Crystallisation Gavin Duffy DIT Kevin St Learning Outcomes



















- Slides: 35

Crystallisation Gavin Duffy DIT, Kevin St.

Learning Outcomes After this lecture you should be able to…. . w Describe crystal growth and nucleation w Define the constituents of a solution and degrees of saturation w Describe the solubility curve w List three methods of achieving supersaturation/crystallisation w Describe nucleation and crystal growth w Give an example of crystallisation w Analyse a crystallisation process in a pharmaceutical plant w Explain how to crystallise by cooling

Crystallisation - Introduction w Crystallization refers to the formation of solid crystals from a homogeneous solution. w It is a solid-liquid separation technique w Used to produce n Sodium chloride n Sucrose from a beet solution n Desalination of sea water n Separating pharmaceutical product from solvents n Fruit juices by freeze concentration w Crystallisation requires much less energy than evaporation n e. g. water, enthalpy of crystallisation is 334 k. J/kg and enthalpy of vaporisation is 2260 k. J/kg

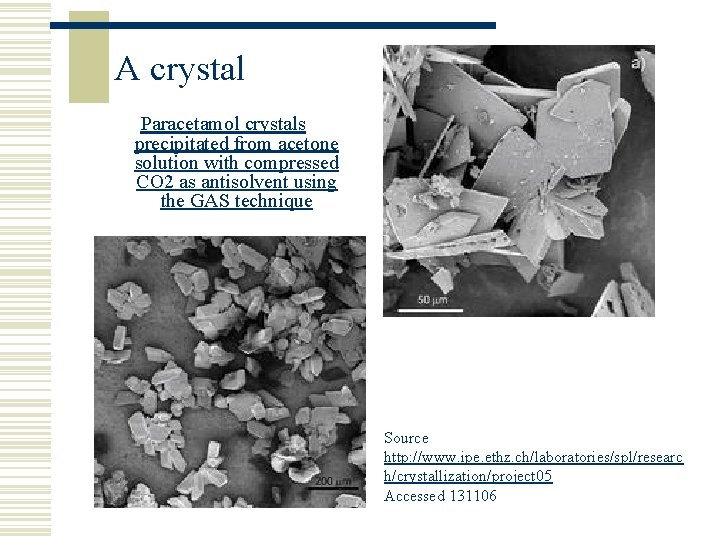
What is a crystal? w A crystal is a solid form of substance (ice) w Some crystals are very regularly shaped and can be classified into one of several shape categories such rhombic, cubic, hexagonal, tetragonal, orthorhombic, etc. w With pharmaceuticals, crystals normally have very irregular shapes due to dendritic growth which is a spiky type appearance like a snowflake. It can be difficult to characterise the size of such a crystal. w Crystals are grown to a particular size that is of optimum use to the manufacturer. Typical sizes in pharmaceutical industry are of the order of 50 m.

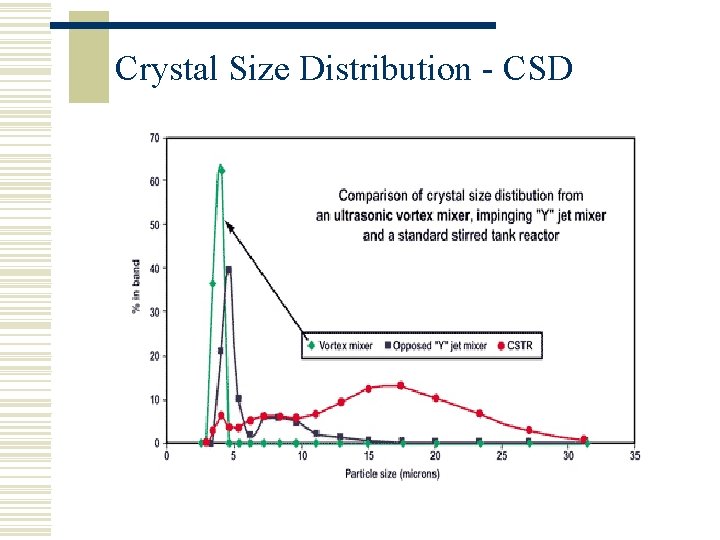
Crystallisation In general, crystallisation should be a straightforward procedure. The objective is to grow crystals of a particular size or crystal size distribution (CSD). If this is not successful, problems that can occur are: w Inconsistency from batch to batch w Difficult to stir and filter w Crystals damaged in filtration/agitation w Creation of polymorphs w Difficult to dry

