Crystal Systems GLY 4200 Fall 2012 1 William
















![Zone Axis Diagram • [001] is the zone axis (100), (110), (010) and related Zone Axis Diagram • [001] is the zone axis (100), (110), (010) and related](https://slidetodoc.com/presentation_image/51b4899a3cc91f5c94b66309905a0913/image-27.jpg)

![Isometric [111] • {111} is equivalent to (111), , 29 Isometric [111] • {111} is equivalent to (111), , 29](https://slidetodoc.com/presentation_image/51b4899a3cc91f5c94b66309905a0913/image-29.jpg)




















- Slides: 60

Crystal Systems GLY 4200 Fall, 2012 1

William Hallowes Miller • • 1801 -1880 British Mineralogist and Crystallographer Published Crystallography in 1838 In 1839, wrote a paper, “treatise on Crystallography” in which he introduced the concept now known as the Miller Indices 2

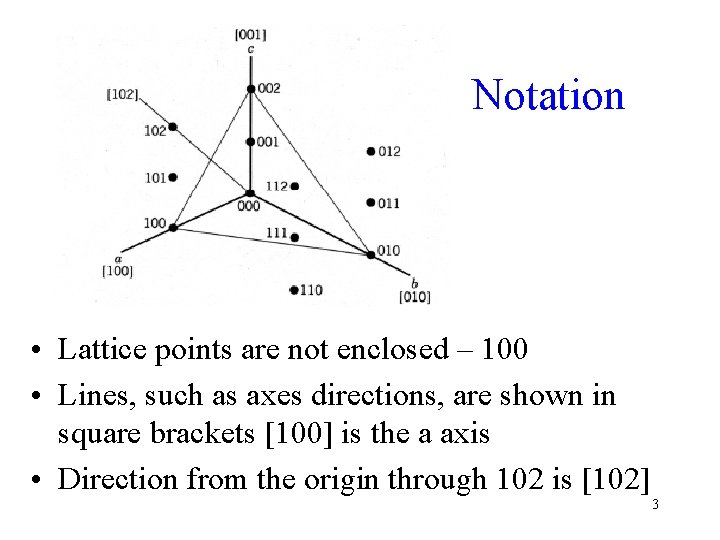
Notation • Lattice points are not enclosed – 100 • Lines, such as axes directions, are shown in square brackets [100] is the a axis • Direction from the origin through 102 is [102] 3

Miller Index • The points of intersection of a plane with the lattice axes are located • The reciprocals of these values are taken to obtain the Miller indices • The planes are then written in the form (h k l) where h = 1/a, k = 1/b and l = 1/c • Miller Indices are always enclosed in ( ) 4

