Crystal Field Theory Electronic Spectra and MO of




![Let’s Look at 4 Co 3+ complexes: Config. [Co(NH 3)6]3+ [Co(NH 3)5(OH 2 Color Let’s Look at 4 Co 3+ complexes: Config. [Co(NH 3)6]3+ [Co(NH 3)5(OH 2 Color](https://slidetodoc.com/presentation_image/275cf8853fdaa6ea188dac1d1b9f93f9/image-9.jpg)



![[Fe(H 2 O)6]3+ [Ni(H 2 O)6]2+ [Co(H 2 O)6]2+ [Zn(H 2 O)6]2+ [Cu(H 2 [Fe(H 2 O)6]3+ [Ni(H 2 O)6]2+ [Co(H 2 O)6]2+ [Zn(H 2 O)6]2+ [Cu(H 2](https://slidetodoc.com/presentation_image/275cf8853fdaa6ea188dac1d1b9f93f9/image-14.jpg)





- Slides: 30

Crystal Field Theory, Electronic Spectra and MO of Coordination Complexes Or why I decided to become an inorganic chemist or Ohhh!!! The Colors!!!



Gemstone owe their color from trace transition-metal ions • Corundum mineral, Al 2 O 3: Colorless • Cr Al : Ruby • Mn Al: Amethyst • Fe Al: Topaz • Ti &Co Al: Sapphire • Beryl mineral, Be 3 Al 2 Si 6 O 18: Colorless • Cr Al : Emerald • Fe Al : Aquamarine



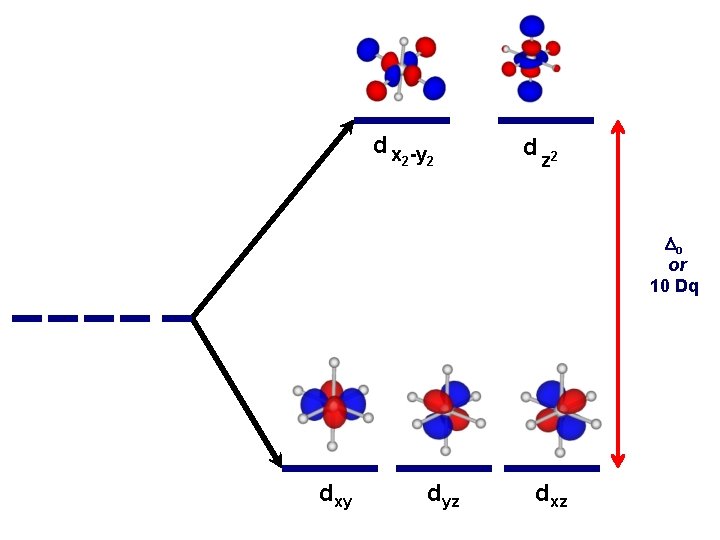
d x -y 2 2 d z 2 Do or 10 Dq dxy dyz dxz

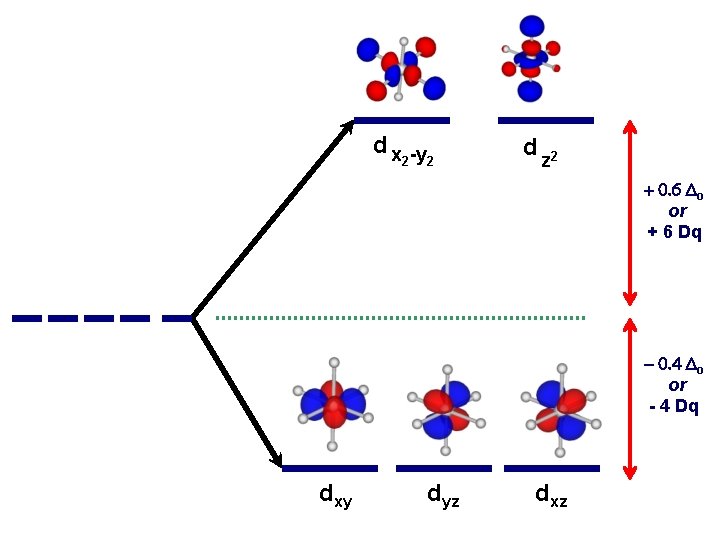
d x -y 2 2 d z 2 + 0. 6 Do or + 6 Dq - 0. 4 Do or - 4 Dq dxy dyz dxz
![Lets Look at 4 Co 3 complexes Config CoNH 363 CoNH 35OH 2 Color Let’s Look at 4 Co 3+ complexes: Config. [Co(NH 3)6]3+ [Co(NH 3)5(OH 2 Color](https://slidetodoc.com/presentation_image/275cf8853fdaa6ea188dac1d1b9f93f9/image-9.jpg)
Let’s Look at 4 Co 3+ complexes: Config. [Co(NH 3)6]3+ [Co(NH 3)5(OH 2 Color of Complex Absorbs d 6 )]3+ [Co(NH 3)5 Br]2+ d 6 [Co(NH 3)5 Cl]2+ d 6 350 -400 400 -500 Values are in nm Greater d 6 520 -570 Splitting 600 -700 600 -650 570 -600

