Crystal Field Theory Analysis of Rovibrational Spectra of

Crystal Field Theory Analysis of Rovibrational Spectra of CO Monomers in Solid Parahydrogen Mario E. Fajardo AFRL/MNME, Energetic Materials Branch, Ordnance Division, U. S. Air Force Research Laboratory, 2306 Perimeter Road, Eglin AFB, FL 32542 -5910. mario. fajardo@eglin. af. mil Takamasa Momose Division of Chemistry, Graduate School of Science, Kyoto University, Kyoto 605 -8502, Japan. * Introduction to MIS in solid parahydrogen (p. H 2) * IR absorption spectra of CO monomers in solid p. H 2 * Crystal Field Theory (CFT) analysis & assignments * Summary 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 1
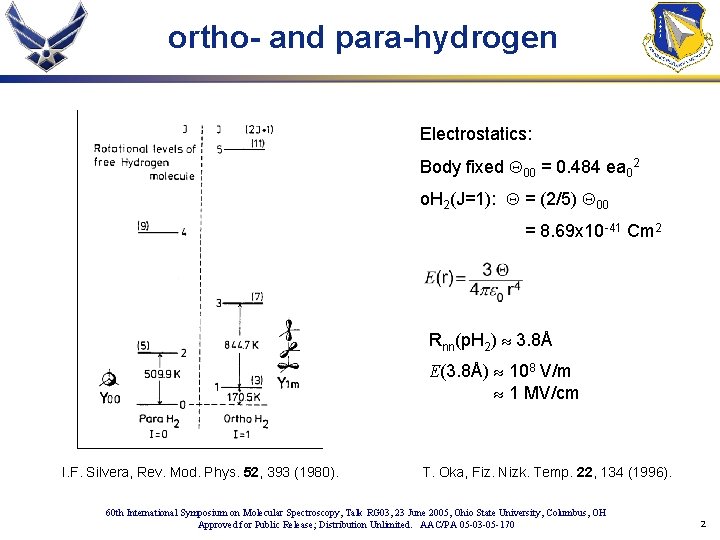
ortho- and para-hydrogen Electrostatics: Body fixed Q 00 = 0. 484 ea 02 o. H 2(J=1): Q = (2/5) Q 00 = 8. 69 x 10 -41 Cm 2 Rnn(p. H 2) » 3. 8Å E(3. 8Å) » 108 V/m » 1 MV/cm I. F. Silvera, Rev. Mod. Phys. 52, 393 (1980). T. Oka, Fiz. Nizk. Temp. 22, 134 (1996). 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 2
Rapid vapor deposition of solid p. H 2 Deposition conditions: Tconv = 15 K Þ ~ 100 ppm o. H 2 Tconv = 19 K Þ ~ 1000 ppm o. H 2 Dnp. H 2/Dt = 200 mmol/hr Dx/Dt = 3. 1 mm/hr Tsub = 2. 4 K ® 2. 9 K Tp. H 2 ® 4. 6 K M. E. Fajardo and S. Tam, J. Chem. Phys. 108, 4237 (1998). S. Tam and M. E. Fajardo, Rev. Sci. Instrum. 70, 1926 (1999). S. Tam and M. E. Fajardo, J. Low Temp. Phys. 122, 345 (2001). Result: mixed fcc/hcp microstructure in as-deposited samples; can anneal to pure hcp. 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 3
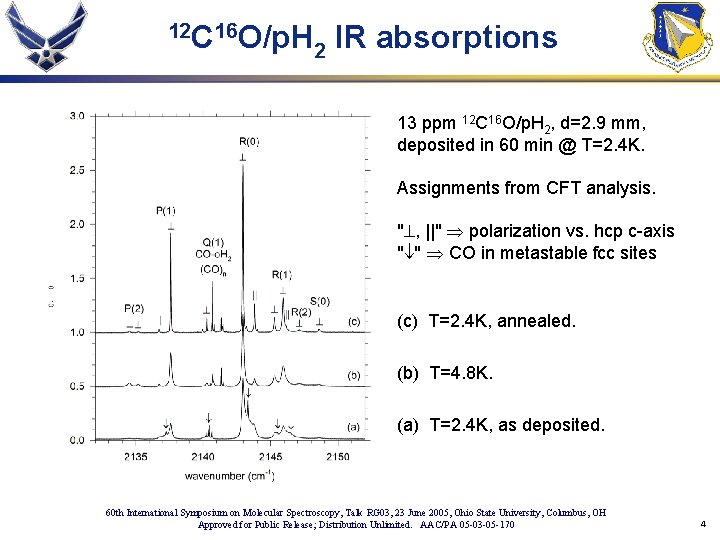
12 C 16 O/p. H 2 IR absorptions 13 ppm 12 C 16 O/p. H 2, d=2. 9 mm, deposited in 60 min @ T=2. 4 K. Assignments from CFT analysis. "^, ||" Þ polarization vs. hcp c-axis "¯" Þ CO in metastable fcc sites (c) T=2. 4 K, annealed. (b) T=4. 8 K. (a) T=2. 4 K, as deposited. 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 4