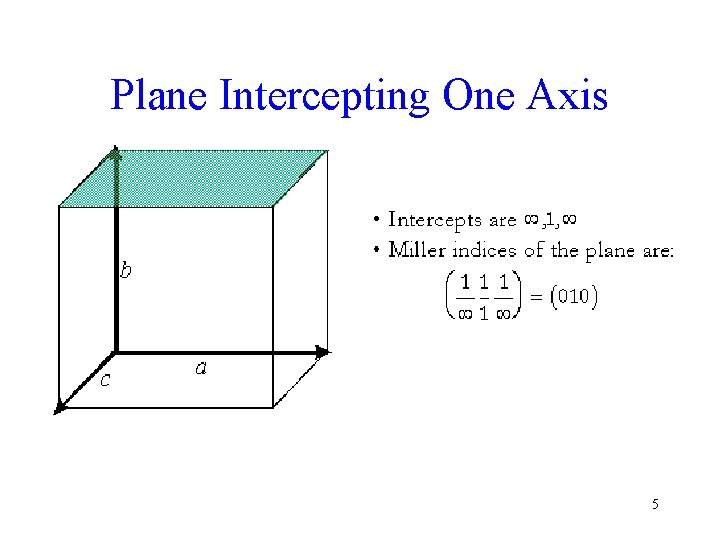
Plane Intercepting One Axis 5

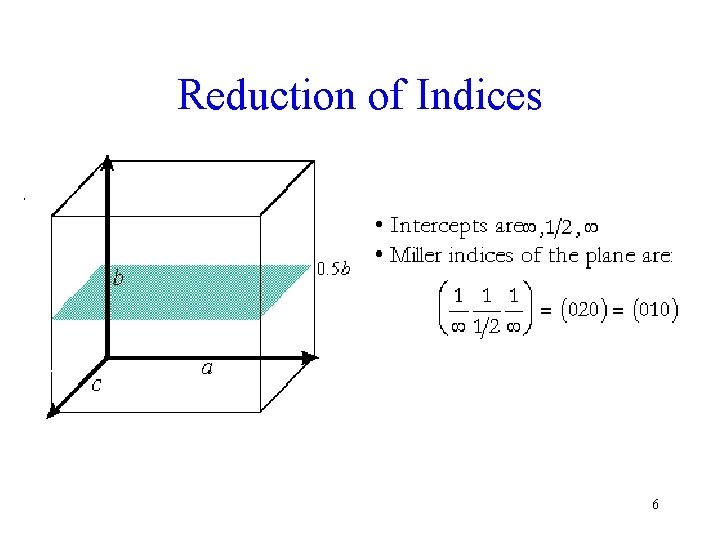
Reduction of Indices 6

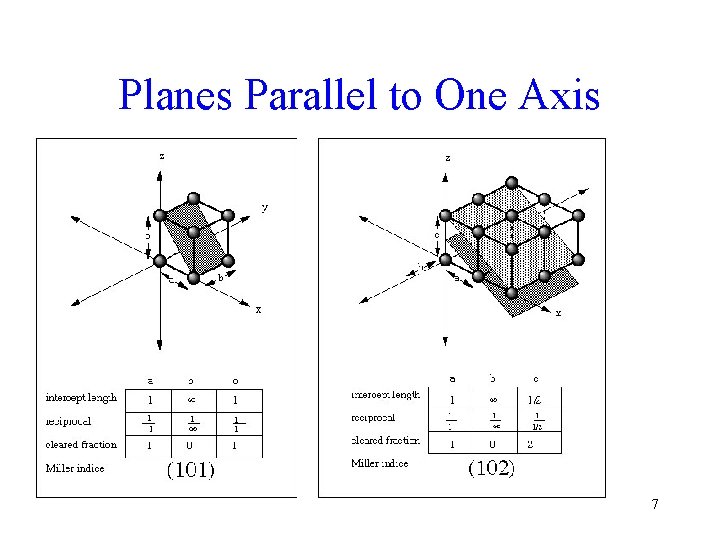
Planes Parallel to One Axis 7

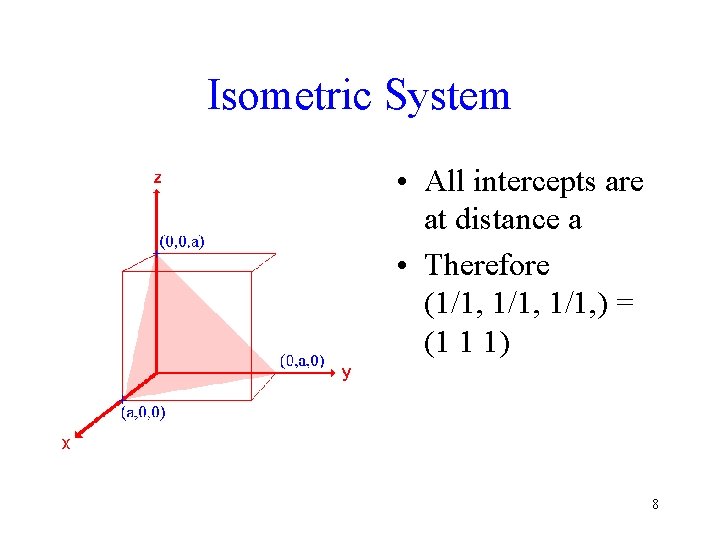
Isometric System • All intercepts are at distance a • Therefore (1/1, ) = (1 1 1) 8

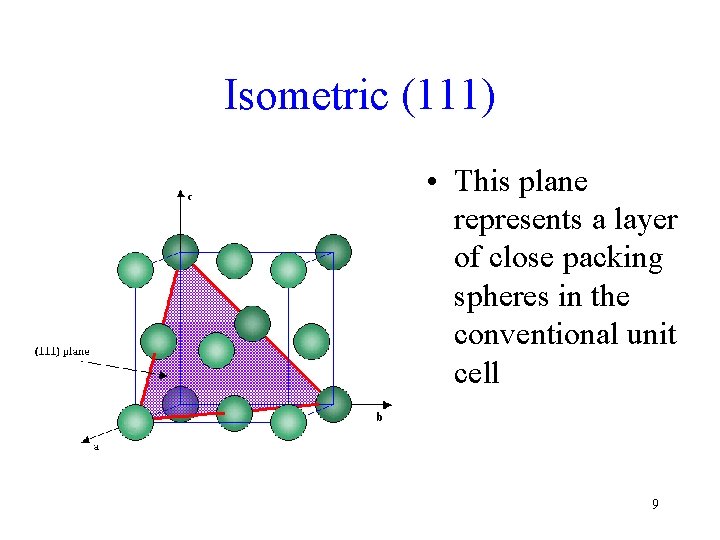
Isometric (111) • This plane represents a layer of close packing spheres in the conventional unit cell 9

