Crystallization COURSE OUTCOME Describe the basic principles and

Crystallization

COURSE OUTCOME �Describe the basic principles and applications of crystallization processes. �Analyze the solubility data and binary mixture phase diagram. �Calculate the yield, heat and mass balances in crystallization. �Distinguish different type of equipment in crystallization process.

Crystallization �Crystallization is the (natural or artificial) process for the formation of solid crystals from a homogenous single phase solution. �Crystallization is also a chemical solid-liquid separation technique, in which isolation of a solute from the liquid solution to a pure solid crystalline phase occurs. �Generally, crystallization is the last chemical purification step. More than 80% of the substances used in pharmaceuti -cals, fine chemicals, agrochemicals, food and cosmetics

� In order for crystallization to take place, a solution must be "supersaturated". �Crystallization is based on the principles of solubility: ü Compounds (solutes) tend to be more soluble in hot liquids (solvents) than they are in cold liquids. ü If a saturated hot solution is allowed to cool, the solute is no longer soluble in the solvent and forms crystals of pure compound. ü Impurities are excluded from the growing crystals and the pure solid crystals can be separated from the dissolved impurities by filtration.
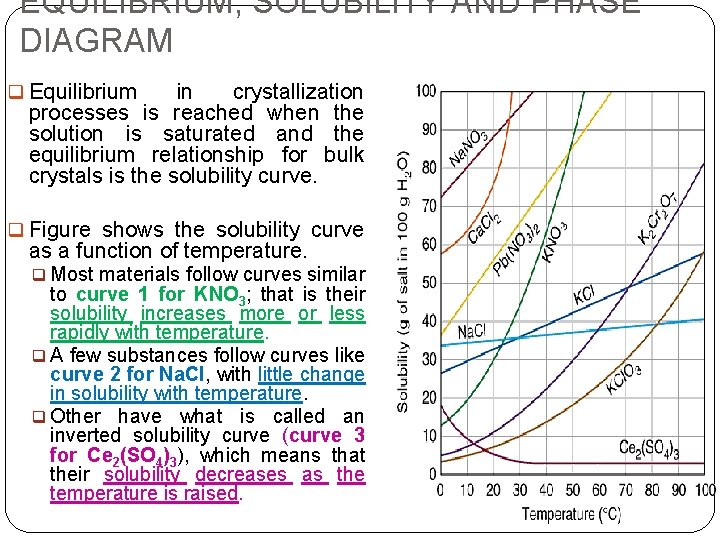
EQUILIBRIUM, SOLUBILITY AND PHASE DIAGRAM q Equilibrium in crystallization processes is reached when the solution is saturated and the equilibrium relationship for bulk crystals is the solubility curve. q Figure shows the solubility curve as a function of temperature. q Most materials follow curves similar to curve 1 for KNO 3; that is their solubility increases more or less rapidly with temperature. q A few substances follow curves like curve 2 for Na. Cl, with little change in solubility with temperature. q Other have what is called an inverted solubility curve (curve 3 for Ce 2(SO 4)3), which means that their solubility decreases as the temperature is raised.