OTHER QUESTIONS So there are two ways to put the electrons Low Spin High Spin Which form for our 4 cobalt(III) complexes? And why the difference between Cl- and Br-?

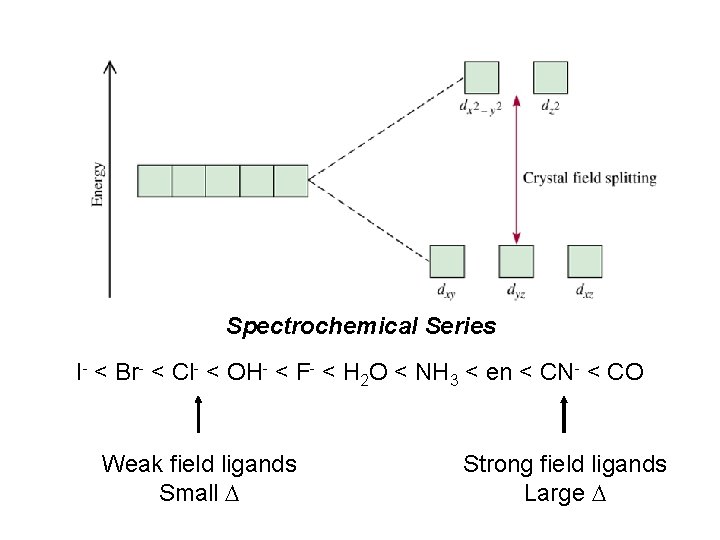
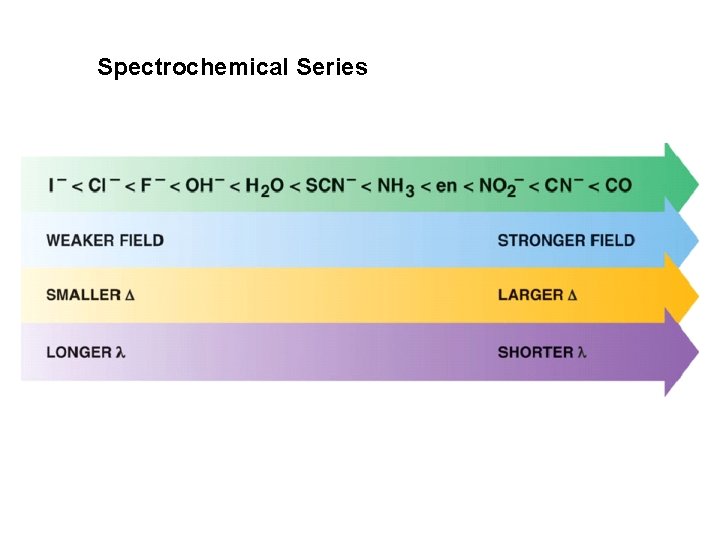
R. Tsuchida (1938) noticed a trend in while looking at a series of Cobalt(III) Complexes. With the general formula : [Co(NH 3)5 X] look at that! The same ones we just looked at…. He arrived a series which illustrates the effect of ligands on Do (10 Dq) He called it: The Spectrochemical Series Tsuchida, R. Bull. Chem. Soc. Jpn. 1938, 13, 388

