CTLA 4 Biology and Targeted Immunotherapy John Janik

CTLA 4 Biology and Targeted Immunotherapy John Janik, M. D. Co-Director Clinical Trials Team Metabolism Branch Center for Cancer Research, National Cancer Institute Bethesda, USA
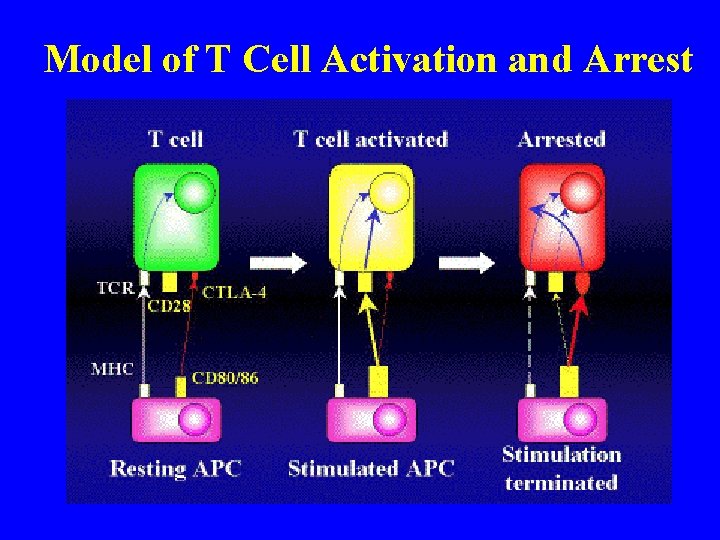
Model of T Cell Activation and Arrest

CD 28 and CTLA 4 Compete for the Same Receptors Expression Half-life B 7. 1 CD 28 Constitutive > 8 Hr 4 m. M CTLA 4 Induced 90 minutes 0. 4 m. M B 7. 2 15 -40 m. M 4 m. M

CTLA 4 Knockout Mouse • Signaling through CTLA 4 is absent • Mice die at 3 -4 weeks of age • Massive infiltration of tissues with lymphocytes • Lymphocytes infiltrate the liver, heart, lung and pancreas

CTLA-4 Blockade in Mouse Tumor Models • Regression of well-established transplantable tumors, and prolongs survival after tumor resection • Effective as single agent in strongly immunogenic tumors • Combination with other agents required for eradication of poorly immunogenic tumors

CTLA-4 Blockade in Mouse Tumor Models • Synergizes – Vaccines (transduced tumor cells, DC, DNA) – Conventional therapies (chemotherapy, local irradiation, surgical resection, hormonal therapy) • Independent of regulatory T cells, and can synergize with T reg depletion
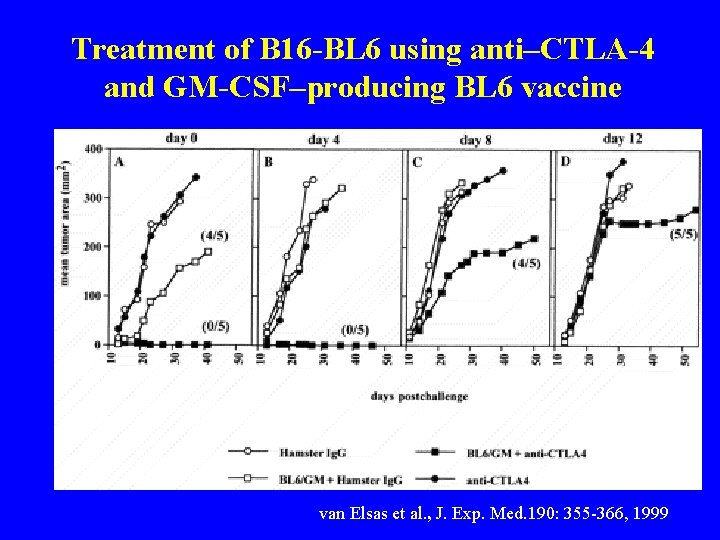
Treatment of B 16 -BL 6 using anti–CTLA-4 and GM-CSF–producing BL 6 vaccine van Elsas et al. , J. Exp. Med. 190: 355 -366, 1999
van Elsas et al. , J. Exp. Med. 190: 355 -366, 1999

