35 6 months >6 -12 months 30 >12 -24 months 25 20 % 15 10 5 0 Uncoated BA / Paclitaxel contrast Paccocath TLR in patients without prior TLR, amputation or death
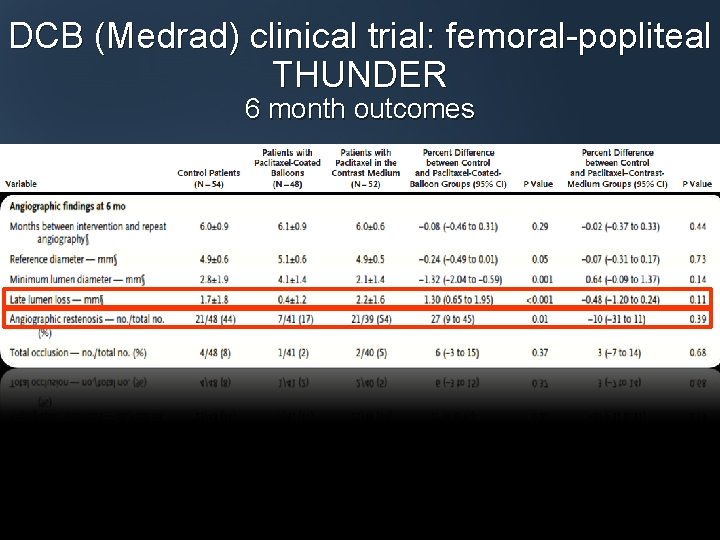
DCB (Medrad) clinical trial: femoral-popliteal THUNDER 6 month outcomes
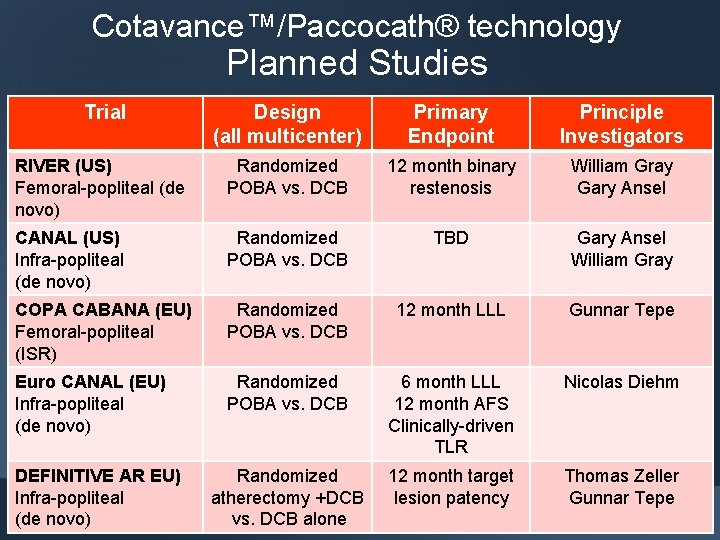
Cotavance™/Paccocath® technology Planned Studies Trial Design (all multicenter) Primary Endpoint Principle Investigators RIVER (US) Femoral-popliteal (de novo) Randomized POBA vs. DCB 12 month binary restenosis William Gray Gary Ansel CANAL (US) Infra-popliteal (de novo) Randomized POBA vs. DCB TBD Gary Ansel William Gray COPA CABANA (EU) Femoral-popliteal (ISR) Randomized POBA vs. DCB 12 month LLL Gunnar Tepe Euro CANAL (EU) Infra-popliteal (de novo) Randomized POBA vs. DCB 6 month LLL 12 month AFS Clinically-driven TLR Nicolas Diehm Randomized atherectomy +DCB vs. DCB alone 12 month target lesion patency Thomas Zeller Gunnar Tepe DEFINITIVE AR EU) Infra-popliteal (de novo)

DEFINITIVE AR study: Rationale for Plaque Excision Incorporation of drug delivery technology as an antirestenotic treatment while maintaining the unique biomechanical properties of the femoropopliteal tract. • Mechanically re-canalize artery without overstretch • Remove diffusion barrier better and more effective and homogenous drug uptake • Reduce likelihood of bail-out stenting and preserve native artery architecture

Paccocath and DA Pilot Study Histology Control: PTX Balloon + stent Test: Silver. Hawk + PTX Balloon In areas of directional atherectomy, minimal neointimal hyperplasia was noted. The healing response was similar to what has been observed in sirolimus and paclitaxel coated stents preclinical work Source: ev 3 internal data; Histology performed by CVPath.