Crystal Size Distribution - CSD

A crystal Paracetamol crystals precipitated from acetone solution with compressed CO 2 as antisolvent using the GAS technique Source http: //www. ipe. ethz. ch/laboratories/spl/researc h/crystallization/project 05 Accessed 131106

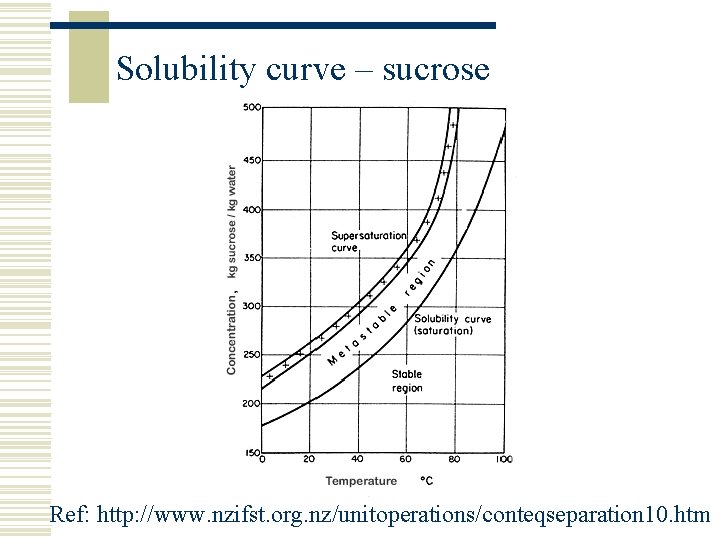
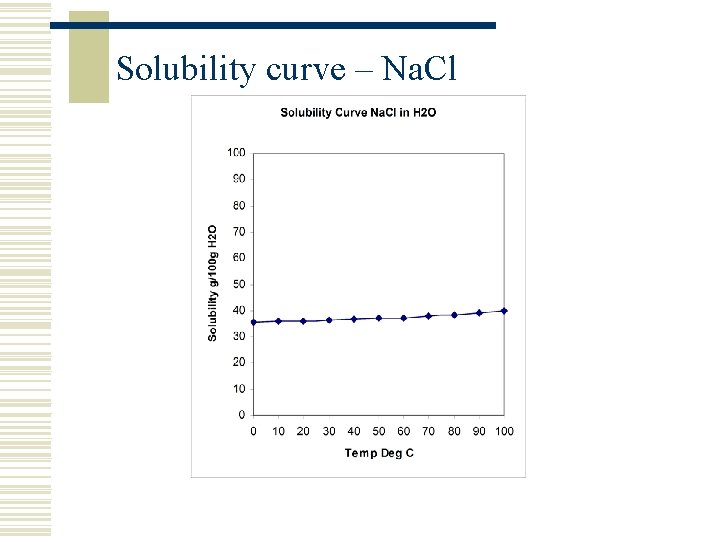
Solutions, Solubility and Solvent w A solid substance (solute) is termed soluble if it can dissolve in a liquid (the solvent) to create a solution w The solution is a homogenous mixture of two or more components w Solubility is normally (but not always) a function of temperature w Solubility can change if the composition of the solvent is changed (e. g. if another solvent is added) w Solubility is usually measured as how many grams of solvent can be dissolved in 100 grams of solute

Solubility curve – sucrose Ref: http: //www. nzifst. org. nz/unitoperations/conteqseparation 10. htm

Solubility curve – Na. Cl

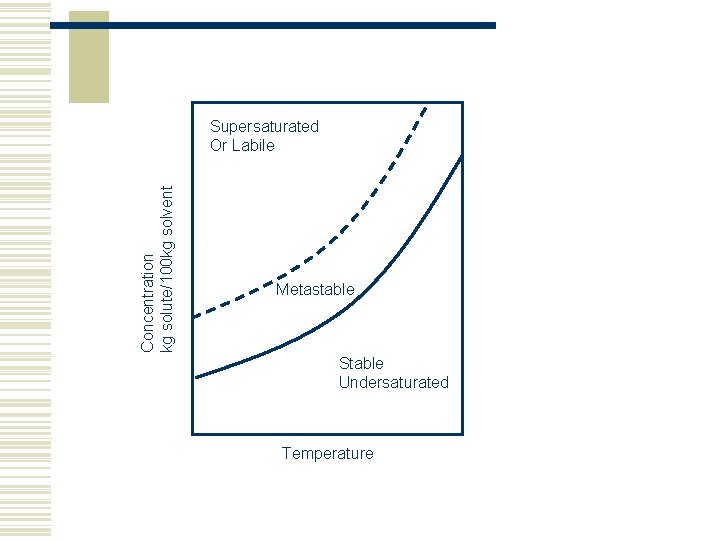
Saturation w An Unsaturated or Undersaturated solution can dissolve more solute w A saturated solution is one which contains as much solute as the solvent can hold w A Supersaturated solution contains more dissolved solute than a saturated solution, i. e. more dissolved solute then can ordinarily be accommodated at that temperature w Two forms of supersaturation n Metastable – just beyond saturation n Labile – very supersaturated w Crystallisation is normally operated in the metastable region