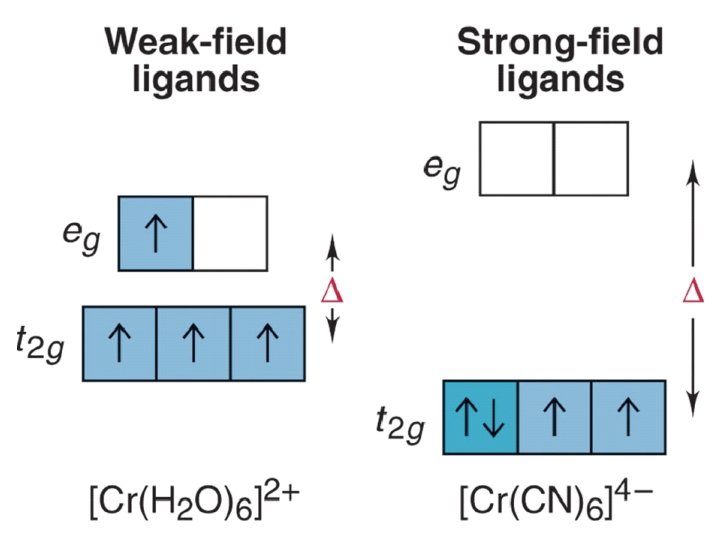
The Spectrochemical Series Ligand effect on Do : Small Do I- < Br- < S 2 - < Cl- < NO 3 - < F- < OH- < H 2 O < CH 3 CN < NH 3 < en < bpy < phen < NO 2 - < PPh 3 < CN- < CO Large Do Or more simply : X<O<N<C Metals also effect Do : Mn 2+ < Ni 2+ < Co 2+ < Fe 2+ < V 2+ < Fe 3+ < Co 3+ < Mn 4+ < Mo 3+ < Rh 3+ < Ru 3+ < Pd 2+ < Ir 3+ < Pt 2+ Fe 3+ << Ru 3+ Ni 2+ << Pd 2+ Important consequences result!!!

Spectrochemical Series I- < Br- < Cl- < OH- < F- < H 2 O < NH 3 < en < CN- < CO Weak field ligands Small D Strong field ligands Large D
![FeH 2 O63 NiH 2 O62 CoH 2 O62 ZnH 2 O62 CuH 2 [Fe(H 2 O)6]3+ [Ni(H 2 O)6]2+ [Co(H 2 O)6]2+ [Zn(H 2 O)6]2+ [Cu(H 2](https://slidetodoc.com/presentation_image/275cf8853fdaa6ea188dac1d1b9f93f9/image-14.jpg)
[Fe(H 2 O)6]3+ [Ni(H 2 O)6]2+ [Co(H 2 O)6]2+ [Zn(H 2 O)6]2+ [Cu(H 2 O)6]2+

S=5/2 S=1/2

S=1 S=2

Spectrochemical Series

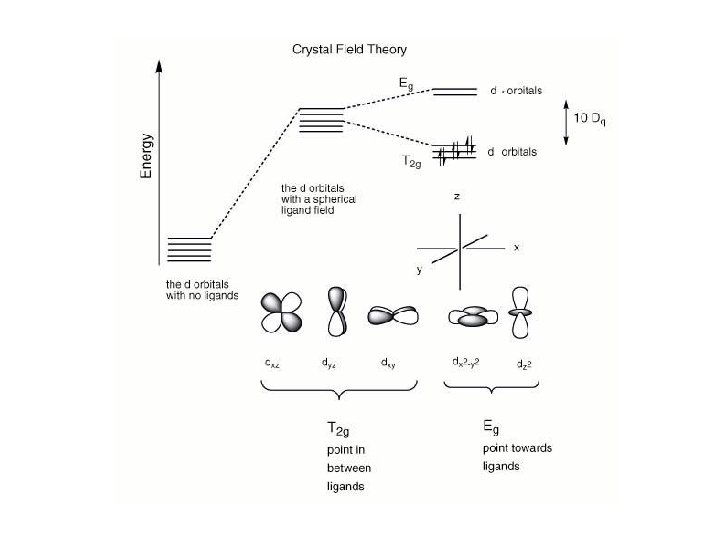
Another important question arises: How does filling electrons into orbitals effect the stability (energy) of the d-orbitals relative to a spherical environment where they are degenerate? We use something called Crystal Field Stabilization Energy (CFSE) to answer these questions For a t 2 gx egy configuration : CFSE = (-0. 4 · x + 0. 6 · y)Do