Lutonix clinical trials • 3 studies fully enrolled: 6 month angiographic follow-up available late 2010 Completed Studies • US peripheral IDE beginning near-term LEVANT I SFA-Popliteal PERVIDEO I Coronary ISR 100 -patient PI: Dierk Scheinert prospective randomized study DCB vs. PTA 40 -patient prospective PI: Detlef Mathey and single arm Laura Mauri De Novo Trial 24 -patient prospective PI: Patrick Serruys Coronary single arm De Novo Investigational - CE Mark Pending
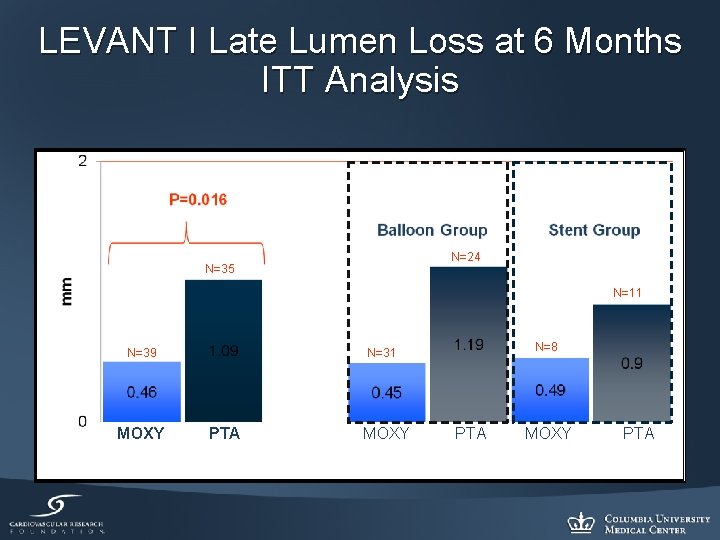
LEVANT I Late Lumen Loss at 6 Months ITT Analysis N=24 N=35 N=11 N=39 MOXY N=8 N=31 PTA MOXY PTA
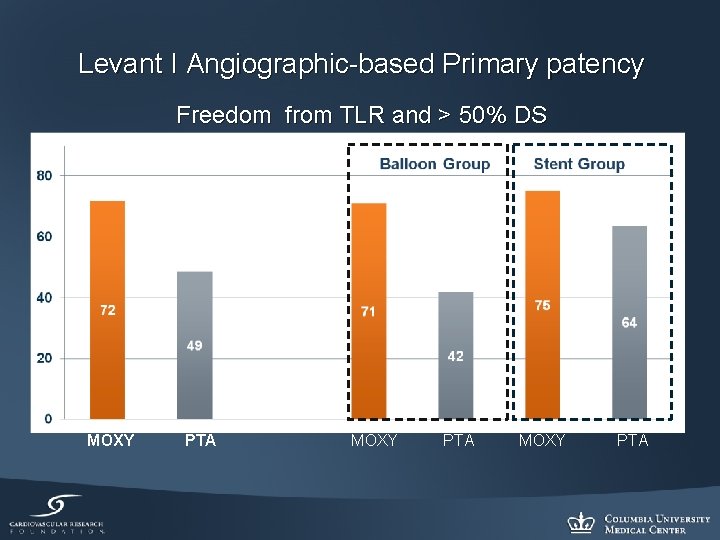
Levant I Angiographic-based Primary patency Freedom from TLR and > 50% DS MOXY PTA
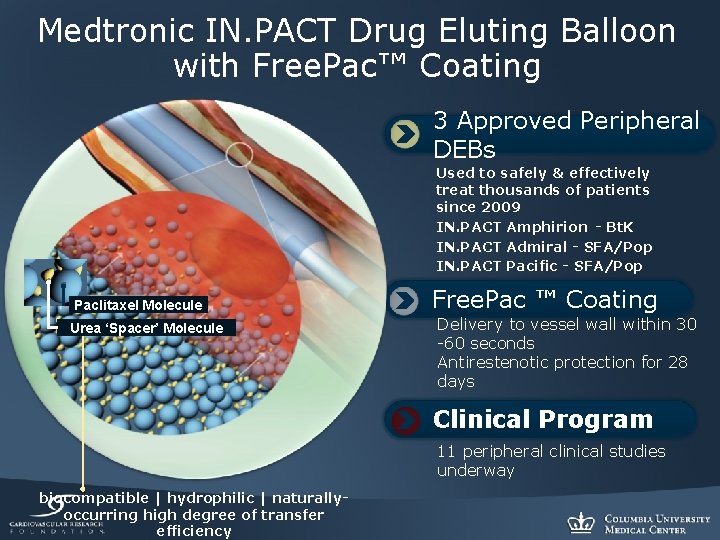
Medtronic IN. PACT Drug Eluting Balloon with Free. Pac™ Coating 3 Approved Peripheral DEBs Used to safely & effectively treat thousands of patients since 2009 IN. PACT Amphirion - Bt. K IN. PACT Admiral - SFA/Pop IN. PACT Pacific - SFA/Pop Paclitaxel Molecule Urea ‘Spacer’ Molecule Free. Pac ™ Coating Delivery to vessel wall within 30 -60 