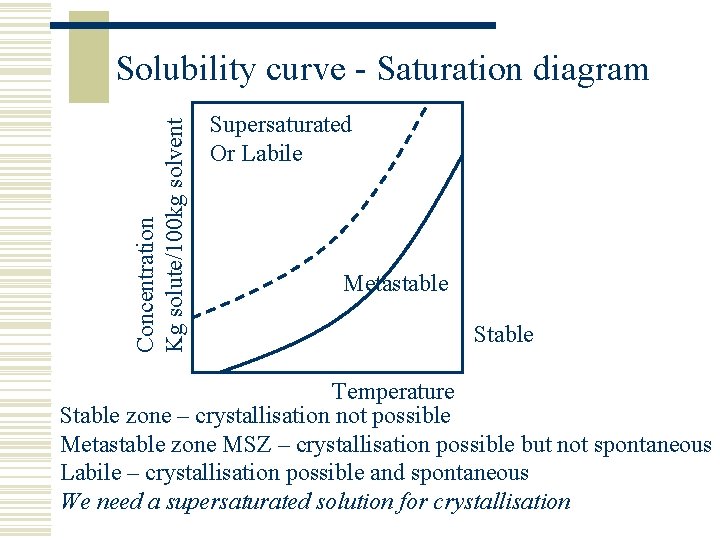
Concentration Kg solute/100 kg solvent Solubility curve - Saturation diagram Supersaturated Or Labile Metastable Stable Temperature Stable zone – crystallisation not possible Metastable zone MSZ – crystallisation possible but not spontaneous Labile – crystallisation possible and spontaneous We need a supersaturated solution for crystallisation

Concentration kg solute/100 kg solvent Supersaturated Or Labile Metastable Stable Undersaturated Temperature

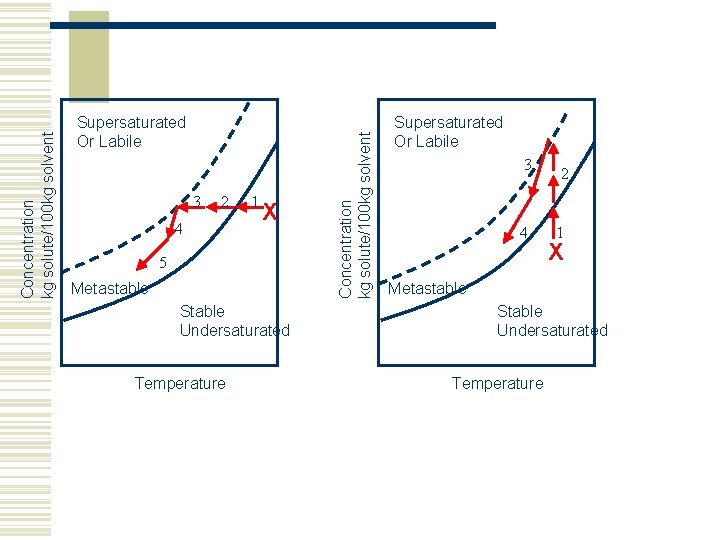
3 2 4 1 X 5 Metastable Stable Undersaturated Temperature Concentration kg solute/100 kg solvent Supersaturated Or Labile 3 4 2 1 X Metastable Stable Undersaturated Temperature

