CRYSTAL STURCTURES CRYSTAL STRUCTURE CAN BE OBTAINED BY

CRYSTAL STURCTUR-ES

CRYSTAL STRUCTURE CAN BE OBTAINED BY ATTACHING ATOMS, GROUP OF ATOMS OR MOLECULES, WHICH ARE CALLED BASIS TO LATTICE SIDES OF THE LATTICE POINTS Crystal=crystal lattice+basis structure
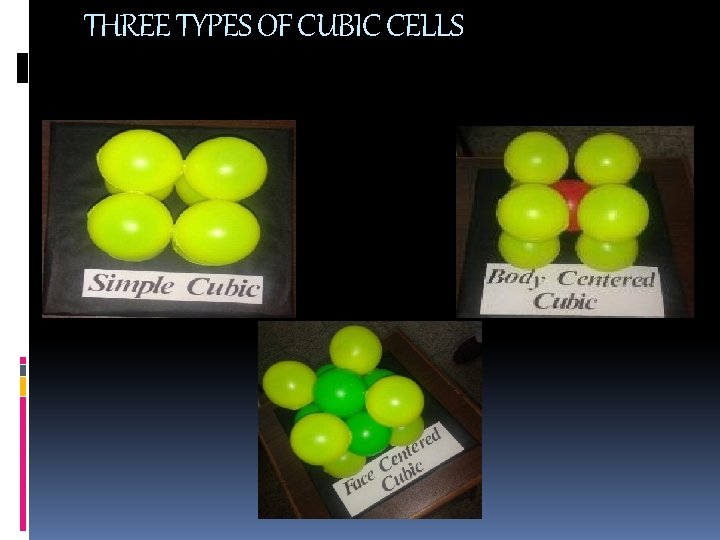
THREE TYPES OF CUBIC CELLS
![• Simple cubic [sc] simple cubic has one primitive point so its primitive • Simple cubic [sc] simple cubic has one primitive point so its primitive](http://slidetodoc.com/presentation_image/4be92c0d7a4f8a30c23e0a311ee3592a/image-4.jpg)
• Simple cubic [sc] simple cubic has one primitive point so its primitive cell. In the unit cell on the left, the atom at the corners are cut because only a portion (1/8) belongs to that cell. the rest belongs to neighboring cells. The co-ordination number of sc is 6

Atomic radius for sc it is half the distance between any two nearest neighbour in the given structure it is expressed in terms of cube edge a a=2 r r=a/2

Atomic packing fraction of simple cubic APF=0. 52 that means that the pecentage of packing is 52% thus, 52% of the volume of the simple cubic unit cell is occupied by atoms and the remaining 48% volume of unit cell isvacant or void space
![BODY CENTERED CUBIC [BCC] BCC STRUCTURE HAS 8 CORNERS ATOMS AND 1 BODY CENTRE BODY CENTERED CUBIC [BCC] BCC STRUCTURE HAS 8 CORNERS ATOMS AND 1 BODY CENTRE](http://slidetodoc.com/presentation_image/4be92c0d7a4f8a30c23e0a311ee3592a/image-7.jpg)
BODY CENTERED CUBIC [BCC] BCC STRUCTURE HAS 8 CORNERS ATOMS AND 1 BODY CENTRE ATOM. EACH CORNER ATOM IS SHARED BY 8 UNIT CELLS. THE CENTER ATOM IS NOT SHARED BY ANY OF THE UNIT CELL. SO THE, NO. OF ATOMS PER UNIT CELL N=1/8 X 8+1 =2

BCC HAS TWO LATTICE POINT SO BCC IS A NONPRIMITIVE CELL. BCC HAS EIGHT NEAREST NEIGHBOURS. EACH ATOMS IS IN CONTACT WITH ITS NEIGHBOURS ONLY ALONG THE BODYDAIGONAL DIRECTIONS. HENCE, THE CO-ORDINATION NO. FOR BCC UNIT CELL IS 8.

ATOMIC PACKING FRACTION OF BCC APF=0. 68 THE PERCENTAGE OF PACAPKING FOR BCC STRUCTURE IS 68% THUS, 68% OF THE VOLUME OF BODY CENTERED CUBIC CELL IS OCCUPIED BY ATOMS AND THE REMAINING 32% OF THE VOLUME IS VACANT OR VOID SPACE

ATOMIC RADIUS FOR BCC UNIT CELL

FACE CENTERED STRUCTURE fcc structure has 8 corner atoms and 6 face centeratoms. Each corner atom is shared by 8 unit cell Each face centered atom is shared by 2 unit cells. Number of atoms present in unit cell is n=(1/8 x 8)+(1/2 x 6) =1+3 =4

CO -ORDINATION NUMBER THE CORNER ATOM IN ITS OWN PLANE TOUCHES 4 FACE CENTERED ATOMS. IN THE PLANE JUST ABOVE , THE CORNER ATOM HAS ANOTHER 4 FACE CENTERED ATOMS AS ITS NEAREST NEIGHBOURS. SIMILARLY, IN THE PLANE JUST BELOW IT HAS 4 MORE FACE CENTERED ATOMS AS AS ITS NEAREST NEIGHBOUR THEREFORE THE NO. OF NEAREST NEIGHBOUR ARE: 4+4+4=12

ATOMIC RADIUS FOR FCC

Atomic paking fraction of fcc APF=O. 74 THE PERCENTAGE OF PACKING FOR FCC STRUCTURE IS 74%. THUS, 74% OF THE VOLUME OF FACE CENTERED CUBIC CELL IS OCCUPIED BY ATOMS AND THE REMAINING 26% OF THE VOLUME IS VACANT OR VOID.

Made by: - GROUP NO: -2 GROUP MEMBERS: 1. NEELAM. CHOUDHARY 2. TUBA. ANSARI 3. NITHU. NADAR 4. RASHIDA. ALI 5. MONIKA. SAHARE 6. MAYURI 7. RUCHIKA 8. PRAJAKTA
- Slides: 15