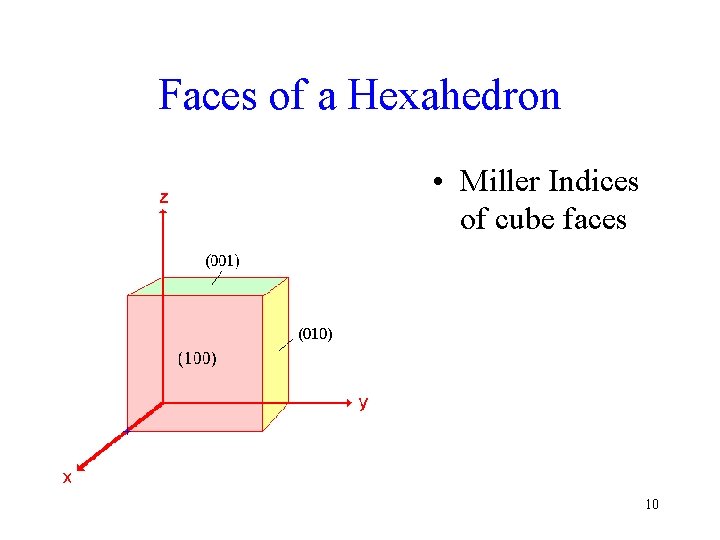
Faces of a Hexahedron • Miller Indices of cube faces 10

Faces of an Octahedron • Four of the eight faces of the octahedron 11

Faces of a Dodecahedron • Six of the twelve dodecaheral faces 12

Octahedron to Cube to Dodecahedron • Animation shows the conversion of one form to another 13

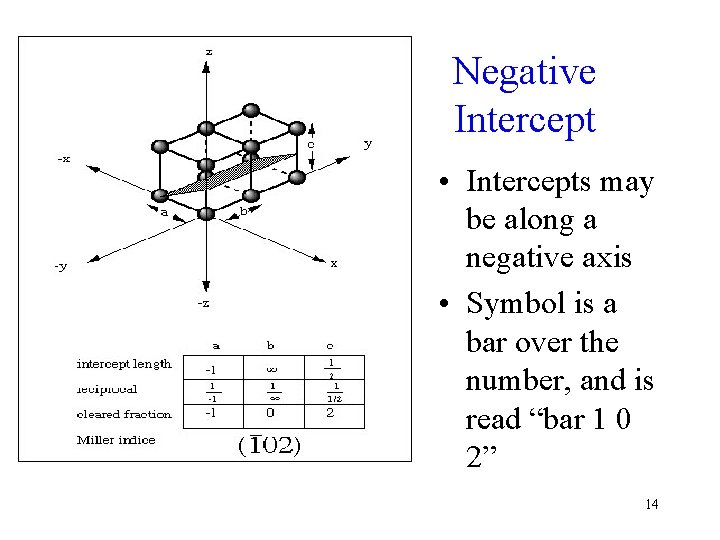
Negative Intercept • Intercepts may be along a negative axis • Symbol is a bar over the number, and is read “bar 1 0 2” 14

Miller Index from Intercepts • Let a’, b’, and c’ be the intercepts of a plane in terms of the a, b, and c vector magnitudes • Take the inverse of each intercept, then clear any fractions, and place in (hkl) format 15

Example • • • a’ = 3, b’ = 2, c’ = 4 1/3, 1/2, 1/4 Clear fractions by multiplication by twelve 4, 6, 3 Convert to (hkl) – (463) 16

Miller Index from X-ray Data • Given Halite, a = 0. 5640 nm • Given axis intercepts from X-ray data § x’ = 0. 2819 nm, y’ = 1. 128 nm, z’ = 0. 8463 nm • Calculate the intercepts in terms of the unit cell magnitude 17