CTLA 4 Antibodies in Clinical Development • Ticilimumab (CP-675, 206) Pfizer (Ig. G 2) • Ipilimumab (MDX-010) Medarex, BMS (Ig. G 1)
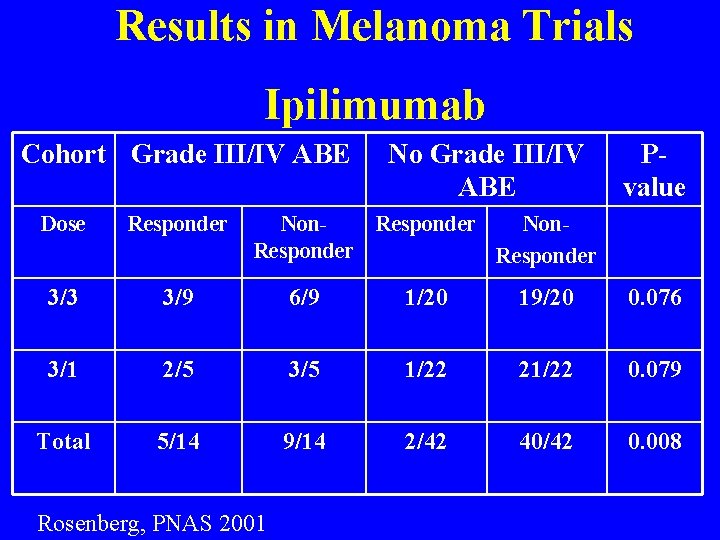
Results in Melanoma Trials Ipilimumab Cohort Grade III/IV ABE No Grade III/IV ABE Pvalue Dose Responder Non. Responder 3/3 3/9 6/9 1/20 19/20 0. 076 3/1 2/5 3/5 1/22 21/22 0. 079 Total 5/14 9/14 2/42 40/42 0. 008 Rosenberg, PNAS 2001
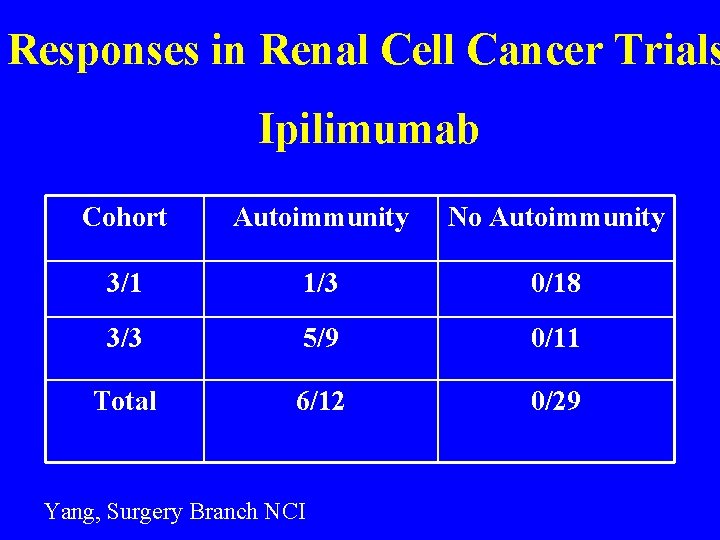
Responses in Renal Cell Cancer Trials Ipilimumab Cohort Autoimmunity No Autoimmunity 3/1 1/3 0/18 3/3 5/9 0/11 Total 6/12 0/29 Yang, Surgery Branch NCI

Autoimmune Breakthrough Events (ABE) • • • Enterocolitis Dermatitis Ocular inflammation Hypophysitis Adrenal insufficiency Hepatitis Pneumonitis Renal insufficiency Aseptic meningitis