EQUILIBRIUM, SOLUBILITY AND PHASE DIAGRAM q The solubility characteristic of a solute in a particular solvent is by far the most important property for determining (1) the best method for causing crystallization and (2) the ease or difficulty is growing crystals. q Crystallization by cooling is only attractive for compounds having a solubility that decreases rapidly with decreasing temperature above ambient temperature. For instance, for Na. Cl, crystallization by cooling would be undesirable because the solubility decreases only by about 10% when the temperature decreases from 100 until 0 o. C. For most soluble inorganic compounds, cooling by evaporation is the preferred technique. q Many important inorganic substances crystallize with water of crystallization. In some systems, several different hydrates are formed, depending on the concentration and temperature and phase equilibrium in such systems can be quite complicated.
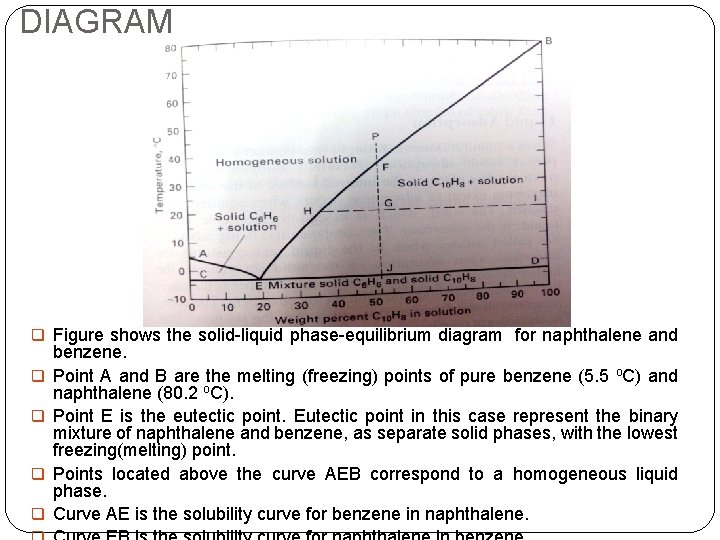
DIAGRAM q Figure shows the solid-liquid phase-equilibrium diagram for naphthalene and q q benzene. Point A and B are the melting (freezing) points of pure benzene (5. 5 o. C) and naphthalene (80. 2 o. C). Point E is the eutectic point. Eutectic point in this case represent the binary mixture of naphthalene and benzene, as separate solid phases, with the lowest freezing(melting) point. Points located above the curve AEB correspond to a homogeneous liquid phase. Curve AE is the solubility curve for benzene in naphthalene.
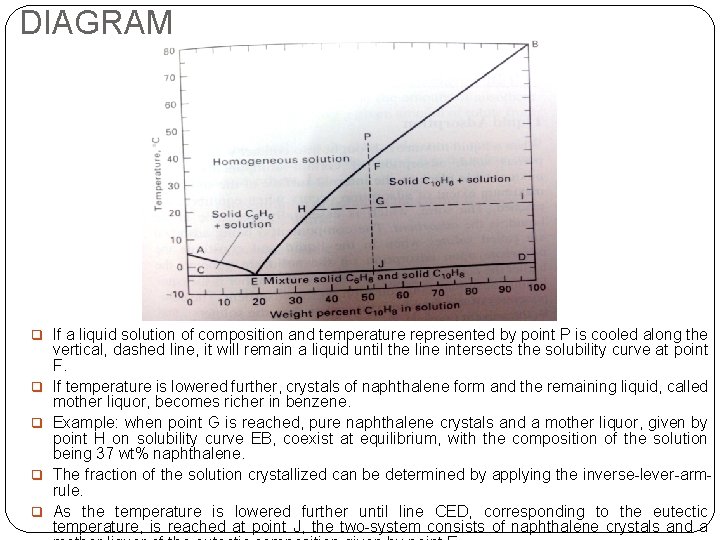
DIAGRAM q If a liquid solution of composition and temperature represented by point P is cooled along the q q vertical, dashed line, it will remain a liquid until the line intersects the solubility curve at point F. If temperature is lowered further, crystals of naphthalene form and the remaining liquid, called mother liquor, becomes richer in benzene. Example: when point G is reached, pure naphthalene crystals and a mother liquor, given by point H on solubility curve EB, coexist at equilibrium, with the composition of the solution being 37 wt% naphthalene. The fraction of the solution crystallized can be determined by applying the inverse-lever-armrule. As the temperature is lowered further until line CED, corresponding to the eutectic temperature, is reached at point J, the two-system consists of naphthalene crystals and a

Mechanism of Crystallization �Crystallization from solution can be thought of as a 3 step processes which are : Ø Supersaturation -The formation of supersaturated solution Ø Nucleation - The formation of crystal nuclei. Ø Crystal growth - The growth of the nuclei to larger size

Supersaturation �Supersaturation refers to a state in which the liquid (solvent) contains more dissolved solids (solute) than can ordinarily be accommodated at that temperature. � Based on Ostwald-Miers, the field of supersaturation consists of three regions and the relationship between supersaturation and spontaneous crystallization led to the diagrammatic representation (Fig. 1). The regions of supersaturation are describe as follows Ø The stable (undersaturated) zone – Crystallization is impossible. The solutions have concentrations lower than the solubility curve at a specific temperature, are considered as being undersaturated. In such solutions, any crystals present will re-dissolve. Ø The metastable (supersaturated) zone- Spontaneous nucleation does not occur. However existing crystals may grow if a surface on which growth can occur is
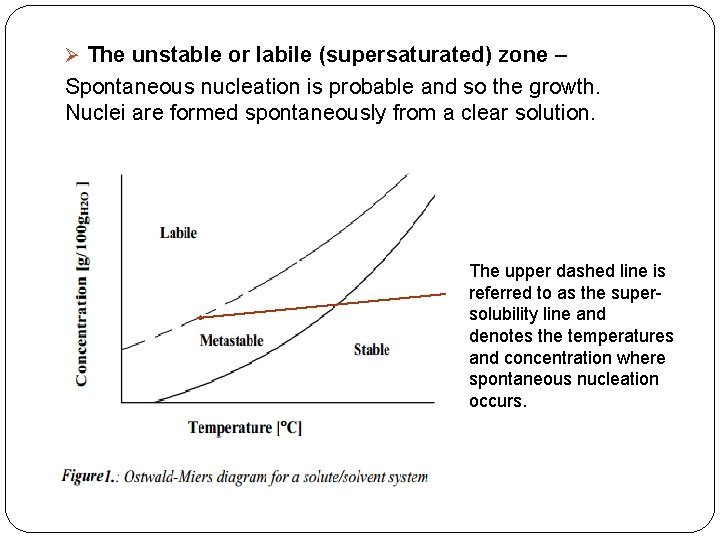
Ø The unstable or labile (supersaturated) zone – Spontaneous nucleation is probable and so the growth. Nuclei are formed spontaneously from a clear solution. The upper dashed line is referred to as the supersolubility line and denotes the temperatures and concentration where spontaneous nucleation occurs.