seconds Antirestenotic protection for 28 days Clinical Program 11 peripheral clinical studies underway biocompatible | hydrophilic | naturallyoccurring high degree of transfer efficiency
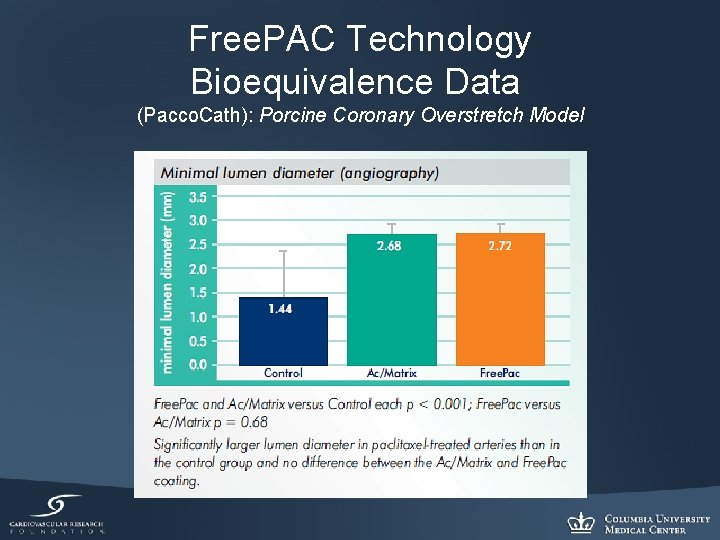
Free. PAC Technology Bioequivalence Data (Pacco. Cath): Porcine Coronary Overstretch Model
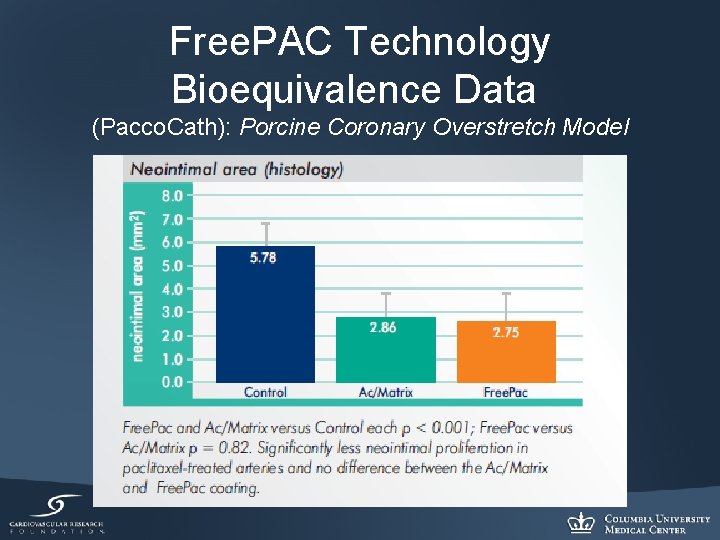
Free. PAC Technology Bioequivalence Data (Pacco. Cath): Porcine Coronary Overstretch Model
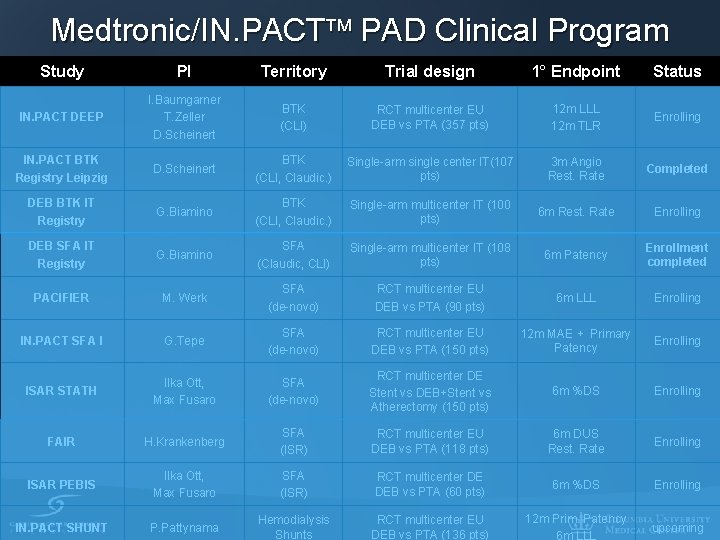
Medtronic/IN. PACT PAD Clinical Program Study PI Territory Trial design 1° Endpoint Status IN. PACT DEEP I. Baumgarner T. Zeller D. Scheinert BTK (CLI) RCT multicenter EU DEB vs PTA (357 pts) 12 m LLL 12 m TLR Enrolling IN. PACT BTK Registry Leipzig D. Scheinert BTK (CLI, Claudic. ) Single-arm single center IT(107 pts) 3 m Angio Rest. Rate Completed DEB BTK IT Registry G. Biamino BTK (CLI, Claudic. ) Single-arm multicenter IT (100 pts) 6 m Rest. Rate Enrolling DEB SFA IT Registry G. Biamino SFA (Claudic, CLI) Single-arm multicenter IT (108 pts) 6 m Patency Enrollment completed PACIFIER M. Werk SFA (de-novo) RCT