Enterocolitis • Frequency 41 of 137 patients treated developed diarrhea • 4 patients had bowel perforation Beck, K. E. et al. J Clin Oncol; 24: 2283 -2289 2006

Enterocolitis • Lymphoma 0% • Melanoma frequency 14% • Renal Cell Carcinoma 28% Beck, K. E. et al. J Clin Oncol; 24: 2283 -2289 2006

Enterocolitis Histologic Characteristics – Neutrophilic inflammation only (46%) – Lymphocytic inflammation only (15%) – Combined neutrophilic and lymphocytic inflammation (38%) Beck, K. E. et al. J Clin Oncol; 24: 2283 -2289 2006
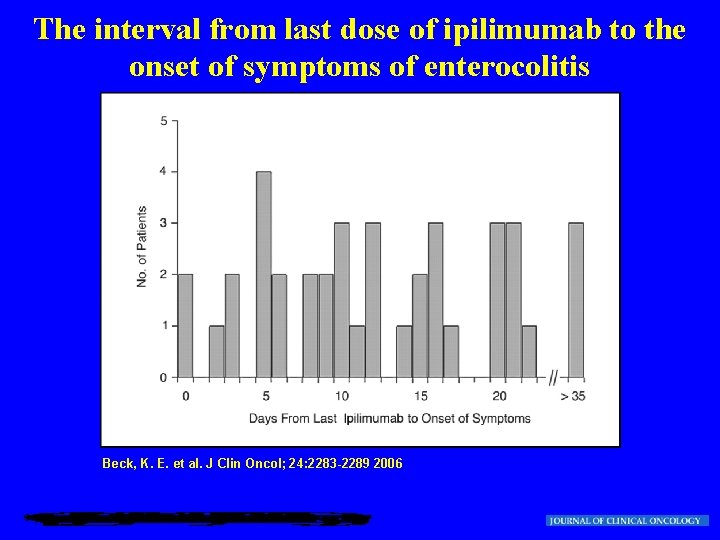
The interval from last dose of ipilimumab to the onset of symptoms of enterocolitis Beck, K. E. et al. J Clin Oncol; 24: 2283 -2289 2006
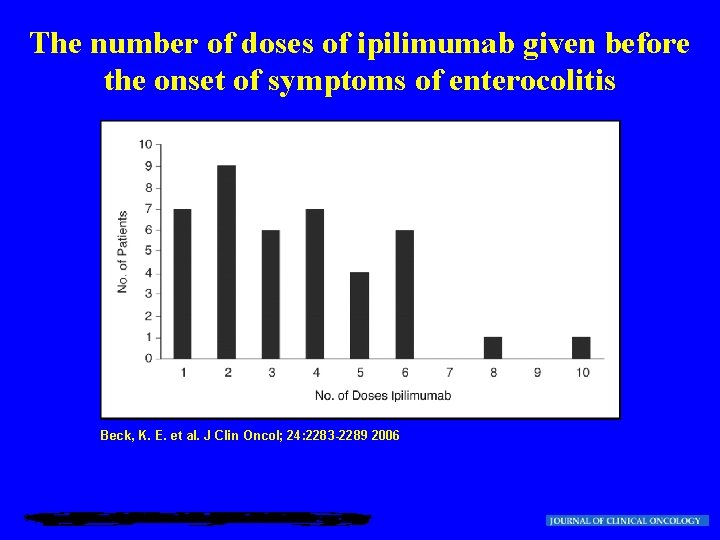