�Supersaturation can be achieved using several methods. These include : Ø Changing solution temperature (cooling crystallization) Ø Evaporation of solvent (evaporative crystallization), Ø Changing the solvent composition (antisolvent crystallization). Ø Chemical reactions (reactive crystallization/precipitation) �Each process has its possibilities and limitations.

Nucleation Primary Nucleation � The formation of a new crystalline entity from a solution starts through the nucleation process. � Nucleation is defined as the series of atomic or molecular processes by which the atoms or molecules of a reactant phase rearrange into a cluster of the product phase large enough as to have the ability to grow irreversibly to a macroscopically larger size. � The cluster is defined as nucleus or critical nuclei. � Nucleation can be homogeneous, in the absence of foreign particles or crystals in the solution, or heterogeneous, in the presence of foreign particles in the solution.
Secondary Nucleation � Secondary nucleation takes place when nucleation is induced by the presence of crystals of the same substance. � Secondary nucleation requires "seeds" or existing crystals to perpetuate crystal growth

Crystal Growth � Crystal growth is the series of processes by which an atom or a molecule is incorporated into the surface of a crystal, causing an increase in size. � These different processes can be summarized into four steps which are: 1) transport of atoms through solution 2) attachment of atoms to the surface 3) movement of atoms on the surface 4) attachment of atoms to edges and kinks. � The first process is the so-called transport process, whereas 2– 4 are referred to as surface processes (and may involve several sub steps). � Since these different steps normally occur in series, the slowest process will control the overall crystal growth.

Crystals �Can appear as polyhedrons, or solids formed by plane faces when allowed to form freely. �Shape is determined by atomic arrangements. Some directions grow faster than others �Morphology can be distinct for the conditions and speed of crystal nucleation/growth. �Relative sizes of a crystal in a particular system can vary considerably –resulting in a variety of crystal shapes. This variation is called a habit. �Crystal habit is influence by the conditions of crystallization, particularly by the impurities present and by the particular solvent or solvents used. �Impurities can inhibit the growth of a crystal in certain directions.
Polyhedrons Shape

CALCULATION IN CRYSTALLIZATION

Material Balances � The solution and the solid crystals are in contact for enough time to reach equilibrium. Hence, the solution is saturated at the final temperature of the process, and the final concentration of the solute in the solution can be obtained from the solubility curve. � The yield of crystals can be calculated knowing the initial concentration of solute, the final temperature, and the solubility at this temperature. � In making the material balances, the calculations are straightforward when the solute crystals are anhydrous. Simple water and solute material balances are made. When the crystallizations are hydrated, some of the water

Example 1 A salt solution weighing 10000 kg with 30 wt% Na 2 CO 3 is cooled to 293 K (20°C). The salt crystallizes as the decahydrate. What will be the yield of Na 2 CO 3 • 10 H 2 O crystals if the solubility is 21. 5 kg anhydrous Na 2 CO 3/100 kg of total water? Do this for the Following cases: (a) Assume that no water is evaporated. (b) Assume that 3% of the total weight of the solution is lost by

Example 1 Solution The molecular weights are 106. 0 for Na 2 CO 3, 180. 2 for 10 H 20, and 286. 2 for Na 2 CO 3 • 10 H 2 O. The process flow diagram is shown in Fig. 3, with W being kg H 2 O evaporated, S kg solution (mother liquor), and C kg (13) crystals of Na 2 CO 3 • 10 H 2 O. Making a material isbalance : of water in the crystals. where (180. 2)/(286. 2) wt fraction (a), where W = 0.

