Crystal the Closed Vial Technology A Revolution in











- Slides: 15

Crystal , the Closed Vial Technology A Revolution in Aseptic Filling Overall Introduction Document January 2009 Copyright © 2008 | Aseptic Technologies | All rights reserved

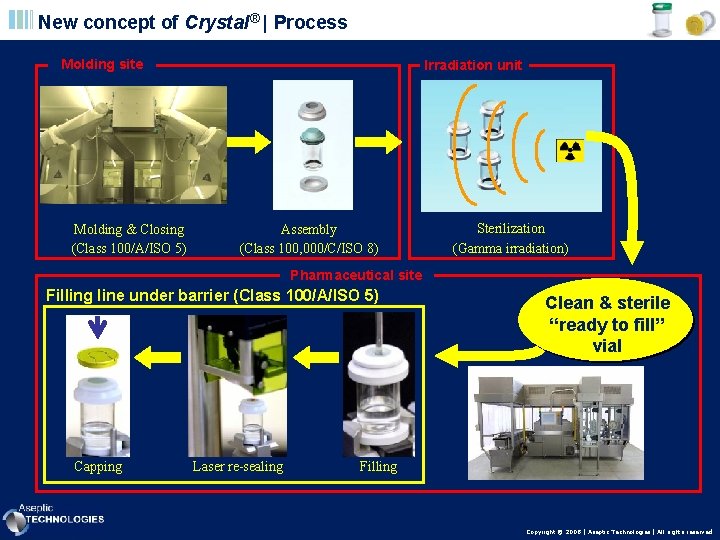
New concept of Crystal® | Process Molding site Molding & Closing (Class 100/A/ISO 5) Irradiation unit Assembly (Class 100, 000/C/ISO 8) Sterilization (Gamma irradiation) Pharmaceutical site Filling line under barrier (Class 100/A/ISO 5) Capping Laser re-sealing Clean & sterile “ready to fill” vial Filling Copyright © 2008 | Aseptic Technologies | All rights reserved

The Crystal® Closed Vial Technology Key advantages The Vial Body made of COC and stopper of thermoplastic elastomer: • Hydrophobic material to prevent electrostatic interaction with soluble proteins • Unbreakable vial • Glass-like transparency • Specially designed stopper minimizing loss of residual volume • Exceptional inertness: low absorption, no ion and very few leachables • Excellent moisture barrier • Bio-compatible • Ability to reduce oxygen concentration to 0. 1% Crystal® vial is particularly suitable for: • High value products • Toxic products Copyright © 2008 | Aseptic Technologies | All rights reserved

The Crystal® Closed Vial Technology Key advantages The Filling process The Crystal® closed vial enables to: • Eliminate most complex steps of aseptic filling – – no no WFI washing hot-air tunnel siliconization stoppering • Limit risk of batch release thanks to process simplification • Secure potent and high-value products Crystal® Technology drastically simplifies aseptic filling operations Copyright © 2008 | Aseptic Technologies | All rights reserved

Vial description Flip away cap Ensures sterility assurance level of the piercing area Top ring Ensures closure integrity Stopper Made of Thermoplastic Elastomer (TPE) Allows laser resealing after filling Vial body Made of Cyclo-olefin co-polymer (COC) Contains volume from 0. 2 to 20 ml Vials up to 50 ml are feasible Bottom ring Ensures mechanical stability Crystal® vial is closed in a class 100 A room before being sterilized by gamma irradiation Copyright © 2008 | Aseptic Technologies | All rights reserved

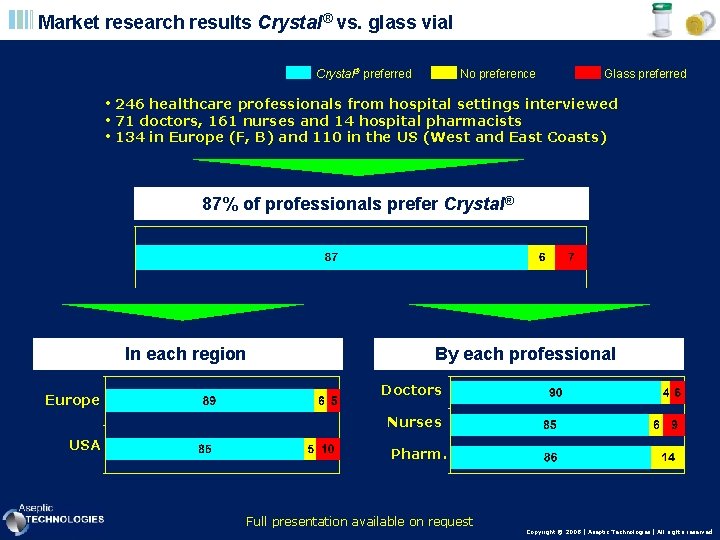
Market research results Crystal® vs. glass vial Crystal preferred No preference Glass preferred • 246 healthcare professionals from hospital settings interviewed • 71 doctors, 161 nurses and 14 hospital pharmacists • 134 in Europe (F, B) and 110 in the US (West and East Coasts) 87% of professionals prefer Crystal® In each region Europe By each professional Doctors Nurses USA Pharm. Full presentation available on request Copyright © 2008 | Aseptic Technologies | All rights reserved