Unit Cell Magnitudes • a’ = 0. 2819/0. 5640, b’ = 1. 128/0. 5640, c’ = 0. 8463/0. 5640 • a’ = 0. 4998, b’ = 2. 000, c’ = 1. 501 • Invert: 1/0. 4998, 1/2. 000, 1/1. 501 = 2, 1/2, 2/3 18

Clear Fractions • • Multiply by 6 to clear fractions 2 x 6 =12, 0. 5 x 6 = 3, 0. 6667 x 6 = 4 (12, 3, 4) Note that commas are used to separate double digit indices; otherwise, commas are not used 19

Law of Huay • Crystal faces make simple rational intercepts on crystal axes 20

Law of Bravais • Common crystal faces are parallel to lattice planes that have high lattice node density 21

Zone Axis • The intersection edge of any two non-parallel planes may be calculated from their respective Miller Indices • Crystallographic direction through the center of a crystal which is parallel to the intersection edges of the crystal faces defining the crystal zone • This is equivalent to a vector cross-product • Like vector cross-products, the order of the planes in the computation will change the result • However, since we are only interested in the 22 direction of the line, this does not matter

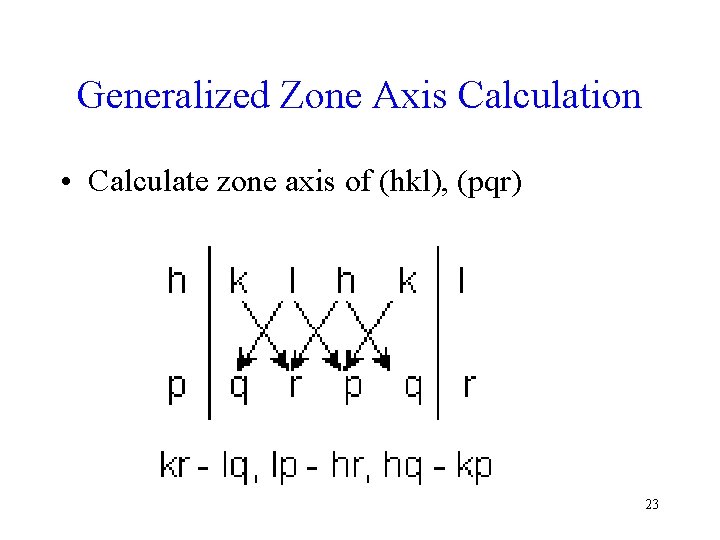
Generalized Zone Axis Calculation • Calculate zone axis of (hkl), (pqr) 23

Zone Axis Calculation • • • Given planes (120) , (201) 1│2 0 1 2│0 2│0 1 2 0│1 (2 x 1 - 0 x 0, 0 x 2 -1 x 1, 1 x 0 -2 x 2) = 2 -1 -4 The symbol for a zone axis is given as [uvw] • So, 24

Common Mistake • Zero x Anything is zero, not “Anything’ • Every year at least one student makes this mistake! 25

Zone Axis Calculation 2 • • • Given planes (201) , (120) 2│0 1 2 0│1 1│2 0 1 2│0 (0 x 0 -2 x 1, 1 x 1 -0 x 2, 2 x 2 -1 x 0) = -2 1 4 Zone axis is This is simply the same direction, in the opposite sense 26
![Zone Axis Diagram 001 is the zone axis 100 110 010 and related Zone Axis Diagram • [001] is the zone axis (100), (110), (010) and related](https://slidetodoc.com/presentation_image/51b4899a3cc91f5c94b66309905a0913/image-27.jpg)
Zone Axis Diagram • [001] is the zone axis (100), (110), (010) and related faces 27

Form • Classes of planes in a crystal which are symmetrically equivalent • Example the form {100} for a hexahedron is equivalent to the faces (100), (010), (001), , , 28
![Isometric 111 111 is equivalent to 111 29 Isometric [111] • {111} is equivalent to (111), , 29](https://slidetodoc.com/presentation_image/51b4899a3cc91f5c94b66309905a0913/image-29.jpg)
Isometric [111] • {111} is equivalent to (111), , 29