The number of doses of ipilimumab given before the onset of symptoms of enterocolitis Beck, K. E. et al. J Clin Oncol; 24: 2283 -2289 2006
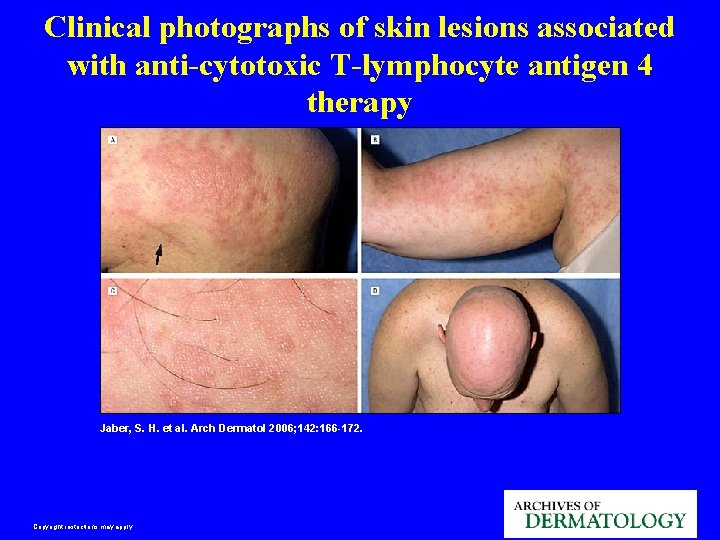
Clinical photographs of skin lesions associated with anti-cytotoxic T-lymphocyte antigen 4 therapy Jaber, S. H. et al. Arch Dermatol 2006; 142: 166 -172. Copyright restrictions may apply.
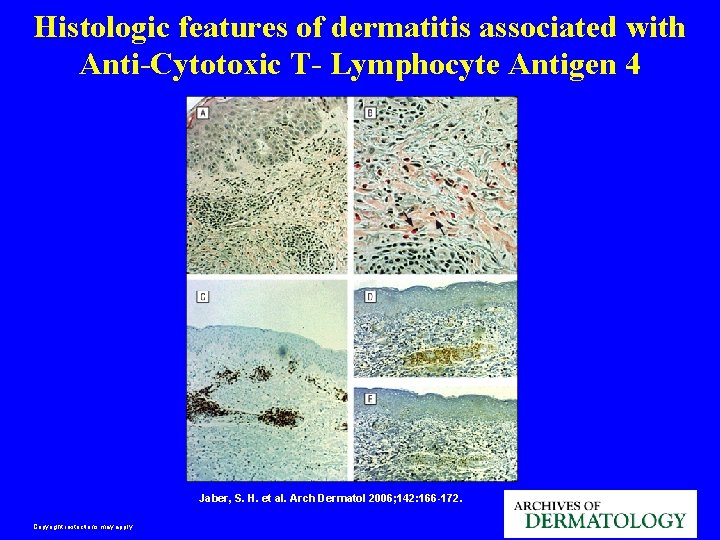
Histologic features of dermatitis associated with Anti-Cytotoxic T- Lymphocyte Antigen 4 Jaber, S. H. et al. Arch Dermatol 2006; 142: 166 -172. Copyright restrictions may apply.
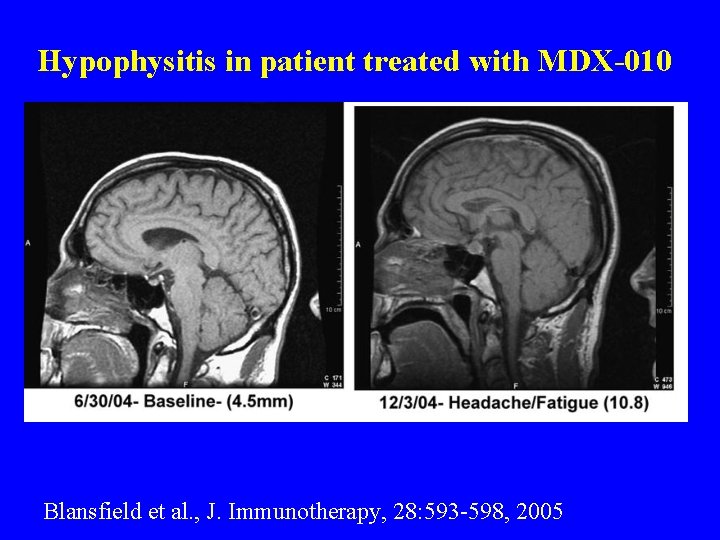
Hypophysitis in patient treated with MDX-010 Blansfield et al. , J. Immunotherapy, 28: 593 -598, 2005