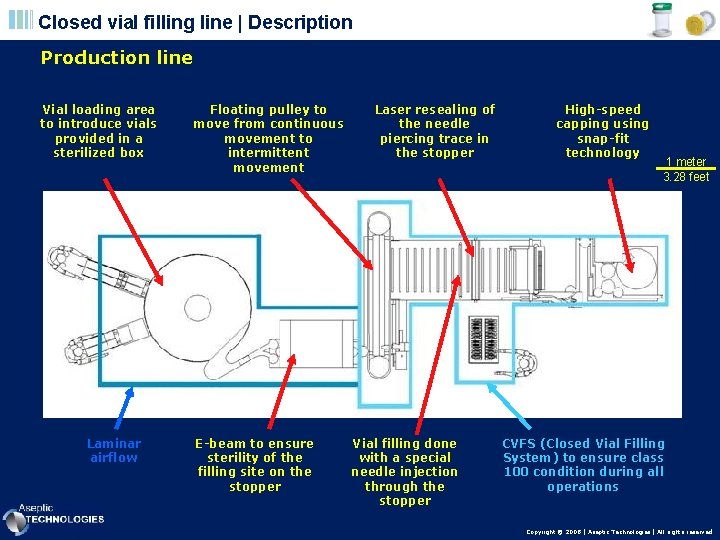
Closed vial filling line | Description Production line Vial loading area to introduce vials provided in a sterilized box Laminar airflow Floating pulley to move from continuous movement to intermittent movement E-beam to ensure sterility of the filling site on the stopper Laser resealing of the needle piercing trace in the stopper Vial filling done with a special needle injection through the stopper High-speed capping using snap-fit technology 1 meter 3. 28 feet CVFS (Closed Vial Filling System) to ensure class 100 condition during all operations Copyright © 2008 | Aseptic Technologies | All rights reserved

Three formats of line available Laboratory line (Crystal L 1) • Designed to fill very small batches (<5, 000 vials) • Capacity: about 10 vials/min • Able to handle all formats of Crystal vials (1 to 50 ml) and glass vials • Equipped with a robot performing filling, re-sealing and capping • Space needed: ~8 m² (machine footprint: 1 m²) • Designed to fill clinical batches and small commercial batches (Crystal C 2 or C 3) • Capacity: up to 75 vials/min • Able to handle all formats of vials (1 to 50 ml) • Equipped with three filling needles and lasers (C 3) • Space needed: 40 m² (machine footprint: 9 m²) Clinical line • Designed to fill large commercial batches (Crystal P 6 to P 24) • Capacity: up to 600 vials/min • Able to handle all formats of vials (1 to 50 ml) • Equipped with 6 to 24 filling needles and lasers • Space needed: 100 m² (machine footprint: 44 m²) • Capacity increase is feasible by adding filling heads and lasers Production line Copyright © 2008 | Aseptic Technologies | All rights reserved

Lyophilization in Crystal® Process 1. Normal vial filled but not laser resealed 2. Penetrator (funnel shape) placed in line on vial top 3. Lyo shelve moved down to push penetrator and re-open piercing trace 4. Lyophilization done through piercing trace 5. Penetrator withdrawn and vial laser re-sealed and capped Pusher Lyo shelve Tests conducted using identical cycles compared to glass vials: üLactose 5 and 10%: successful üSucrose 5 and 10%: successful üViruses: same virus titer as in glass Copyright © 2008 | Aseptic Technologies | All rights reserved

Key advantages of Crystal® Technology For whom? • For your regulatory • Provide a top-class sterility assurance • For your supply chain • Secure the products inside an • For your • Simplify entire filling process (lean • For your QA/QC team • Quicker initial validation and less follow • For your marketing • Facilitate handle by healthcare team SAFER team manufacturing team EASIER Why? team level thanks to a closed ready to fill container unbreakable and coded container manufacturing) -up validation work specialists • For your financial team • Reduce capital expenses INCREASE PROFIT • For your • Reduce operating expenses • For your sales team • Boost sales thanks to better perception manufacturing team despite vials more expensive than unclean non-sterile glass vials by end-users Copyright © 2008 | Aseptic Technologies | All rights reserved