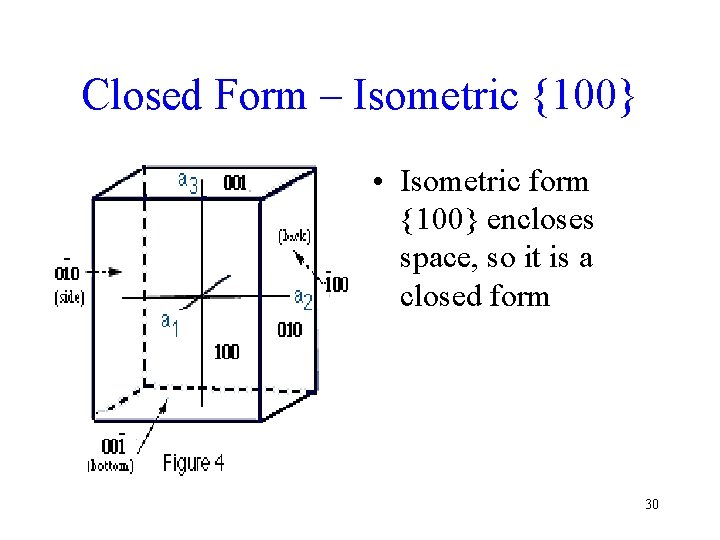
Closed Form – Isometric {100} • Isometric form {100} encloses space, so it is a closed form 30

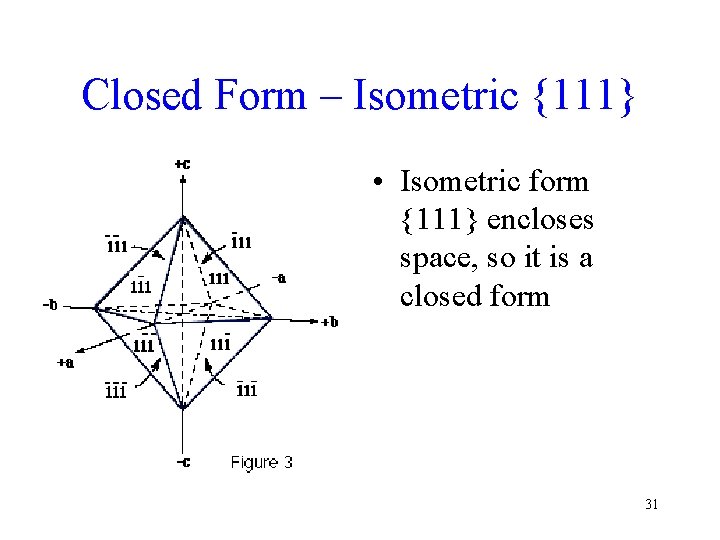
Closed Form – Isometric {111} • Isometric form {111} encloses space, so it is a closed form 31

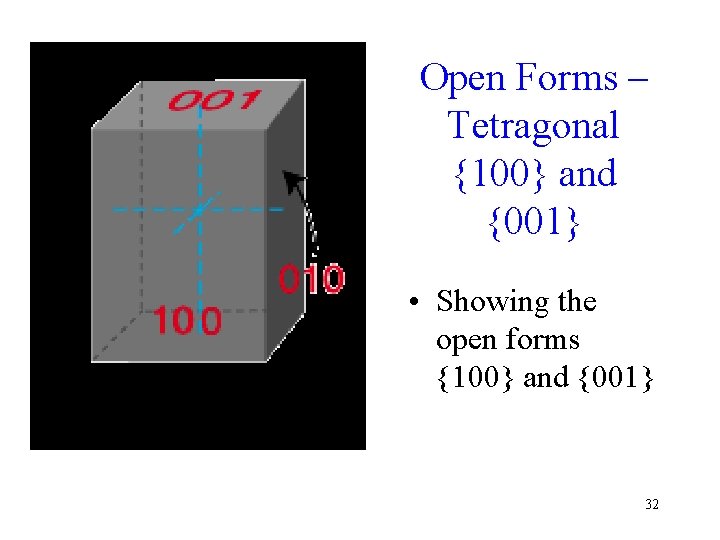
Open Forms – Tetragonal {100} and {001} • Showing the open forms {100} and {001} 32

Pedion • Open form consisting of a single face 33

Pinacoid • Open form consisting of two parallel planes • Platy specimen of wulfenite – the faces of the plates are a pinacoid 34

Benitoite • The mineral benitoite has a set of two triangular faces which form a basal pinacoid 35

Dihedron • Pair of intersecting faces related by mirror plane or twofold symmetry axis § Sphenoids - Pair of intersecting faces related by two-fold symmetry axis § Dome - Pair of intersecting faces related by mirror plane 36