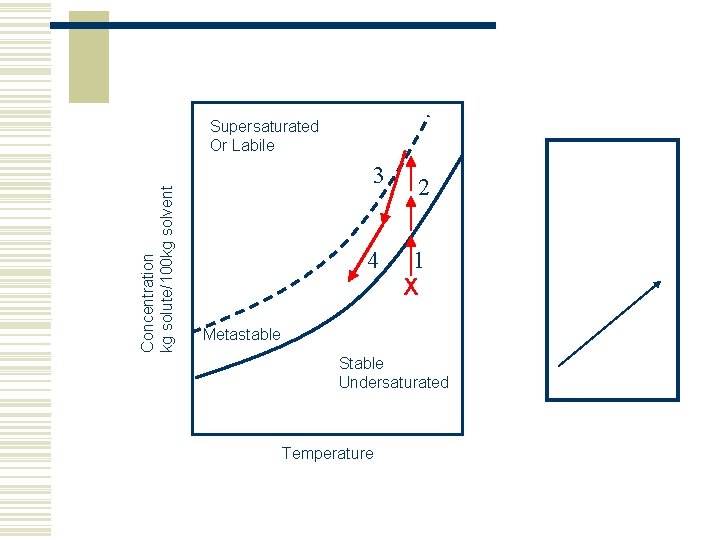
Concentration kg solute/100 kg solvent Supersaturated Or Labile 3 2 4 1 X Metastable Stable Undersaturated Temperature

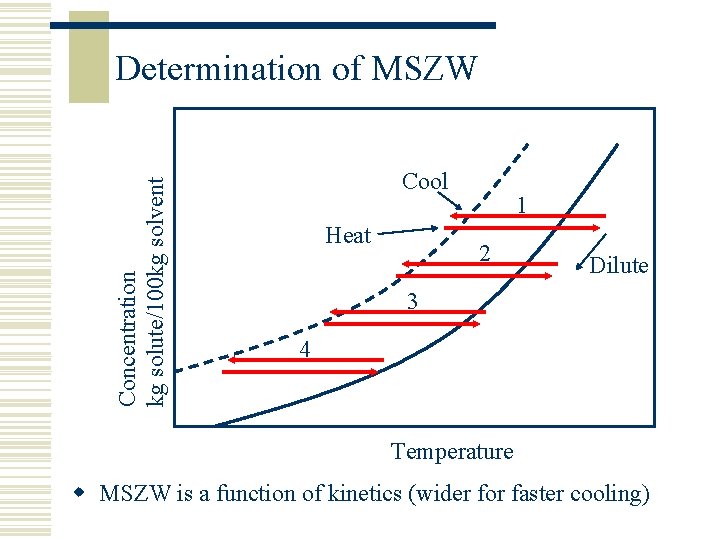
Concentration kg solute/100 kg solvent Determination of MSZW Cool Heat 1 2 Dilute 3 4 Temperature w MSZW is a function of kinetics (wider for faster cooling)

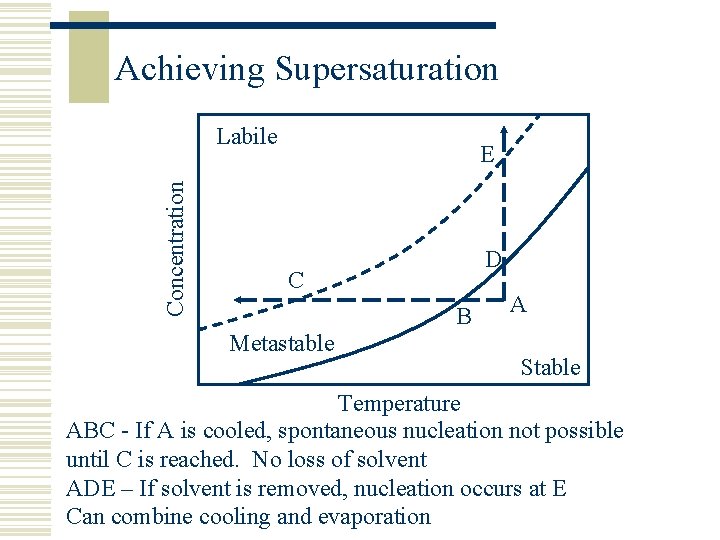
Achieving Supersaturation Concentration Labile E D C B Metastable A Stable Temperature ABC - If A is cooled, spontaneous nucleation not possible until C is reached. No loss of solvent ADE – If solvent is removed, nucleation occurs at E Can combine cooling and evaporation