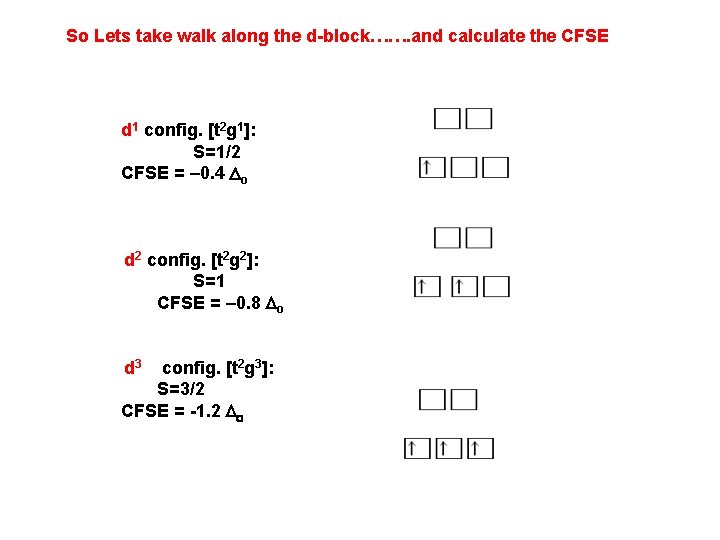
So Lets take walk along the d-block……. and calculate the CFSE d 1 config. [t 2 g 1]: S=1/2 CFSE = – 0. 4 Do d 2 config. [t 2 g 2]: S=1 CFSE = – 0. 8 Do d 3 config. [t 2 g 3]: S=3/2 CFSE = -1. 2 Do

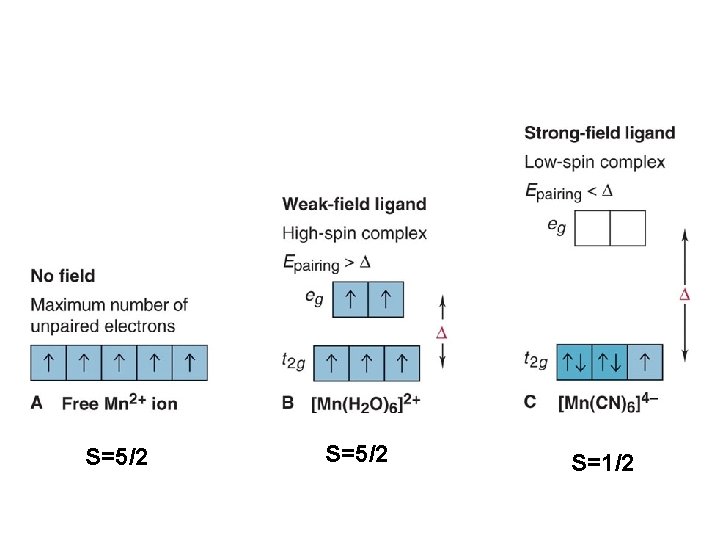
BUT WHEN YOU GET TO: d 4 THERE ARE TWO OPTIONS!!!!! Low Spin High Spin CFSE = -1. 6 Do + P CFSE = -0. 6 Do When is one preferred over the other ? ? ? It depends. (P 14, 900 cm-1 / e- pair) Do P = Do both are equally stabilized P > Do high spin (weak field) stabilized NOTE: the text uses the symbol P, for spin pairing energy P< low spin (weak field) stabilized

