CSALT Study grazoprevir elbasvir in genotype 1 with
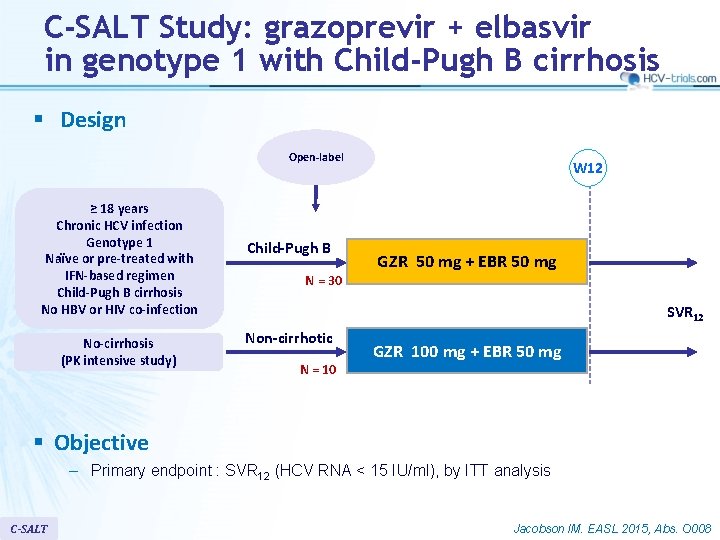
C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis § Design Open-label ≥ 18 years Chronic HCV infection Genotype 1 Naïve or pre-treated with IFN-based regimen Child-Pugh B cirrhosis No HBV or HIV co-infection No-cirrhosis (PK intensive study) Child-Pugh B N = 30 W 12 GZR 50 mg + EBR 50 mg SVR 12 Non-cirrhotic N = 10 GZR 100 mg + EBR 50 mg § Objective – Primary endpoint : SVR 12 (HCV RNA < 15 IU/ml), by ITT analysis C-SALT Jacobson IM. EASL 2015, Abs. O 008
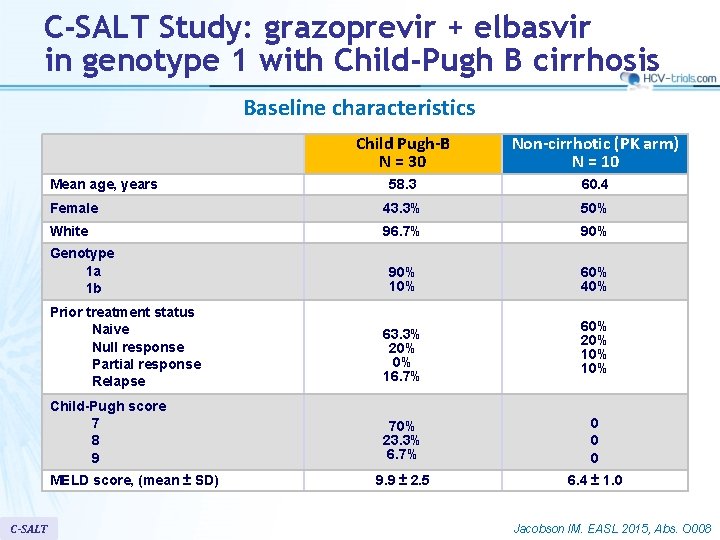
C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis Baseline characteristics Child Pugh-B N = 30 Non-cirrhotic (PK arm) N = 10 58. 3 60. 4 Female 43. 3% 50% White 96. 7% 90% 10% 60% 40% Mean age, years Genotype 1 a 1 b Prior treatment status Naive Null response Partial response Relapse 63. 3% 20% 0% 16. 7% Child-Pugh score 7 8 9 70% 23. 3% 6. 7% 0 0 0 9. 9 ± 2. 5 6. 4 ± 1. 0 MELD score, (mean ± SD) C-SALT 60% 20% 10% Jacobson IM. EASL 2015, Abs. O 008
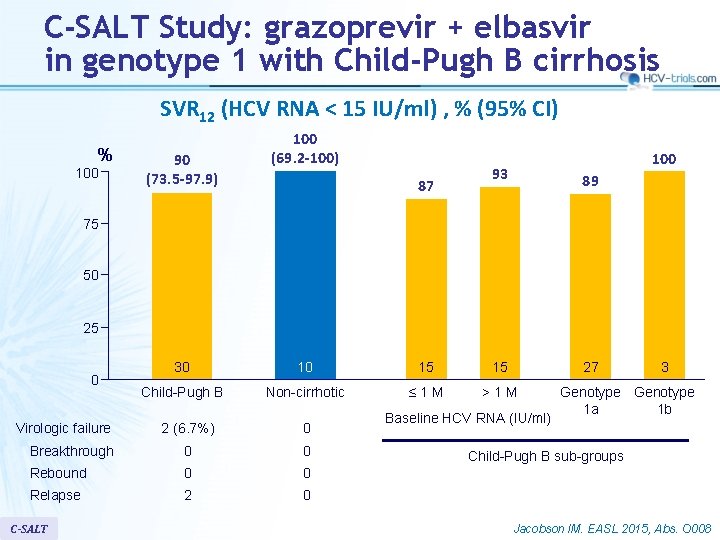
C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis SVR 12 (HCV RNA < 15 IU/ml) , % (95% CI) % 100 90 (73. 5 -97. 9) 100 (69. 2 -100) 87 100 93 89 75 50 25 0 Virologic failure 30 10 15 15 Child-Pugh B Non-cirrhotic ≤ 1 M >1 M 2 (6. 7%) 0 Breakthrough 0 0 Rebound 0 0 Relapse 2 0 C-SALT 27 Baseline HCV RNA (IU/ml) 3 Genotype 1 a 1 b Child-Pugh B sub-groups Jacobson IM. EASL 2015, Abs. O 008
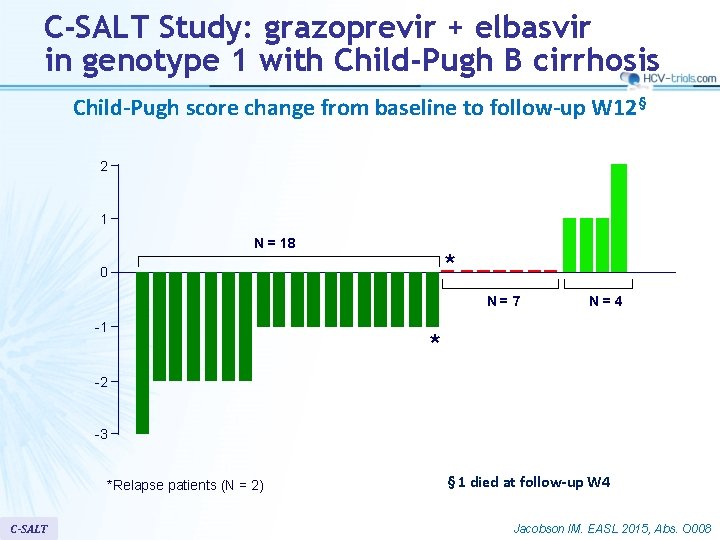
C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis Child-Pugh score change from baseline to follow-up W 12§ 2 1 N = 18 * 0 N=7 -1 N=4 * -2 -3 *Relapse patients (N = 2) C-SALT § 1 died at follow-up W 4 Jacobson IM. EASL 2015, Abs. O 008

