CRYO 4 PERSISTENT AF Study Cryoballoon Ablation in

CRYO 4 PERSISTENT AF Study Cryoballoon Ablation in Persistent Atrial Fibrillation Patients Single Procedure Outcomes Following Pulmonary Vein Isolation in the Prospective Multicenter CRYO 4 PERSISTENT AF Study (Clinical. Trials. gov NCT 02213731)

Contents § § Background Study Design Primary Objective Results § § Demographics Efficacy Safety QOL / Arrhythmic Symptoms § Conclusions

Study Design CRYO 4 PERSISTENT AF (NCT 02213731) – Prospective, multi-center, non-randomized, single arm study – Designed to assess the single procedure outcomes of PVI using Arctic Front Advance™ cryoballoon – Patients with symptomatic Per. AF – 11 sites in Europe (France, Germany, and Greece) – 101 patients met I/E criteria, demonstrated 100% AF burden, were treated per protocol, and included in the primary analysis Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain.

Primary Objective • Freedom from all arrhythmias (AF/AT/AFL) at 12 months – Core lab adjudicated recurrence of AF/AT/AFL ≥ 30 sec and/or DCCV (outside of 3 month blanking period) – Repeat ablation post index-procedure (inclusive of 3 -month blanking period) • Only use of Arctic Front Advance cryoballoon is allowed to obtain PVI – No additional left atrial empirical lesions and/or complex fractionated electrogram ablations Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain.
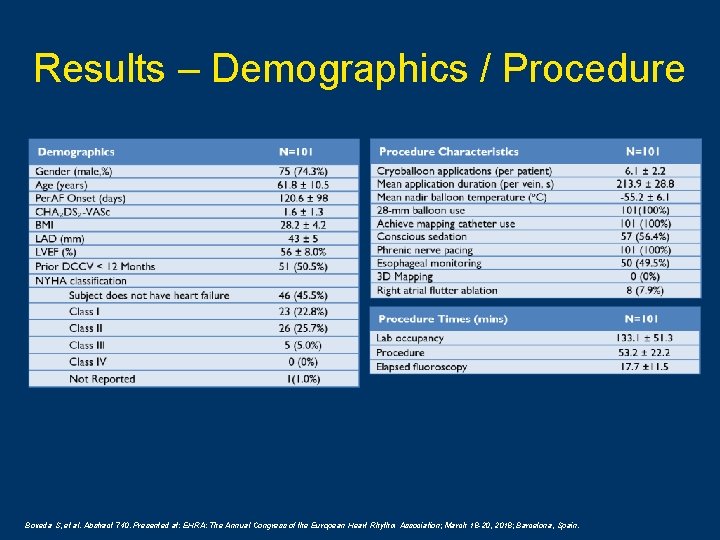
Results – Demographics / Procedure Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain.
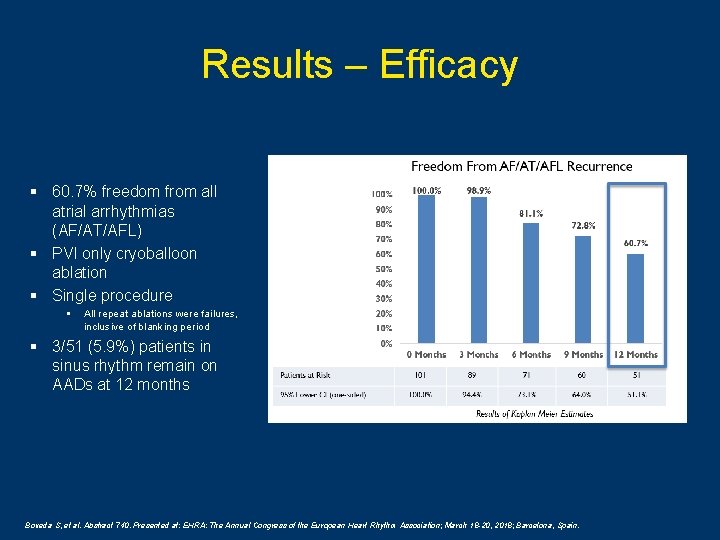
Results – Efficacy § 60. 7% freedom from all atrial arrhythmias (AF/AT/AFL) § PVI only cryoballoon ablation § Single procedure § All repeat ablations were failures, inclusive of blanking period § 3/51 (5. 9%) patients in sinus rhythm remain on AADs at 12 months Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain.
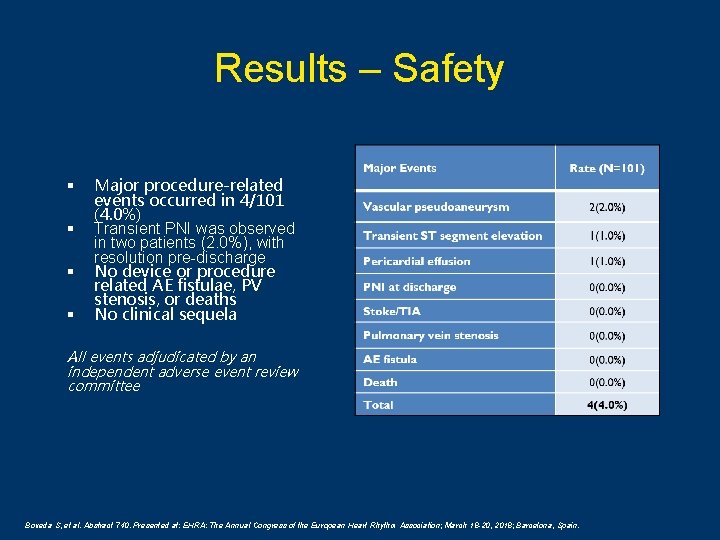
Results – Safety § § Major procedure-related events occurred in 4/101 (4. 0%) Transient PNI was observed in two patients (2. 0%), with resolution pre-discharge No device or procedure related AE fistulae, PV stenosis, or deaths No clinical sequela All events adjudicated by an independent adverse event review committee Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain.
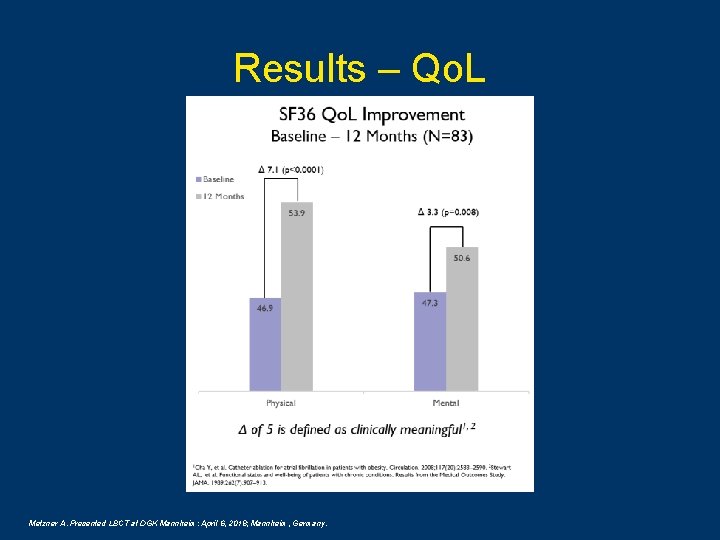
Results – Qo. L Metzner A. Presented LBCT at DGK Mannheim: April 6, 2018; Mannheim, Germany.
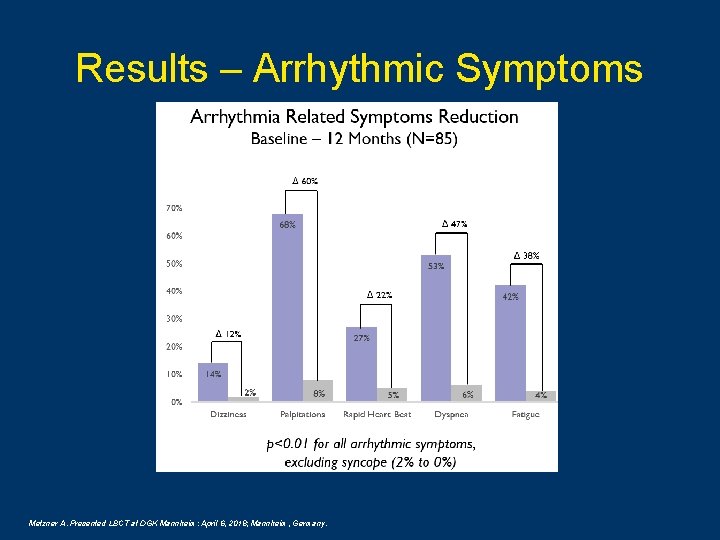
Results – Arrhythmic Symptoms Metzner A. Presented LBCT at DGK Mannheim: April 6, 2018; Mannheim, Germany.