Dome • Open form consisting of two intersecting planes, related by mirror symmetry • Very large gem golden topaz crystal is from Brazil and measures about 45 cm in height • Large face on right is part of a dome 37

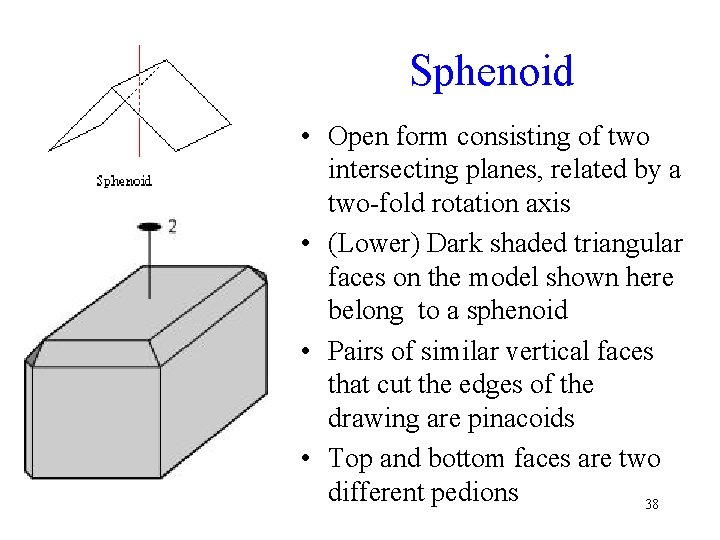
Sphenoid • Open form consisting of two intersecting planes, related by a two-fold rotation axis • (Lower) Dark shaded triangular faces on the model shown here belong to a sphenoid • Pairs of similar vertical faces that cut the edges of the drawing are pinacoids • Top and bottom faces are two different pedions 38

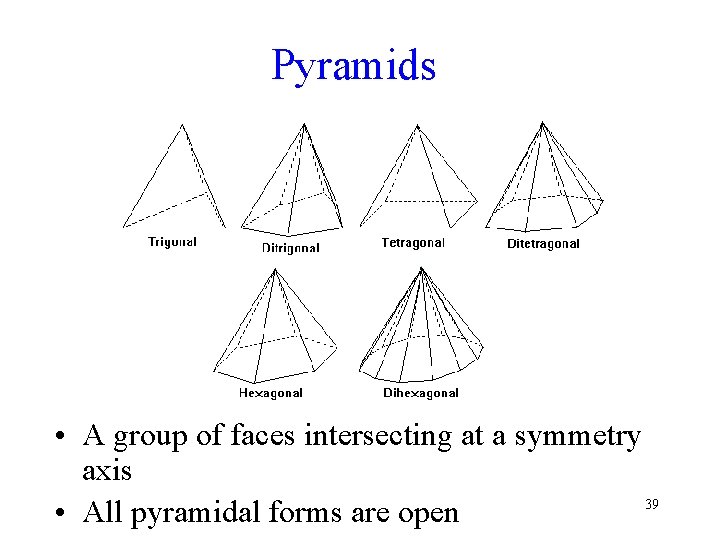
Pyramids • A group of faces intersecting at a symmetry axis 39 • All pyramidal forms are open

Apophyllite Pyramid • Pyramid measures 4. 45 centimeters tall by 5. 1 centimeters wide at its base 40

Uvite • Three-sided pyramid of the mineral uvite, a type of tourmaline 41

Prisms • A prism is a set of faces that run parallel to an axes in the crystal • There can be three, four, six, eight or even twelve faces • All prismatic forms are open 42

Diprismatic Forms • Upper – Trigonal prism • Lower – Ditrigonal prism – note that the vertical axis is an A 3, not an A 6 43

Citrine Quartz • The six vertical planes are a prismatic form • This is a rare doubly terminated crystal of citrine, a variety of quartz 44

Vanadinite • Forms hexagonal prismatic crystals 45

Galena • Galena is isometric, and often forms cubic to rectangular crystals • Since all faces of the form {100} are equivalent, this is a closed form 46

Fluorite • Image shows the isometric {111} form combined with isometric {100} • Either of these would be closed forms if uncombined 47