Crystallisation Techniques w In general crystallisation is achieved by n Cooling a solution l If supersaturation is a function of temperature n Removal of the solvent by evaporation l Where supersaturation is independent of temperature (e. g. common salt) n Addition of another solvent to reduce solubility l When solubility is high and above methods are not desirable, or in combination with above methods l The new solvent is called the anti solvent and is chosen such that the solubility is less in this new solution than it was before

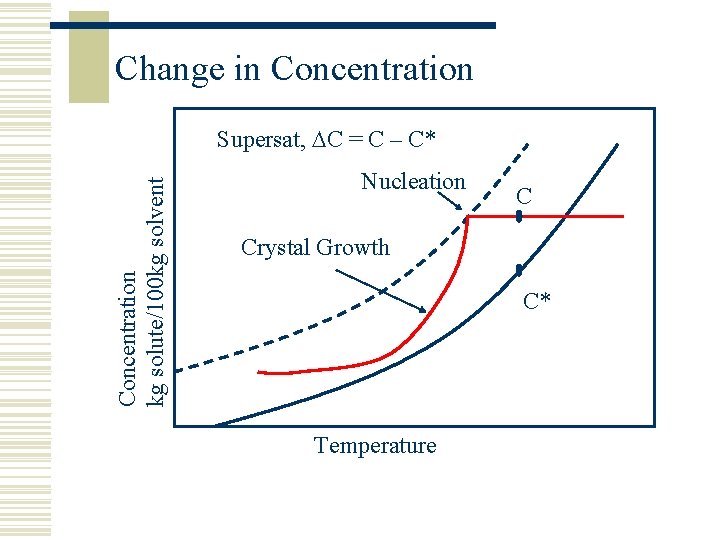
Change in Concentration kg solute/100 kg solvent Supersat, C = C – C* Nucleation C Crystal Growth C* Temperature

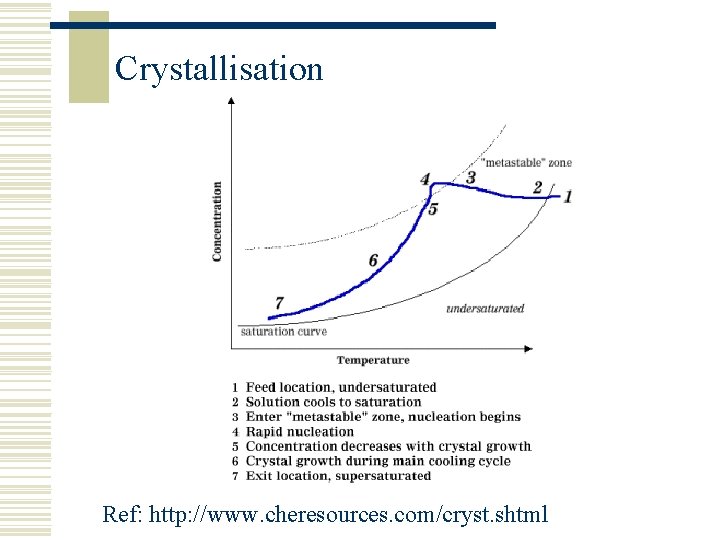
Crystallisation Ref: http: //www. cheresources. com/cryst. shtml

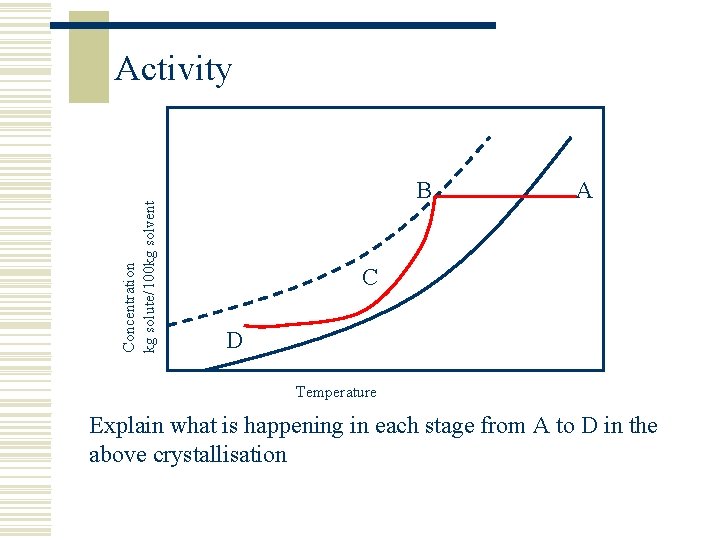
Concentration kg solute/100 kg solvent Activity B A C D Temperature Explain what is happening in each stage from A to D in the above crystallisation

Supersaturation, C w Supersaturation is the driving force for n Nucleation n Crystal Growth w Creation and control of supersaturation is the key to successful crystallisation w High Crystal Growth + High Nucleation w High nucleation means a lot of fines (filtration problems) w High crystal growth means inclusion of impurities w C is usually maintained at a low level in the pharmaceutical industry so the right CSD is achieved

Activity – How would you crystallise? w Have a look at the solubility curves provided w List the three techniques for achieving supersaturation w Which would you use and why?