Intrapatient Dose-escalation with Ipilimumab • Hypothesis – Higher doses may increase autoimmune events with higher response rate • Design – 3 mg/kg x 2 If no ABE • 5 mg/kg x 2 If no ABE • 9 mg/kg x 2 • 16/46 pts developed ABE, 5 responses • No correlation between autoimmune events and response Maker, J Immunotherapy 2006

A pilot study of Ipilimumab in patients with progressive cancer following cancer vaccine therapy John Janik, M. D. Co-Director Clinical Trials Team Metabolism Branch Center for Cancer Research, National Cancer Institute Bethesda, USA

Study objectives • Determine the toxicity of Ipilimumab • Determine effect of Ipilimumab on tumorspecific T cell responses • Monitor clinical tumor responses

Treatment schema Dose 3 mg/kg 1. 5 mg/kg Day 0 28 1. 5 mg/kg 56 1. 5 mg/kg 84
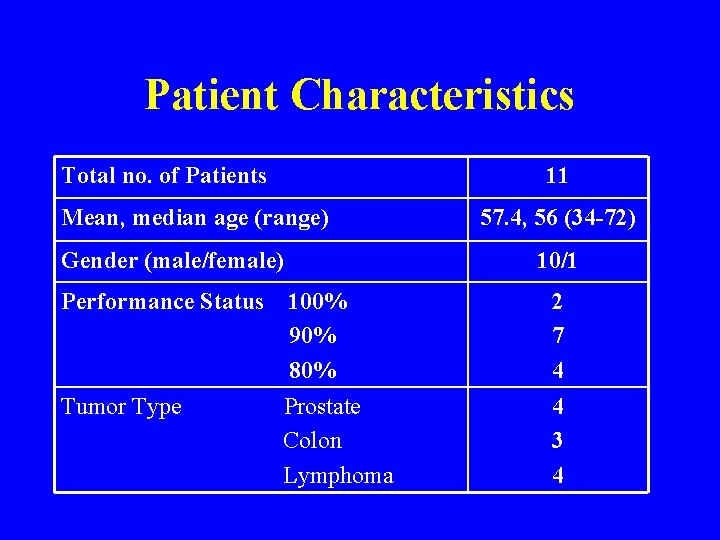
Patient Characteristics Total no. of Patients 11 Mean, median age (range) Gender (male/female) 57. 4, 56 (34 -72) 10/1 Performance Status 100% 90% 80% 2 7 4 Tumor Type Prostate Colon Lymphoma 4 3 4
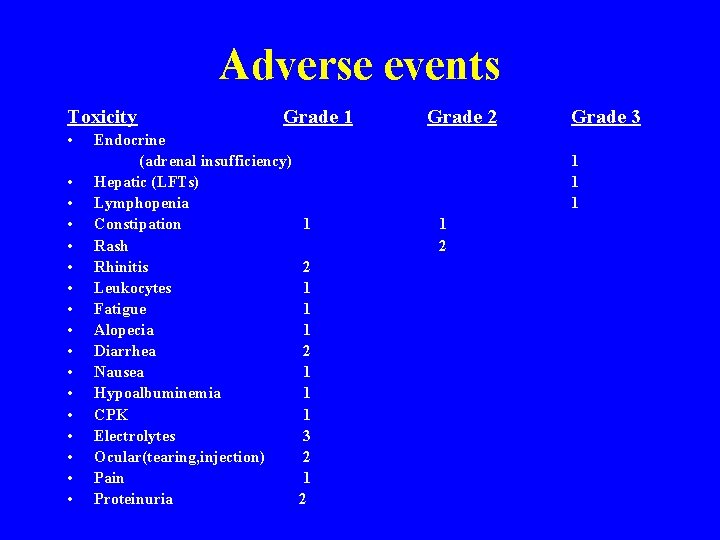
Adverse events Toxicity • • • • • Grade 1 Endocrine (adrenal insufficiency) Hepatic (LFTs) Lymphopenia Constipation Rash Rhinitis Leukocytes Fatigue Alopecia Diarrhea Nausea Hypoalbuminemia CPK Electrolytes Ocular(tearing, injection) Pain Proteinuria Grade 2 Grade 3 1 1 2 1 1 1 3 2 1 2
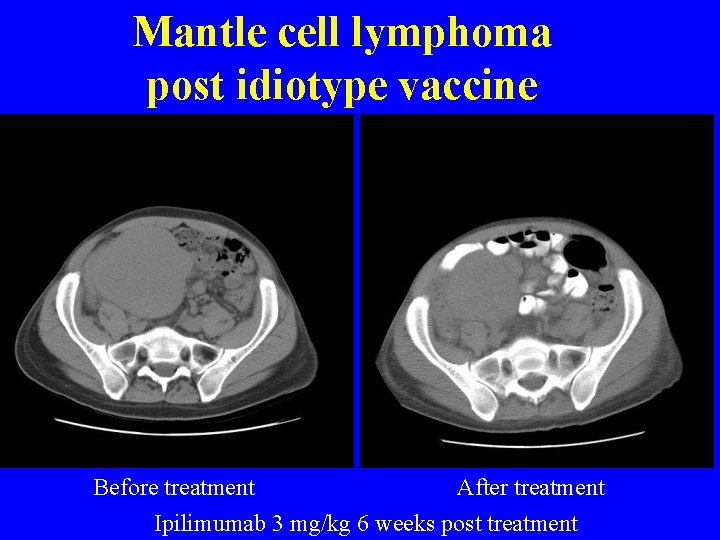
Mantle cell lymphoma post idiotype vaccine Before treatment After treatment Ipilimumab 3 mg/kg 6 weeks post treatment