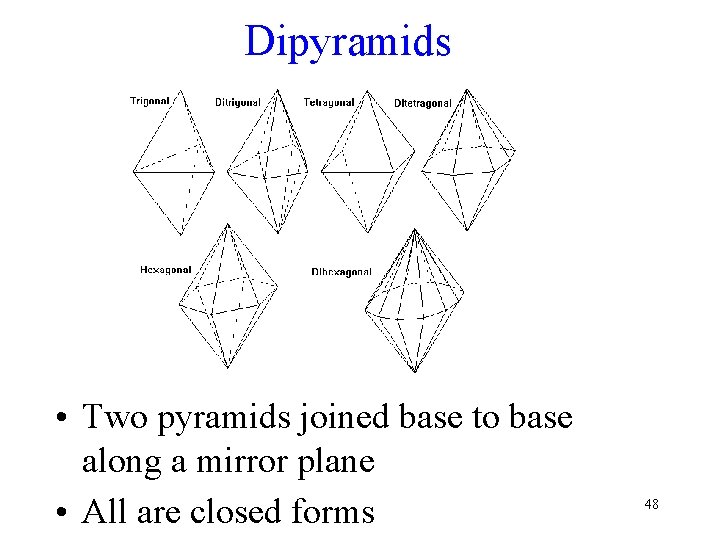
Dipyramids • Two pyramids joined base to base along a mirror plane • All are closed forms 48

Hanksite • Tetragonal dipyramid 49

Disphenoid • A solid with four congruent triangle faces, like a distorted tetrahedron • Midpoints of edges are twofold symmetry axes • In the tetragonal disphenoid, the faces are isosceles triangles and a fourfold inversion axis joins the midpoints of the bases of the 50 isosceles triangles.

Dodecahedrons • A closed 12 -faced form • Dodecahedrons can be formed by cutting off the edges of a cube • Form symbol for a dodecahedron is isometric{110} • Garnets often display this form 51

Tetrahedron • The tetrahedron occurs in the class bar 4 3 m and has the form symbol {111}(the form shown in the drawing) or {1 bar 11} • It is a four faced form that results form three bar 4 axes and four 3 -fold axes • Tetrahedrite, a copper sulfide mineral 52

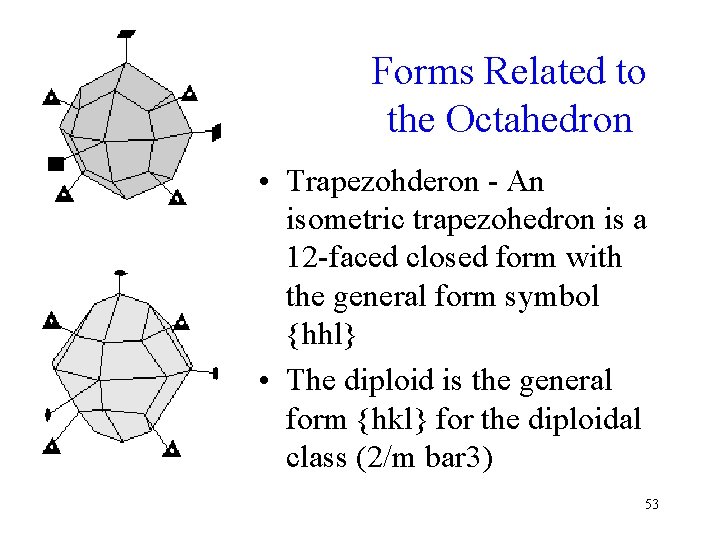
Forms Related to the Octahedron • Trapezohderon - An isometric trapezohedron is a 12 -faced closed form with the general form symbol {hhl} • The diploid is the general form {hkl} for the diploidal class (2/m bar 3) 53

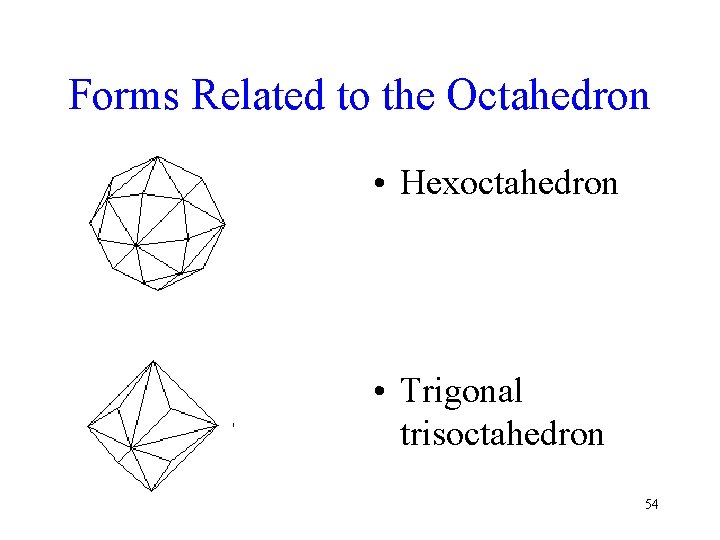
Forms Related to the Octahedron • Hexoctahedron • Trigonal trisoctahedron 54