Devonshire Crystal Field Theory Physical assumptions: (1) Molecular center-of-mass (C. M. ) remains fixed at trapping site center. (2) Rigid, undistorted trapping site. Potential for diatomic rotor in Oh crystal field: Vcry(q, f) = -K 4 {P 40(cos q) + (1/168)P 44(cos q) cos(4 f)} A. F. Devonshire, Proc. Roy. Soc. (London) A 153, 601 (1936). 4 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 5
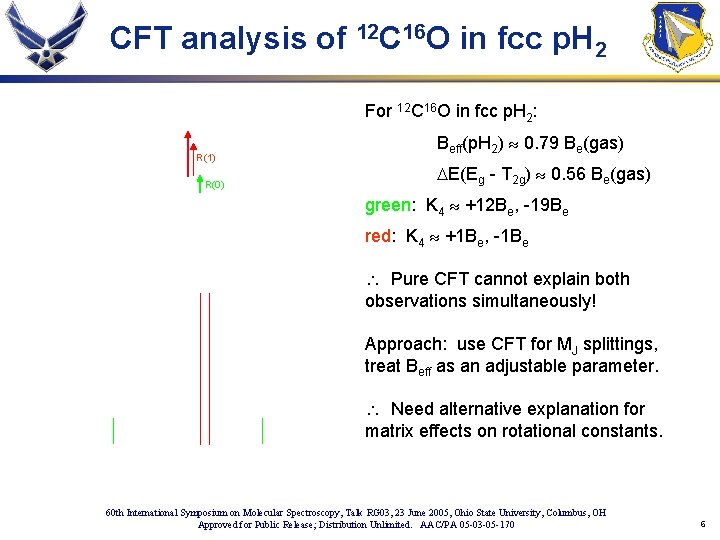
CFT analysis of 12 C 16 O in fcc p. H 2 For 12 C 16 O in fcc p. H 2: R(1) R(0) Beff(p. H 2) » 0. 79 Be(gas) DE(Eg - T 2 g) » 0. 56 Be(gas) green: K 4 » +12 Be, -19 Be red: K 4 » +1 Be, -1 Be Pure CFT cannot explain both observations simultaneously! Approach: use CFT for MJ splittings, treat Beff as an adjustable parameter. Need alternative explanation for matrix effects on rotational constants. 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 6

Diatomic rotor in D 3 h crystal field six adjustable fitting parameters: vibration: we rotation: Be, De, ae crystal field: e 2, e 3 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 7

CFT analysis of 12 C 16 O in hcp p. H 2 constant w 0 Be De ae d e 2 e 3 a b c d e 12 C 16 O values (cm-1) 2140. 295(2)b [2143. 2711]c 1. 577(1) [1. 9313] 0. 0271(1) [0. 00000612] 0. 0129(6) [0. 01750] 1. 026(4) 2. 523(7) 82% 4400 x! ~Be Values in parentheses are the standard deviation (1 s) of the least squares fitting for the last digit. Values in square brackets are those in the gas phase. [J. A. Coxon and P. G. Hajigeorgiou, J. Chem. Phys. 121, 2992 (2004). ] The band origin of the gas phase is calculated by we-2 wexe+(13/4) weye. The ground state rotational constant is B 0=Be-(1/2)ae and that in the excited state is Bv=Be-(3/2)ae. Fit limited to J=0, 1, & 2 levels. 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 8
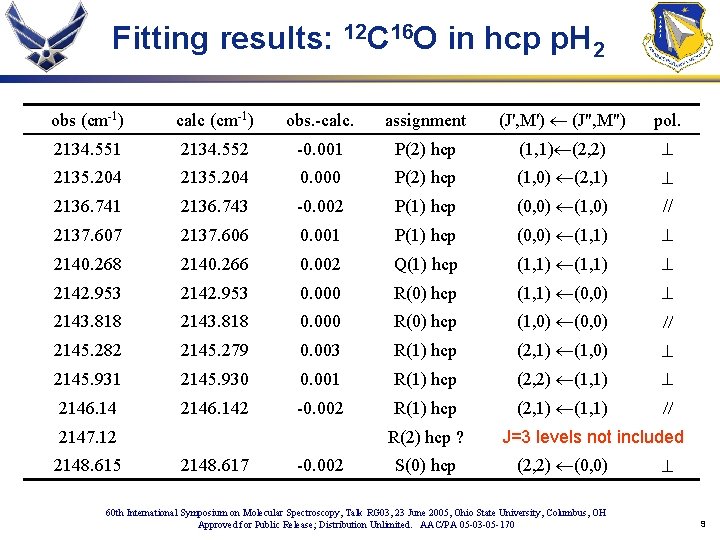
