Crystal Formation Crystals are formed from saturated solutions























- Slides: 26


Crystal Formation Crystals are formed from saturated solutions. Saturated solutions are created when a soluble solid is stirred into water until the water cannot dissolve anymore of that substance at that temperature.

Crystal Formation When the solution is cooled , the water holds less dissolved solute , so some of the substance will come out of the solution. If the particles fit together in a crystalline structure , a crystal will form.

Crystal Formation Crystal shape and color is unique for different substances. This allows us to identify different substances easily.

Crystal Formation Large crystals form when a hot saturated solution is slowly cooled. Small crystals form when a hot saturated solution is quickly cooled.

Crystal Formation Some examples of crystals we see daily are:

Crystal Formation Some examples of crystals we see daily are: Quartz batteries in watches LCD TV’s – Liquid Crystal Display

Physical and Chemical Changes Physical changes are changes in states of matter. These changes are reversible because the substance can go back to its original form. Example:

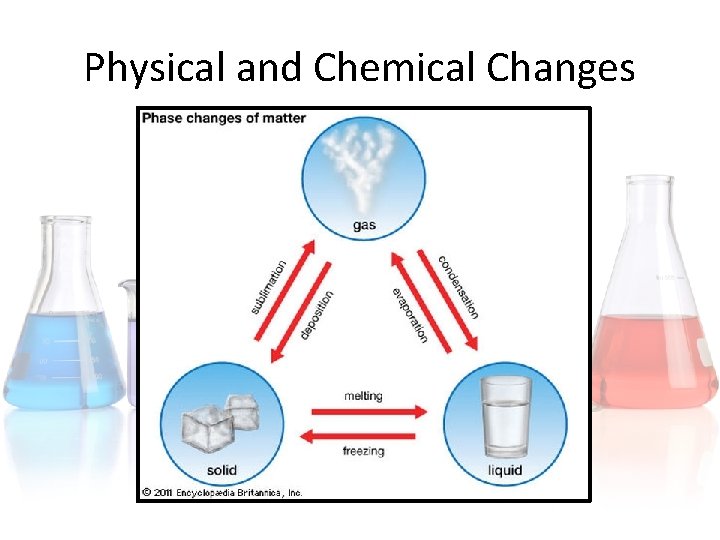
Physical and Chemical Changes in states of matter can be described in the following chart. Note: you do not need to know what sublimation and deposition are.

Physical and Chemical Changes

Physical and Chemical Changes https: //www. youtube. com/watch? v=M 8 tyjw. B 42 X 4 &app=desktop Chemical changes are changes in the molecule structure of matter. They are said to be irreversible because it is nearly impossible to return to its original state. Example:

Physical and Chemical Changes in states of matter are easy to see. Every year, we see snow (solid) melt in spring. Chemical changes are sometimes not as easy to identify. Luckily, there are some ways we can tell chemical change happened.

Physical and Chemical Changes Evidence of Chemical Change We know chemical change has happened when we see one or more of these four things: 1. Color change Examples: - Mixing food coloring - Mixing milk into coffee

Physical and Chemical Changes Evidence of Chemical Change 2. Bubbles Examples: - Mixing vinegar and baking soda - Mixing Mentos candies And Coke

Physical and Chemical Changes Evidence of Chemical Change 3. Energy Released Examples: - Explosions - Fire - Bright light - Extreme heat

Physical and Chemical Changes Evidence of Chemical Change 4. Precipitate A precipitate is a solid that gets left behind after a chemical reaction. Examples: - Starch and Iodine

Physical and Chemical Changes Evidence of Chemical Change Other examples of chemical change: Rust Burning Wood

Physical and Chemical Changes Evidence of Chemical Change Other examples of chemical change: Browning Bananas Digesting Food

Physical and Chemical Changes • studyjams. scholastic. com/studyjams/sci ence/matter/changes-of-matter. htm

Acids and Bases

Acids and Bases • Acids and bases are two special kinds of chemicals. • Almost all liquids are either acids or bases. • Alkaline is another word for base.

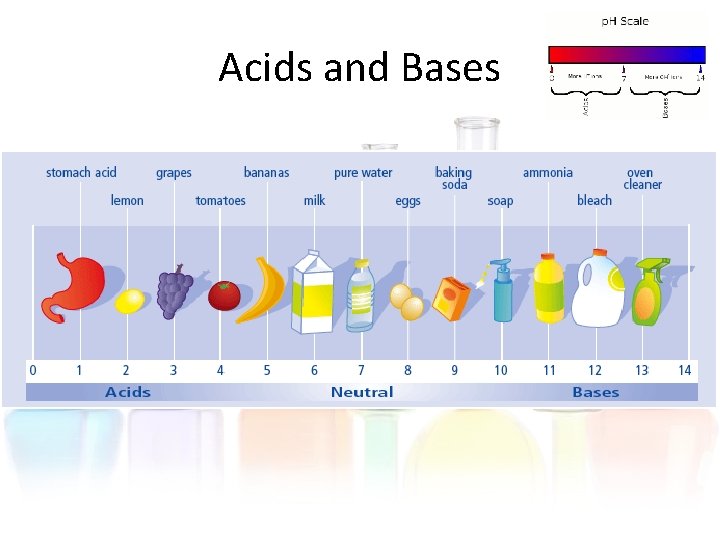
Acids and Bases • Whether a liquid is an acid or base depends on the type of ions in it. • If it has a lot of hydrogen ions, then it is an acid. • If it has a lot of hydroxide ions, then it is a base.

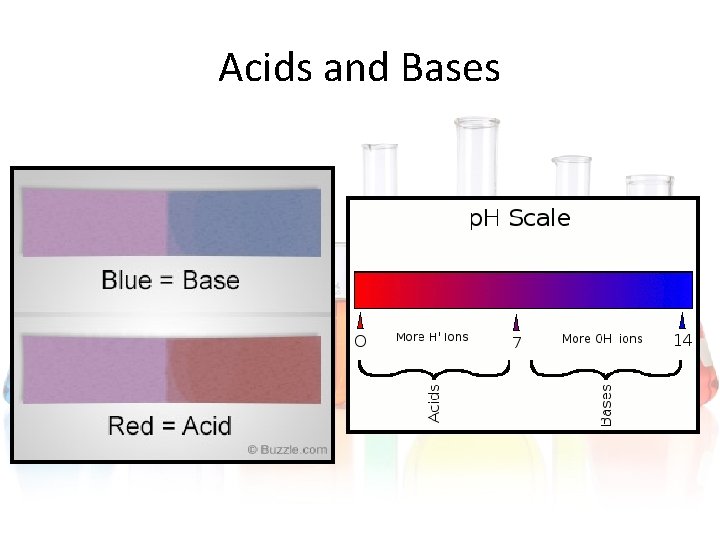
Acids and Bases • We measure the amount of hydroxide or hydrogen ions with the p. H scale. • The p. H scale ranges from 0 to 14. • Acids have a p. H of 0 to 7. • Bases have a p. H of 7 to 14.

Acids and Bases • To test where a substance is on the p. H scale, we use litmus tests. • Litmus tests are pieces of paper that change color to show if the substance is an acid or base. • Litmus paper turns blue if it’s a base. • Litmus paper turns red if it’s an acid.

Acids and Bases

Acids and Bases