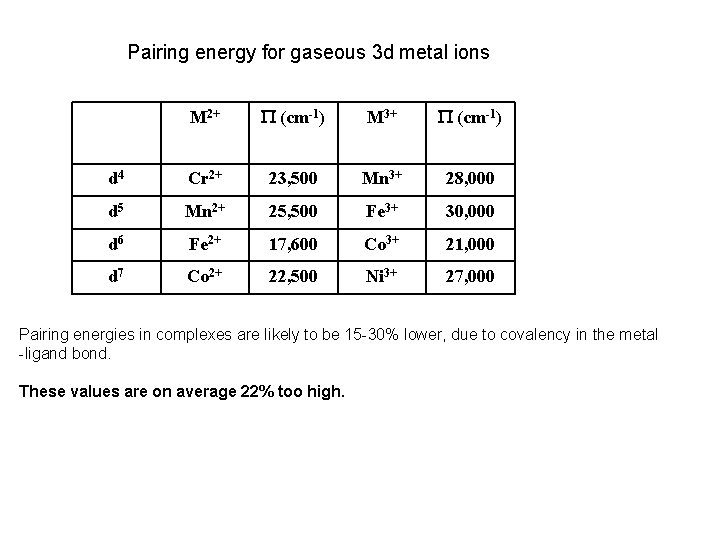
P , Spin Pairing Energy is composed of two terms (a) The coulombic repulsion – This repulsion must be overcome when forcing electrons to occupy the same orbital. As 5 -d orbitals are more diffuse than 4 -d orbitals which are more diffuse than 3 -d orbitals, the pairing energy becomes smaller as you go down a period. As a rule 4 d and 5 d transition metal complexes are generally low spin! (b) The loss of exchange energy – The exchange energy (Hünd’s Rule) is proportional to the number of electrons having parallel spins. The greater this number, the more difficult it becomes to pair electrons. Therefore, d 5 (Fe 3+ , Mn 2+) configurations are most likely to form high spin complexes.

Pairing energy for gaseous 3 d metal ions M 2+ P (cm-1) M 3+ P (cm-1) d 4 Cr 2+ 23, 500 Mn 3+ 28, 000 d 5 Mn 2+ 25, 500 Fe 3+ 30, 000 d 6 Fe 2+ 17, 600 Co 3+ 21, 000 d 7 Co 2+ 22, 500 Ni 3+ 27, 000 Pairing energies in complexes are likely to be 15 -30% lower, due to covalency in the metal -ligand bond. These values are on average 22% too high.


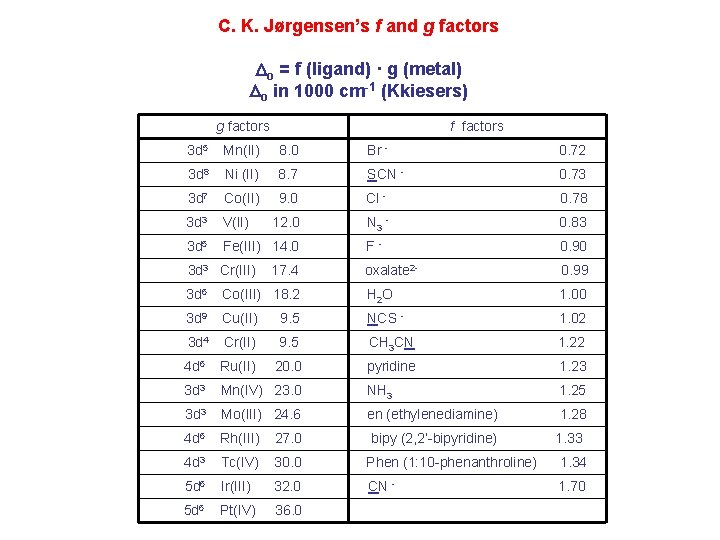
C. K. Jørgensen’s f and g factors Do = f (ligand) · g (metal) Do in 1000 cm-1 (Kkiesers) g factors f factors 3 d 5 Mn(II) 8. 0 Br - 0. 72 3 d 8 Ni (II) 8. 7 SCN - 0. 73 3 d 7 Co(II) 9. 0 Cl - 0. 78 3 d 3 V(II) 12. 0 N 3 - 0. 83 3 d 5 Fe(III) 14. 0 F- 0. 90 oxalate 2 - 0. 99 3 d 3 Cr(III) 17. 4 3 d 6 Co(III) 18. 2 H 2 O 1. 00 3 d 9 Cu(II) 9. 5 NCS - 1. 02 3 d 4 Cr(II) 9. 5 CH 3 CN 1. 22 4 d 6 Ru(II) 20. 0 pyridine 1. 23 3 d 3 Mn(IV) 23. 0 NH 3 1. 25 3 d 3 Mo(III) 24. 6 en (ethylenediamine) 1. 28 4 d 6 Rh(III) 27. 0 4 d 3 Tc(IV) 30. 0 Phen (1: 10 -phenanthroline) 1. 34 5 d 6 Ir(III) 32. 0 CN - 1. 70 5 d 6 Pt(IV) 36. 0 bipy (2, 2’-bipyridine) 1. 33