Nucleation w Crystallisation starts with Nucleation w There are two types of nucleation – Primary and Secondary w Primary relates to the birth of the crystal, where a few tens of molecules come together to start some form of ordered structure w Secondary nucleation can only happen if there are some crystals present already. It can occur at a lower level of supersaturation than primary nucleation. w Often, industrial crystallisers jump straight to secondary nucleation by ‘seeding’ the crystalliser with crystals prepared earlier

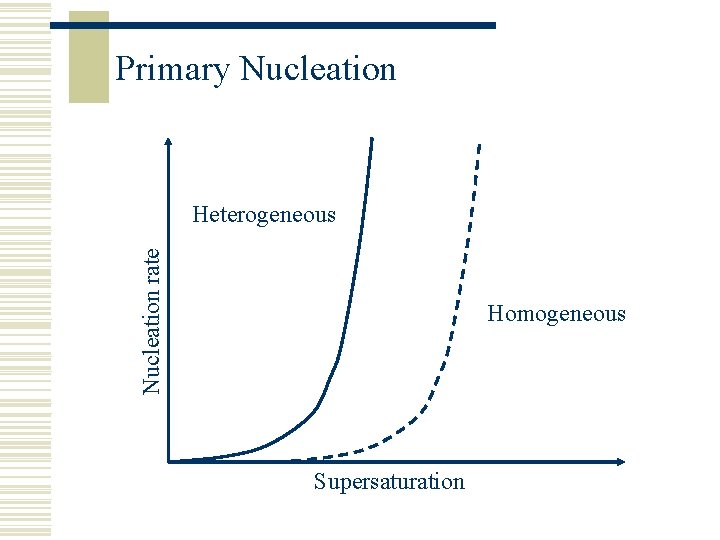
Primary Nucleation w The birth of a new crystal is complex and involves the clustering of a few tens of molecules held together by intermolecular forces w Homogeneous – small amounts of the new phase are formed without any help from outside w Heterogeneous – nucleation is assisted by suspended particles of a foreign substance or by solid objects such as the wall of the container or a rod immersed in the solution – these objects catalyse the process of nucleation so it occurs at lower levels of supersaturation w Homogenous conditions are difficult to create so heterogeneous nucleation is more normal in industrial crystallisation (if it is not seeded)

Primary Nucleation rate Heterogeneous Homogeneous Supersaturation

Secondary Nucleation w Secondary nucleation is an alternative path to primary nucleation and occurs when seed crystals are added w Nucleation occurs at a lower supersaturation than primary when crystals are already present w Secondary nucleation is due to: n Contact nucleation – crystals are created by impact with agitator or vessel wall. Nuclei are created by striking a crystal – the number created is related to the supersaturation and the energy of impact. Can occur at low supersaturation. n Shear nucleation – shear stresses in the boundary layer of fluid flow create new crystals/nulcei. Embryos are created and swept away that would have been incorporated into an existing crystal w Very important in industrial crystallisers as this is the main type of crystal growth used w Difficult to predict or model nucleation rates

Supersaturation and Crystal Growth w For low supersaturation primary nucleation is not widespread. Secondary nucleation on existing crystals is more likely. Result is small numbers of large crystals w For high supersaturation primary nucleation is widespread. This results in many crystals of small size. w Slow cooling with low supersaturation creates large crystals w Fast cooling from high supersaturation creates small crystals w Agitation reduces crystal size by creating more dispersed nucleation w Rate of cooling can affect purity of product - see handout on slow cooling v rapid cooling

Seeding w The type or quality of seed used can influence the crystallisation process w Good seed results in a good crystallisation, i. e. a particle size distribution that does not include fines w Bad seed can increase the amount of fines produced w Good and Bad can be defined by the seed crystal size w Source of seed can be n Material left from the last batch (no tight control on particle size) n Specially prepared material or material from a good batch (tight particle size distribution)