Fitting results: 12 C 16 O in hcp p. H 2 obs (cm-1) calc (cm-1) obs. -calc. assignment (J', M') ¬ (J", M") pol. 2134. 551 2134. 552 -0. 001 P(2) hcp (1, 1)¬(2, 2) ^ 2135. 204 0. 000 P(2) hcp (1, 0) ¬(2, 1) ^ 2136. 741 2136. 743 -0. 002 P(1) hcp (0, 0) ¬(1, 0) // 2137. 607 2137. 606 0. 001 P(1) hcp (0, 0) ¬(1, 1) ^ 2140. 268 2140. 266 0. 002 Q(1) hcp (1, 1) ¬(1, 1) ^ 2142. 953 0. 000 R(0) hcp (1, 1) ¬(0, 0) ^ 2143. 818 0. 000 R(0) hcp (1, 0) ¬(0, 0) // 2145. 282 2145. 279 0. 003 R(1) hcp (2, 1) ¬(1, 0) ^ 2145. 931 2145. 930 0. 001 R(1) hcp (2, 2) ¬(1, 1) ^ 2146. 142 -0. 002 R(1) hcp (2, 1) ¬(1, 1) // 2147. 12 2148. 615 R(2) hcp ? 2148. 617 -0. 002 S(0) hcp J=3 levels not included (2, 2) ¬(0, 0) 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 ^ 9
Energy levels: 12 C 16 O in hcp p. H 2 ? 8. 73 calc 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 10
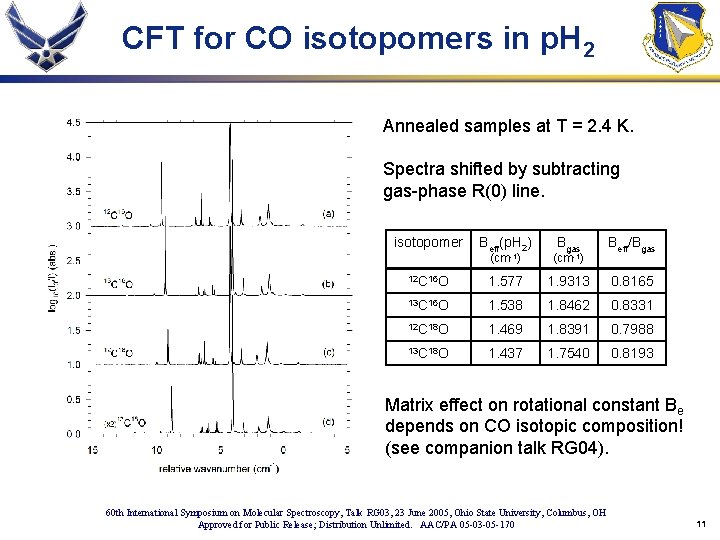
CFT for CO isotopomers in p. H 2 Annealed samples at T = 2. 4 K. Spectra shifted by subtracting gas-phase R(0) line. isotopomer Beff(p. H 2) (cm-1) Bgas (cm-1) Beff/Bgas 12 C 16 O 1. 577 1. 9313 0. 8165 13 C 16 O 1. 538 1. 8462 0. 8331 12 C 18 O 1. 469 1. 8391 0. 7988 13 C 18 O 1. 437 1. 7540 0. 8193 Matrix effect on rotational constant Be depends on CO isotopic composition! (see companion talk RG 04). 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 11

Summary * CO monomers exist as hindered rotors in solid p. H 2. * Detailed spectroscopic assignments accomplished using Crystal Field Theory for diatomic in D 3 h field. * Obtained excellent fits to experimental data. (six parameters vs. ten energy levels) e 2, e 3 ~ B e Beff(p. H 2) » 0. 8 Be(gas) De(p. H 2) » 4400 De(gas) * Matrix effect on rotational constant Be depends on CO isotopomer! (subject of companion talk RG 04. . . ) 60 th International Symposium on Molecular Spectroscopy, Talk RG 03, 23 June 2005, Ohio State University, Columbus, OH Approved for Public Release; Distribution Unlimited. AAC/PA 05 -03 -05 -170 12
- Slides: 12