multicenter EU DEB vs PTA (90 pts) 6 m LLL Enrolling IN. PACT SFA I G. Tepe SFA (de-novo) RCT multicenter EU DEB vs PTA (150 pts) 12 m MAE + Primary Patency Enrolling ISAR STATH Ilka Ott, Max Fusaro SFA (de-novo) RCT multicenter DE Stent vs DEB+Stent vs Atherectomy (150 pts) 6 m %DS Enrolling FAIR H. Krankenberg SFA (ISR) RCT multicenter EU DEB vs PTA (118 pts) 6 m DUS Rest. Rate Enrolling ISAR PEBIS Ilka Ott, Max Fusaro SFA (ISR) RCT multicenter DE DEB vs PTA (60 pts) 6 m %DS Enrolling IN. PACT SHUNT P. Pattynama Hemodialysis Shunts RCT multicenter EU DEB vs PTA (136 pts) 12 m Prim. Patency upcoming
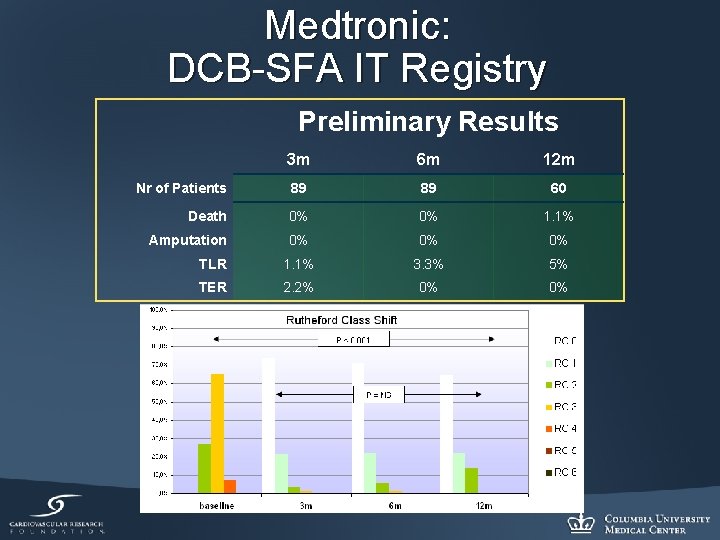
Medtronic: DCB-SFA IT Registry Preliminary Results 3 m 6 m 12 m Nr of Patients 89 89 60 Death 0% 0% 1. 1% Amputation 0% 0% 0% TLR 1. 1% 3. 3% 5% TER 2. 2% 0% 0%

N = 91 Werk M et al
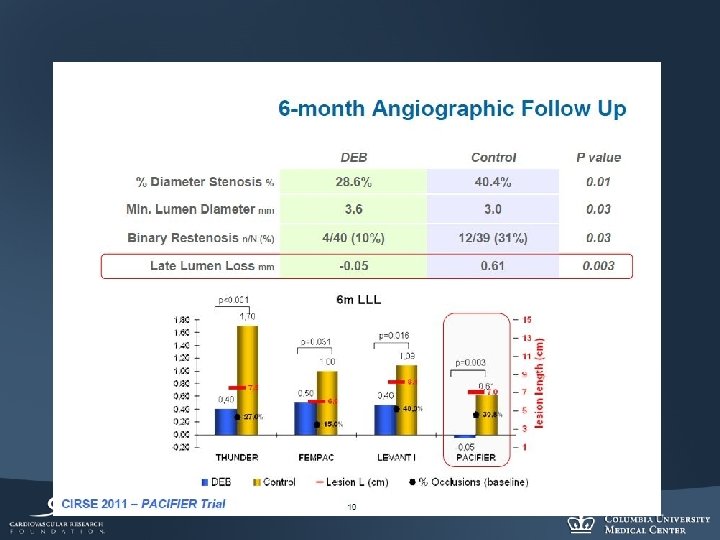
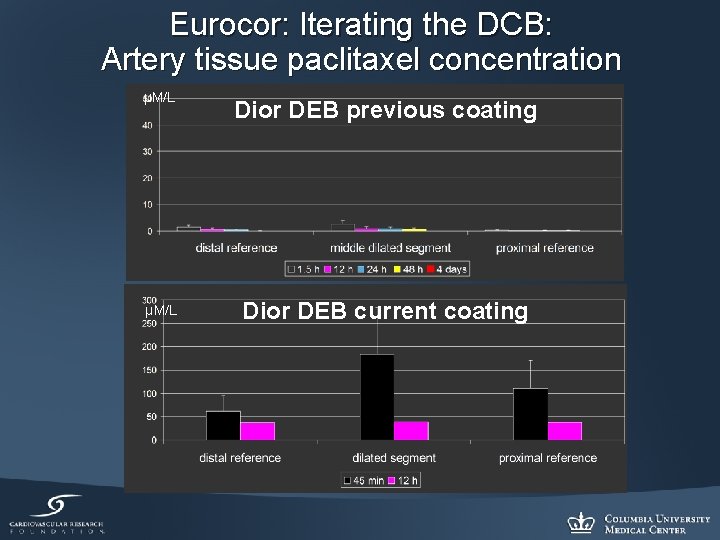
Eurocor: Iterating the DCB: Artery tissue paclitaxel concentration µM/L Dior DEB previous coating Dior DEB current coating

Eurocor PAD EU clinical trial plan Study Design Location Principle Investigator Freeway Stent Study Randomized POBA vs. DCB post-stent SFA-popliteal Professor Tacke Freeride Randomized POBA vs. DEB SFA-popliteal Professor Schulte PACUBA I Randomized POBA vs. DEB SFA-ISR Professor Lammer

Cook DCB: Advance 18 PTX Balloon Catheter • Nylon balloon/catheter with drug applied directly (no carrier, no polymer). Drug-coated PTA Balloon Study • Prospective, randomized trial in SFA/popliteal: ¡ Lesions up to 19 cm in length Enrolling up to 150 patients (Germany and Russia) • Primary endpoint ¡ Late lumen loss at 6 months ¡

Conclusion • A single-exposure of paclitaxel during balloon angioplasty can be demonstrated to result in both instantaneous and long term drug retention ¡ Via this mechanism, non-stent drug delivery has shown effectiveness in both pre-clinical and clinical settings in peripheral circulation ¡ Delivery mechanisms for antiproliferative agents appear to be important to the biologic efficacy of this therapy, and differ by device/excipient • Current and future clinical trials of DCB in PAD will test both proprietary formulations and adjunct therapies
Thank you
- Slides: 38