Note: Rh 3+ and Ir 3+ are a lot different than Co 3+ g 3 d < g 4 d ≤ g 5 d EXAMPLE: Calculate the Do (10 Dq) for [Rh(OH 2)6]3+ in cm-1 and nm. for [Rh(pyr)3 Cl 3]

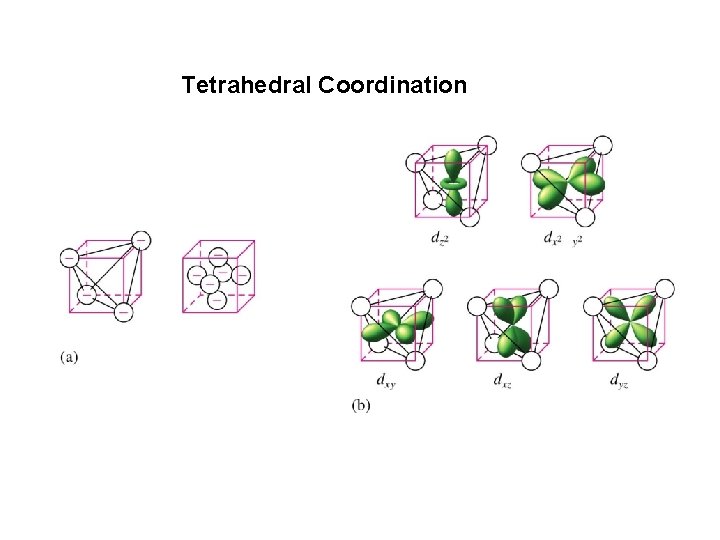
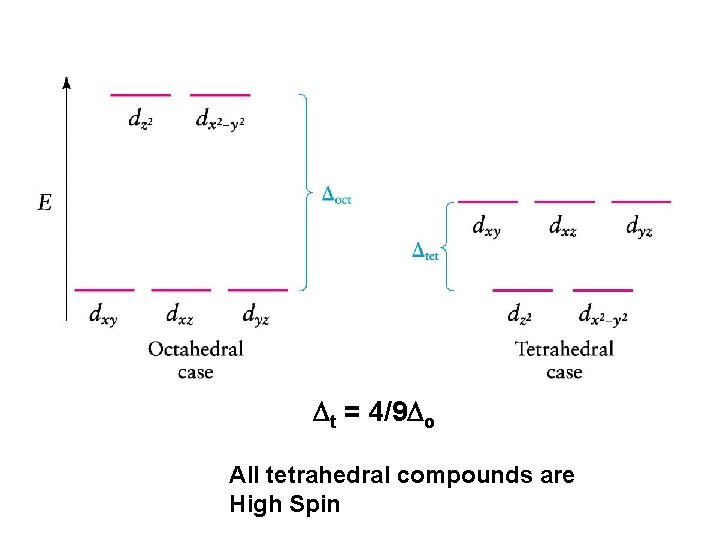
Tetrahedral Coordination

Dt = 4/9 Do All tetrahedral compounds are High Spin

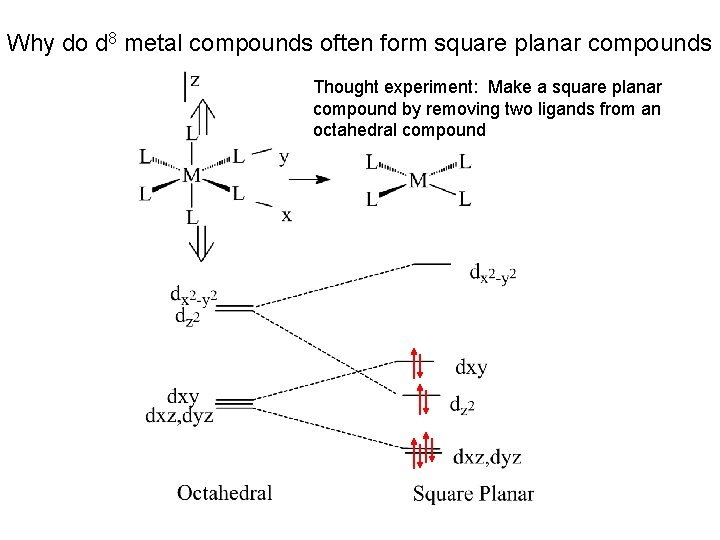
Why do d 8 metal compounds often form square planar compounds Thought experiment: Make a square planar compound by removing two ligands from an octahedral compound