Conclusions This is the first prospective multicenter study reporting single procedure PVI only effectiveness of cryoballoon ablation in persistent AF patients using the Arctic Front Advance exclusively. In this cohort of Per. AF patients, cryoballoon is an effective treatment with freedom from all atrial arrhythmias of 61% at 12 months with predictable procedure times. Cryoballoon demonstrated a low device and procedure related complication rate of 4% with no long term clinical sequela. Patients treated with the cryoballoon demonstrated a clinically significant improvement in arrhythmia-related symptoms and QOL from baseline to 12 months post-ablation. Boveda S, et al. Abstract 740. Presented at: EHRA: The Annual Congress of the European Heart Rhythm Association; March 18 -20, 2018; Barcelona, Spain. Metzner A. Presented LBCT at DGK Mannheim: April 6, 2018; Mannheim, Germany.
Indications The Arctic Front Advance™ Cardiac Cryo. Ablation Catheter catheter is indicated for the treatment of patients with atrial fibrillation (AF). Contraindications Use of Arctic Front Advance™ Cryoballoon is contraindicated as follows: In the ventricle because of the danger of catheter entrapment in the chordae tendinae In patients with active systemic infections In conditions where the manipulation of the catheter within the heart would be unsafe (e. g. , intracardiac mural thrombus) In patients with cryoglobulinemia In patients with one or more pulmonary vein stents Brief Statement Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please contact your Medtronic Sales Representative and/or consult Medtronic’s website at www. medtronic. com.
- Slides: 11