SOLUTION: Making a balance for Na 2 CO 3, (14) Solving the two equations simultaneously, C = 6370 kg of Na 2 C • 10 H 2 O crystals and S = 3630 kg solution. For part (b), W = 0. 03(10000) = 300 kg H 2 O. Equation (13) becomes (15) Equation (14) does not change, since no salt is in the W stream Solving Eqs. (14) and (15)simultaneously, C = 6630 kg of Na 2 C • 10 H 2 O crystals and S = 3070 kg solution.

Heat Balance � When a compound whose solubility increases as temperature increases dissolves, there is an absorption of heat, called the heat of solution–occurs when the solubility decreases as the temperature increases � At equilibrium the heat of crystallization is equal to the negative of the heat of solution at the same concentration in solution. � The enthalpy H 1 of the entering solution at the initial temperature is read off the chart, where H 1 is k. J for the total feed. The enthalpy H 2 of the final mixture of crystals and mother liquor at the final temperature is also read off. If some evaporation occurs, the enthalpy Hv of the water vapor is obtained from the steam tables. Then the total heat absorbed q in k. J is � If q is positive, heat must be added to the system. If it is negative, heat is evolved or given off. � Heat absorbed, q = +’ve , Heat given off , q = -’ve

Example 2 A feed solution of 2268 kg at 327. 6 K(54. 4°C) containing 48. 2 kg Mg. SO 4/100 kg total water is cooled to 293. 2 K (20°C), where Mg. SO 4 • 7 H 2 O crystals are removed. The solubility of the salt is 35. 5 kg Mg. SO 4/100 kg total water. The average heat capacity of the feed solution can be assumed as 2. 93 k. J/kg • K. The heat of solution at 291. 2 K(18°C) is 13. 31 x 103 k. J/kgmol Mg. SO 4 • 7 H 2 O. Calculate the yield of crystals and make a heat balance to determine the total heat absorbed, q , assuming that no water is vaporized

SOLUTION: Solution Making a water balance and a balance for Mg. SO 4 using equations similar to (13) and (14) in Example 1, C = 616. 9 kg Mg. SO 4 • 7 H 2 O crystals and S = 1651. 1 kg solution. To make a heat balance, a datum of 293. 2 K (20°C) will be used. The molecular weight of Mg. SO 4 • 7 H 20 is 246. 49. The enthalpy of the feed is H 1:

SOLUTION: The heat of solution is -(13. 31 x 10 )/246. 49 = -54. 0 k. J/kg crystals. 3 Then the heat of crystallization is -(-54. 0) = +54. 0 k. J/kg crystals, or 54. 0(616. 9) = 33312 k. J. This assumes that the value at 291. 2 K is the same as at 293. 2 K. The total heat absorbed, q, is Since q is negative, heat is given off and must be removed.

Equipment for Crystallization Tank Crystallization � Hot saturated solutions are allowed to cool in open tanks � After a period of time, the mother liquor is drained and the crystals removed � Nucleation and the size of crystals are difficult to control � Labor cost are very high � Has limited application; used to produce certain fine chemical and pharmaceutical products.

Equipment for Crystallization Scraped surface crystallizers � One type of scraped surface crystallizer is the Swenson-Walker crystallizer, which consists of an open trough 0. 6 m wide with a semicircular bottom having a cooling jacket inside. � Slow-speed spiral agitator rotates and suspends the growing crystals on turning. � Blades pass close to the wall and break off any deposits of crystals on the cooled wall. � Used in crystallizing ice cream and plasticizing margarine.

Equipment for Crystallization Circulating-liquid crystallizer evaporated- � Supersaturation is generated by evaporation. � Circulating liquid is drawn by the screw pump down inside the tube side of condensing steam heater � Heated liquid then flows into the vapor space, where flash evaporation occurs, giving some supersaturation. � The vapor leaving is condensed. � The supersaturated liquid flow down the downflow tube and then up through the bed fluidized and agitated crystals, which are growing in size. � The living saturated liquid then goes back as a recycle stream to the heater, where it is joined by the entering feed.

Equipment for Crystallization Circulating-magma vacuum crystallizer � The magma or suspension of crystals is circulated out the main body through a circulating pipe by a screw pump � Magma flows through a heater, where its temperature is raised 2 -6 K. � The heated liquor then mixes with body slurry and boiling occurs at liquid surface � This cause supersaturation in the swirling liquid near the surface, which results in deposits on the swirling suspended crystals until they leave again via the circulating pipe � The vapors leave through the top � A steam-jet ejector provides the vacuum
- Slides: 30