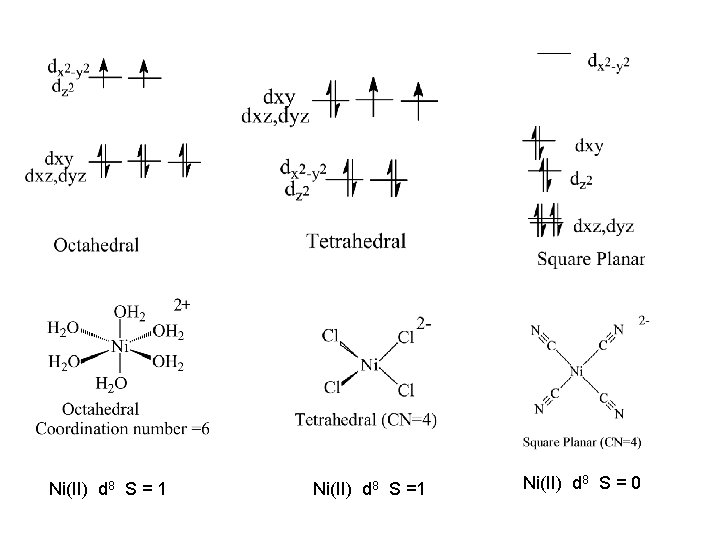
Ni(II) d 8 S = 1 Ni(II) d 8 S = 0

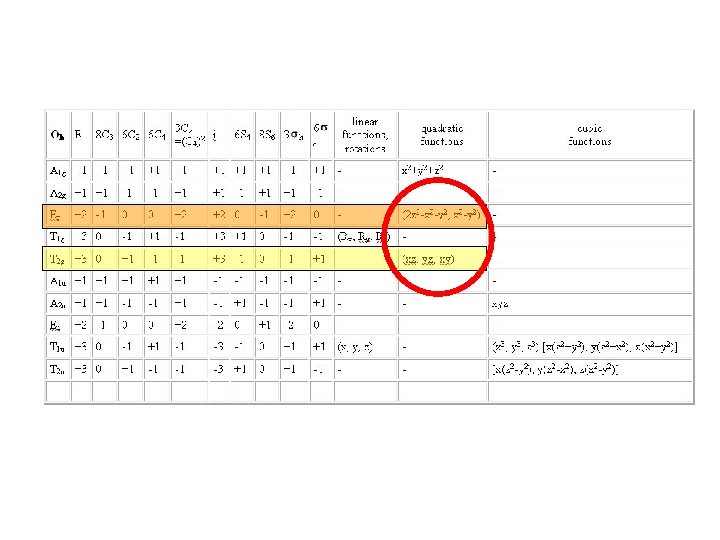
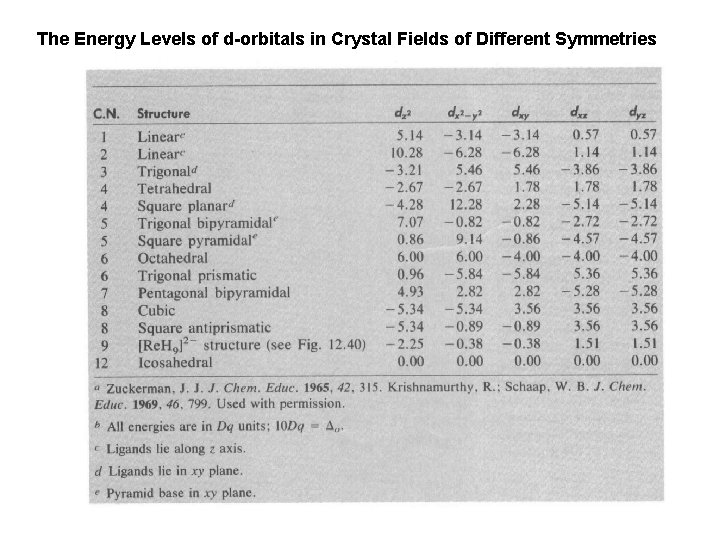
The Energy Levels of d-orbitals in Crystal Fields of Different Symmetries