Treatment schema Dose 3 mg/kg Day 0 3 mg/kg 21 3 mg/kg 42 3 mg/kg 63 3 mg/kg 84 3 mg/kg 105
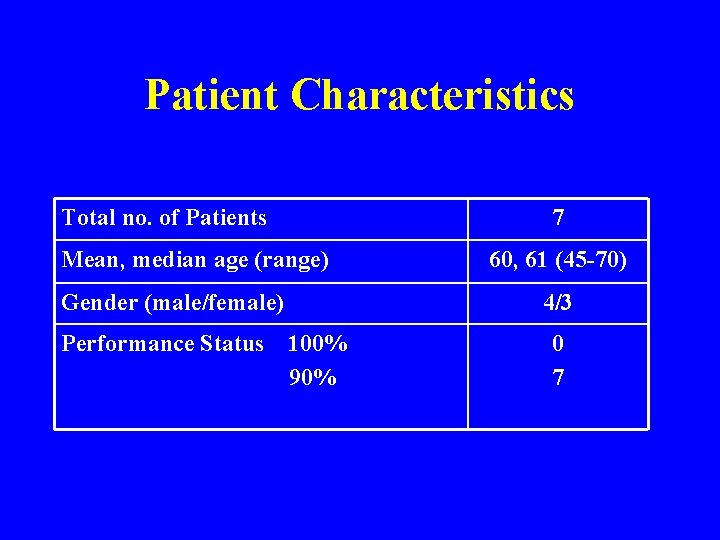
Patient Characteristics Total no. of Patients 7 Mean, median age (range) Gender (male/female) Performance Status 60, 61 (45 -70) 4/3 100% 90% 0 7
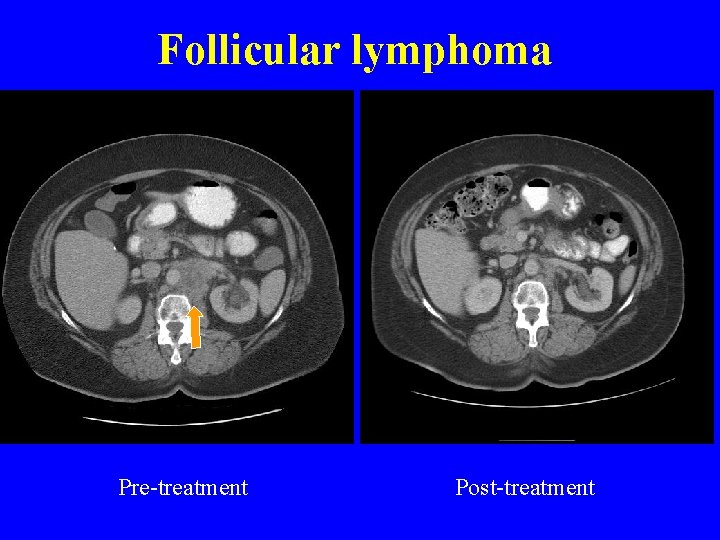
Follicular lymphoma Pre-treatment Post-treatment