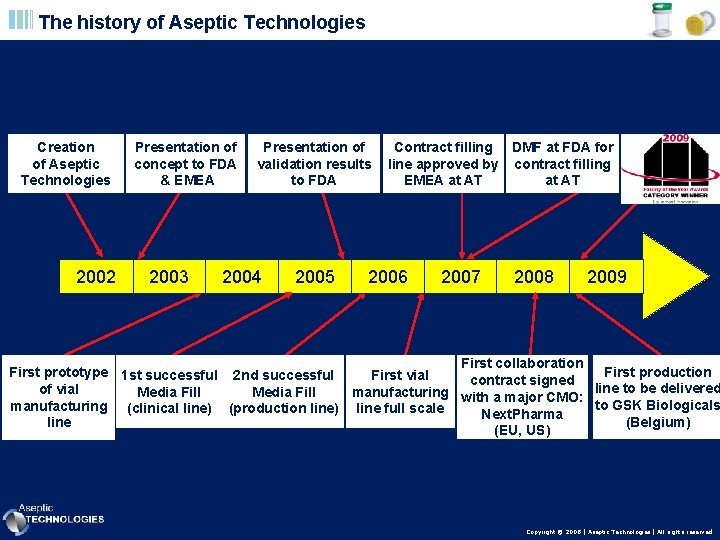
The history of Aseptic Technologies Creation of Aseptic Technologies 2002 Presentation of concept to FDA & EMEA 2003 Presentation of validation results to FDA 2004 2005 Contract filling DMF at FDA for line approved by contract filling EMEA at AT 2006 2007 2008 2009 First collaboration First production First prototype 1 st successful 2 nd successful First vial contract signed of vial Media Fill manufacturing with a major CMO: line to be delivered to GSK Biologicals manufacturing (clinical line) (production line) line full scale Next. Pharma (Belgium) line (EU, US) Copyright © 2008 | Aseptic Technologies | All rights reserved

Current status of the Crystal® Technology • Use of technology - One Production line (36, 000 vials per hour) sold to GSK Biologicals for vaccine filling - Stability tests started with two vaccines from GSK Biologicals and several products from biotechs and contract manufacturers • Vial - Vial made by injection molding (1 and 2 ml) available at industrial scale - Larger volumes (10 to 100 ml) made by injection blow molding - Design and feasibility pilot phase finalized - Small scale production pilot under development • Line - Mechanical and pharmaceutical validation performed - Media fill simulation: no contamination in more than 100, 000 vials filled • Filling capabilities - Clinical line installed in our facility - Inspected by EMEA, approval granted for stability batches - Two DMF filed at FDA (both CBER and CDER) - Several partnerships with contract manufacturer, including Next. Pharma to fill at Aseptic Technologies for partner clients - Aseptic Technologies site elected as “Facility of the Year” by ISPE for equipment innovation Copyright © 2008 | Aseptic Technologies | All rights reserved

Aseptic Technologies • Launched in June 2002 with a capital of 25 Mio EUR with the objective to develop the closed vial and its filling line, capital increased to 55 Mio EUR in 2005 • Subsidiary of Glaxo. Smith. Kline Biologicals • Current headcount 22 people • 4, 000 m² of facility of which 400 m² of clean room • Collaboration with several partners in the conception and production of vials and lines - IBA: mini e-beam - Technord: automation - Lasea: laser technology - Sirris: material studies - Rexam: vial manufacturing • Core technology licensed by Medical Instill Technologies Inc. Copyright © 2008 | Aseptic Technologies | All rights reserved

The Crystal® Closed Vial Technology • Has a proven track record of biological drug stability • Protects your products from physical and chemical damages • Reduces the risk on product loss - Residual volume - Broken vial - Batch rejection EMEA approved 2 DMF filed at FDA Filling suite in our facility to support your compatibility/stability tests in the Crystal® vials Copyright © 2008 | Aseptic Technologies | All rights reserved

Aseptic Technologies S. A. Rue Camille Hubert, 7 -9 B-5032 Gembloux Belgium www. aseptictech. com Copyright © 2008 | Aseptic Technologies | All rights reserved Benoît Verjans Commercial Director Tel: +32 81 409 4 direct Fax: +32 81 409 411 Benoit. verjans@aseptictech. com