C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis Pharmacokinetics § Plasma samples collected over 24 hours at treatment W 4 • PK group (non-cirrhotic, N = 9) with GZR 100 mg • Child-Pugh B (N = 9) with GZR 50 mg GMR (90% CI) Child-Pugh B / Non-cirrhotic Grazoprevir C 2 hr C 24 hr AUC 0 -24 1. 06 (0. 53, 2. 11) 1. 71 (0. 87, 3. 33) 1. 25 (0. 70, 2. 24) Elbasvir C 2 hr C 24 hr AUC 0 -24 0. 93 (0. 64, 1. 33) 1. 04 (0. 67, 1. 60) 0. 90 (0. 63, 1. 60) § GZR exposure was slightly higher (not significant) in patients with Child-Pugh B cirrhosis receiving 50 mg dose compared to non-cirrhotic patients receiving 100 mg dose § EBR (50 mg) PK was similar in both patient populations C-SALT Jacobson IM. EASL 2015, Abs. O 008

C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis Adverse events, N (%) Child-Pugh B N = 30 Non cirrhotic (PK) N = 10 0 0 4 (13. 3%)* 0 30% 16. 7% 10% 10% 30% 20% 0 50% Grade 3 -4 ALT/AST elevation, N (%) 0 0 Grade 3 -4 bilirubin elevation, N (%) 4 (13. 3) 0 Death**, N (%) 1 (3. 3) 0 Discontinuation due to an AE, N (%) Serious adverse event, N (%) Adverse events, N (%) Fatigue Arthralgia Nausea Pyrexia Headache * All unrelated to study treatment : hepatocellular carcinoma, N = 1; ascites and encephalopathy, N = 1 ; bacterial peritonitis, cerebral infarction and hepatic failure (**death at follow-up W 4) N = 1 ; hematemesis, N = 1 C-SALT Jacobson IM. EASL 2015, Abs. O 008

C-SALT Study: grazoprevir + elbasvir in genotype 1 with Child-Pugh B cirrhosis § Summary – High rates of virologic response were observed in Child-Pugh B patients receiving a combination of once-daily GZR 50 mg + EBR 50 mg – The regimen was well tolerated with no evidence of hepatotoxicity – Plasma GZR exposure was slightly higher in Child-Pugh B patients receiving 50 mg compared to non-cirrhotic patients receiving 100 mg – EBR exposure was similar in both Child-Pugh B and non-cirrhotic groups – This regimen was highly effective and well-tolerated in a traditionally hard-to-treat patient group with no currently approved treatment options C-SALT Jacobson IM. EASL 2015, Abs. O 008
- Slides: 7