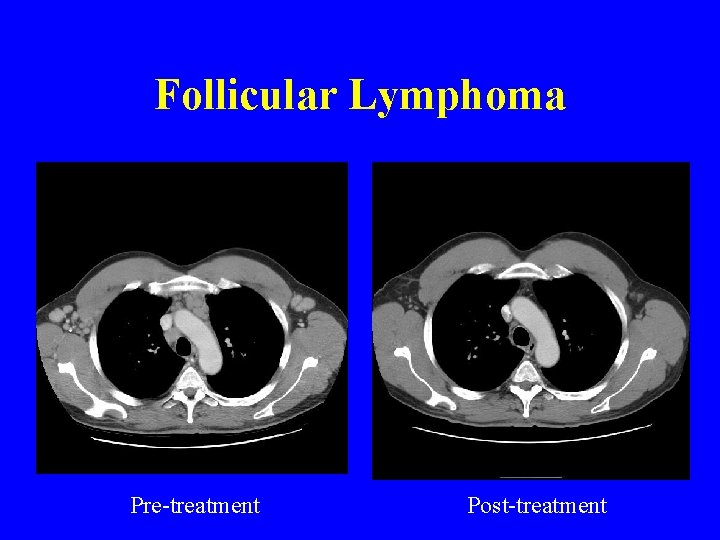
Follicular Lymphoma Pre-treatment Post-treatment
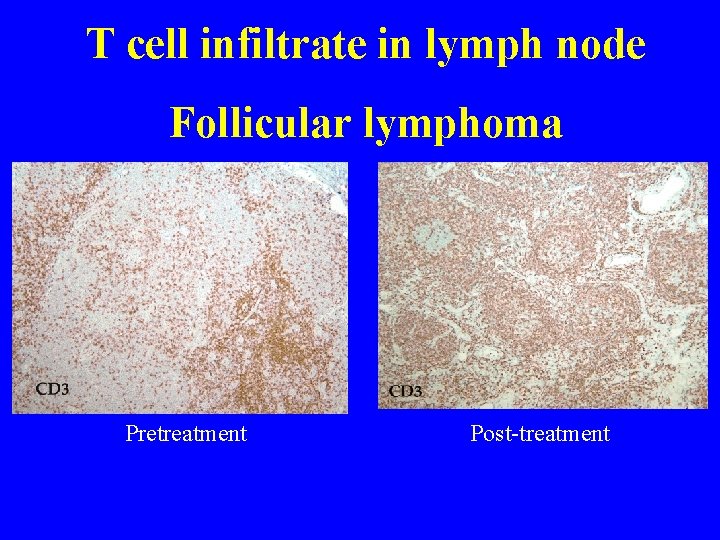
T cell infiltrate in lymph node Follicular lymphoma Pretreatment Post-treatment
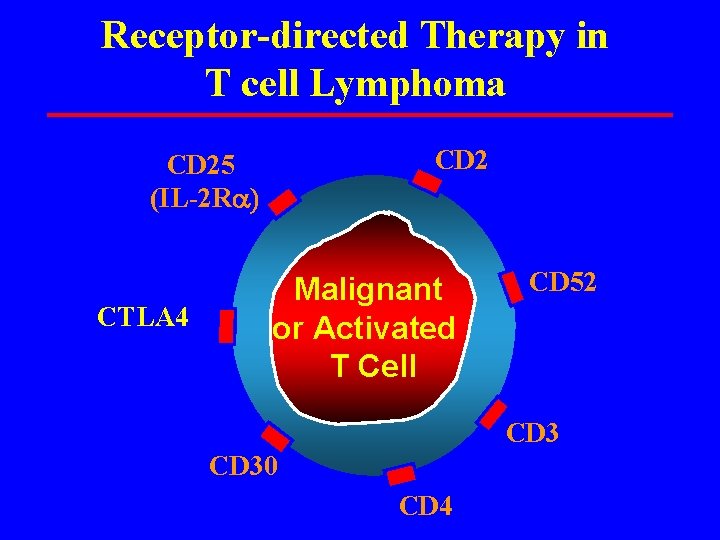
Receptor-directed Therapy in T cell Lymphoma CD 25 (IL-2 Ra CTLA 4 Malignant or Activated T Cell CD 52 CD 30 CD 4