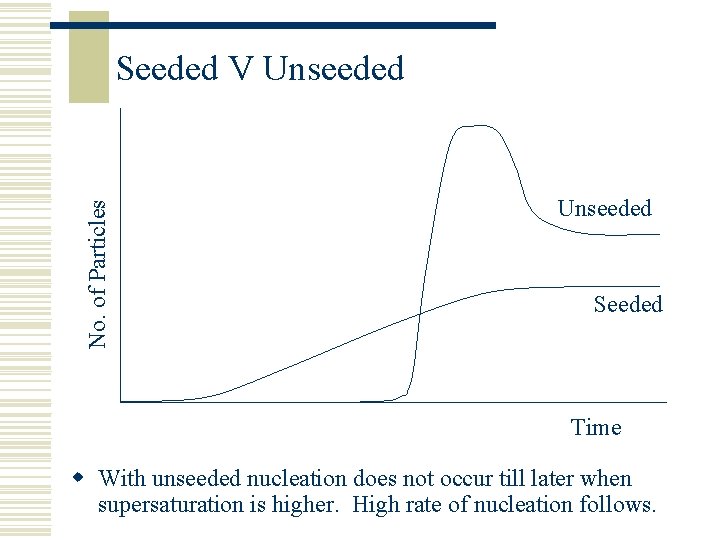
When to Seed? w Seed can be added dry to the crystalliser w Allow time for dispersion throughout the crystalliser – this can take several hours w Never seed to the right of the solubility curve – the solution is not yet ready w Never seed to the left of the solubility curve – nucleation is already happening w Seed half way between the two

No. of Particles Seeded V Unseeded Seeded Time w With unseeded nucleation does not occur till later when supersaturation is higher. High rate of nucleation follows.

Seeding – advantages, disadvantages w Advantages include n Point of nucleation from batch to batch is repeatable n Reduces the number of fines n Improves predictability of scale up n Can prevent polymorphism w Disadvantages n Experience has shown that not any seed will do, good quality seed is needed n Extra addition point on vessel or hand hole is usually opened to manually add seed which could create health and safety issues

Crystal Growth w Once nucleation has occurred crystal growth can happen w The objective of crystallisation is to produce the required crystal size distribution (CSD) w The actual CSD required depends on the process w Crystal growth rate has proved difficult to model and empirical relationships developed from laboratory tests are generally used w Two steps to crystal growth n Diffusion of solute from bulk solution to the crystal surface n Deposition of solute and integration into crystal lattice

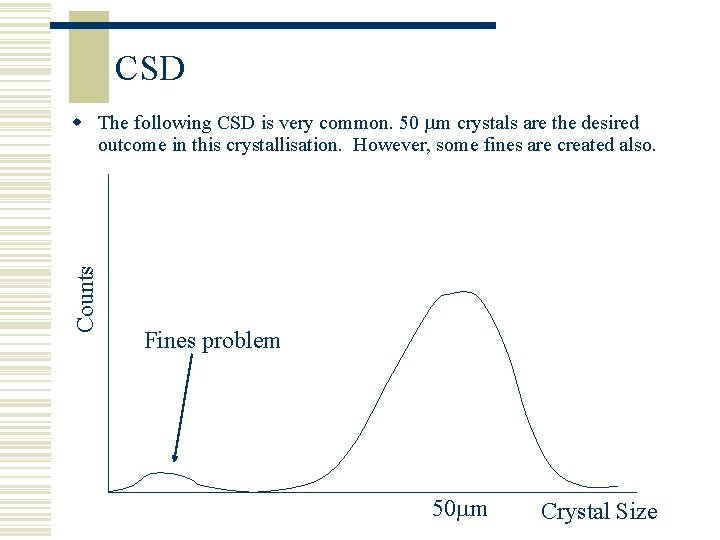
CSD Counts w The following CSD is very common. 50 m crystals are the desired outcome in this crystallisation. However, some fines are created also. Fines problem 50 m Crystal Size

Impurities and Crystal Growth w Impurities can prevent crystal growth w If concentration of impurities is high enough crystals will not grow w Should not be an issue in the pharmaceutical industry w For example, the production of non crystalline sweets such as lollipops (sugar crystals give an unwanted grainy texture) n Addition of acid breaks sucrose into fructose and glucose n This makes it difficult for sucrose crystals to form because the impurities damage the structure n Addition of other sugars creates the same result