Pyritohedron • The pyritohedron is a 12 faced form that occurs in the crystal class 2/m bar 3 • The possible forms are {h 0 l} or {0 kl} and each of the faces that make up the form have 5 sides 55

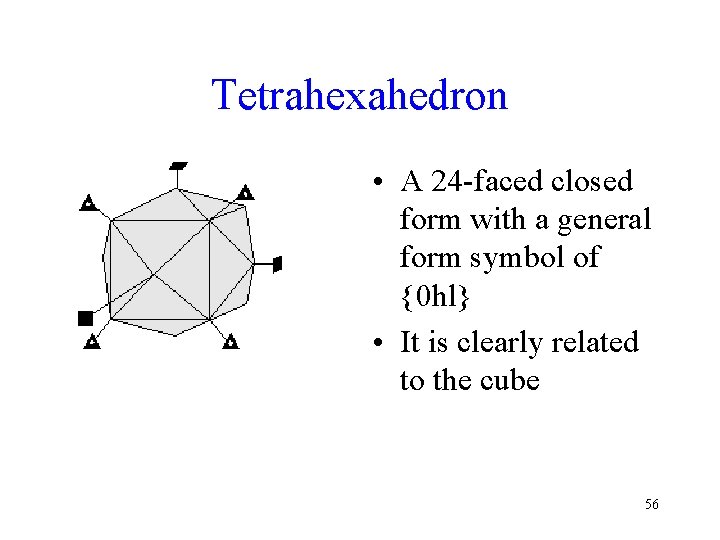
Tetrahexahedron • A 24 -faced closed form with a general form symbol of {0 hl} • It is clearly related to the cube 56

Scalenohedron • A scalenohedron is a closed form with 8 or 12 faces • In ideally developed faces each of the faces is a scalene triangle • In the model, note the presence of the 3 -fold rotoinversion axis perpendicular to the 3 2 -fold axes 57

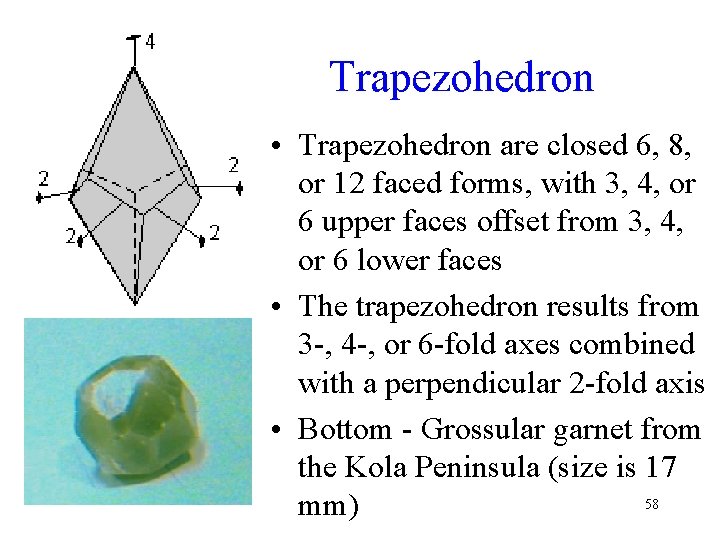
Trapezohedron • Trapezohedron are closed 6, 8, or 12 faced forms, with 3, 4, or 6 upper faces offset from 3, 4, or 6 lower faces • The trapezohedron results from 3 -, 4 -, or 6 -fold axes combined with a perpendicular 2 -fold axis • Bottom - Grossular garnet from the Kola Peninsula (size is 17 58 mm)

Rhombohedron • A rhombohedron is 6 -faced closed form wherein 3 faces on top are offset by 3 identical upside down faces on the bottom, as a result of a 3 -fold rotoinversion axis • Rhombohedrons can also result from a 3 -fold axis with perpendicular 2 -fold axes • Rhombohedrons only occur in the crystal classes bar 3 2/m , 32, and bar 3. 59

Application to the Core • From EOS, v. 90, #3, 1/20/09 60