Conclusions • Ipilimumab has activity in melanoma (14%/198 pts), renal cell carcinoma (15%/41 pts) and B cell lymphoma • Well characterized safety profile – Autoimmune breakthrough events are characteristic (20 -30%) • Higher doses of Ipilimumab produce greater frequency of autoimmunity but does not increase response rate • Autoimmunity can be managed successfully in the majority of patients with steroid therapy • Responses can be observed without autoimmunity

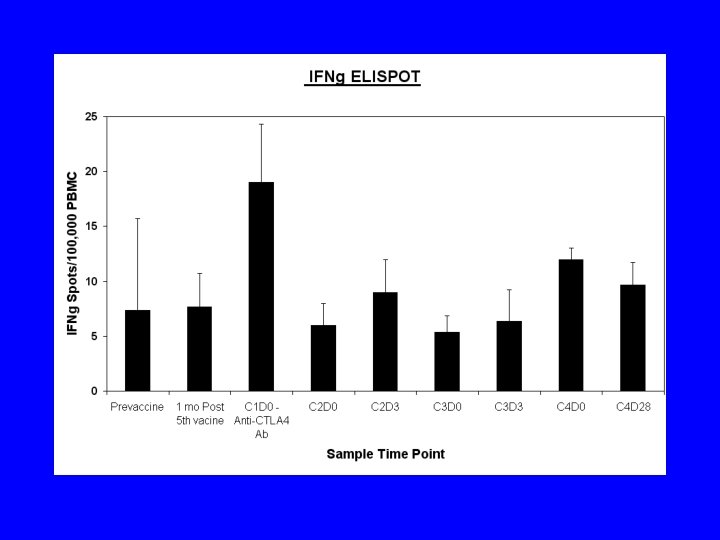
Conclusions • Ipilimumab was well tolerated in patients with lymphoma - one dose-limiting toxicity autoimmune toxicity • No consistent change in vaccine specific T cell responses • Three objective responses in 9 patients (33%) with follicular lymphoma (9 months, 7 months, 13+ months) • Retreatment can induce a second remission

Acknowledgements • Metabolism Branch – – – – John C. Morris Deirdre O’Mahony Wendy Gao T. A. Waldmann Wyndham H. Wilson Larry Kwak Barry Gause Sattva Neelapu • Flow cytometry – Tom Fleisher – Margaret Brown • Pathology – Stefania Pittaluga • Statistics – Paul Albert • Ophthamology – Robert Nussenblatt
- Slides: 37