CRT 2016 Washington DC February 2016 Prospects for

CRT 2016, Washington, DC. February 2016 Prospects for CV Risk Factor Reduction with PCSK 9 and CEPT Inhibitor Therapies Heart and Vascular Institute A. Michael Lincoff, M. D. Director, C 5 Research (Cleveland Clinic Coordinating Center for Clinical Research) Vice Chairman for Clinical Research, Lerner Research Institute Vice Chairman of Cardiovascular Medicine Professor of Medicine AML

Speaker Disclosure – A. Michael Lincoff, MD Relationships with Industry Research Funding: Astra. Zeneca, CSL Behring, Eli Lilly, Esperion, Pfizer, Roche Consultant: Abbott, Amgen, Sarepta, Sermonix, St. Jude Medical Supervisory – C 5 Research Director: Amgen, Atricure, Cardiovascular Systems, Centocor, Covidien, Edwards Lifesciences, Early Sense, Ethicon, Janssen, Juventas, Medicines Company, Medtronic, Novartis, Orexigen, Stemedica, Takeda AML
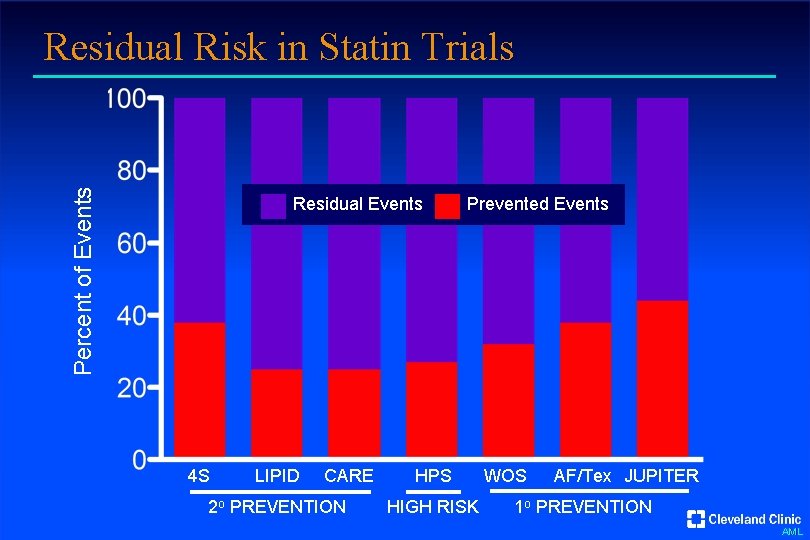
Percent of Events Residual Risk in Statin Trials Residual Events 4 S LIPID CARE 2 o PREVENTION Prevented Events HPS HIGH RISK WOS AF/Tex JUPITER 1 o PREVENTION AML

Dyslipidemia Rx Beyond Statins CETP Inhibitors PCSK 9 Inhibitors AML
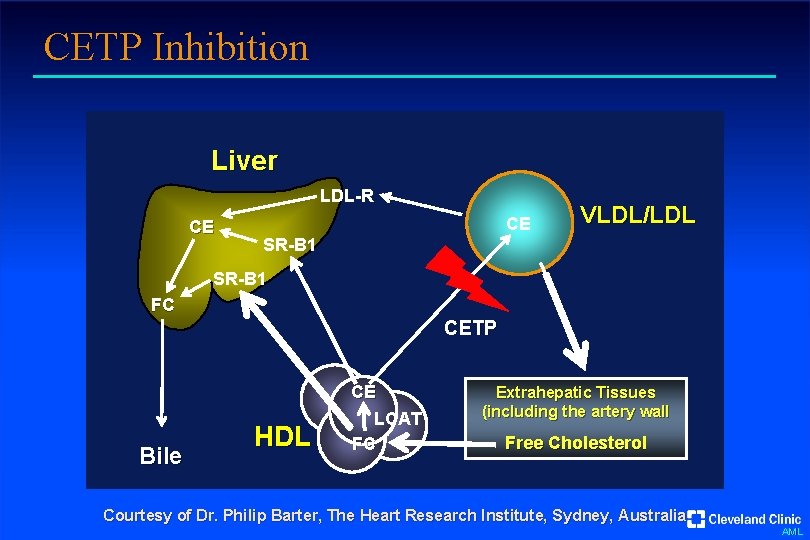
CETP Inhibition Liver LDL-R CE CE VLDL/LDL SR-B 1 FC CETP Bile HDL CE LCAT FC Extrahepatic Tissues (including the artery wall Free Cholesterol Courtesy of Dr. Philip Barter, The Heart Research Institute, Sydney, Australia AML
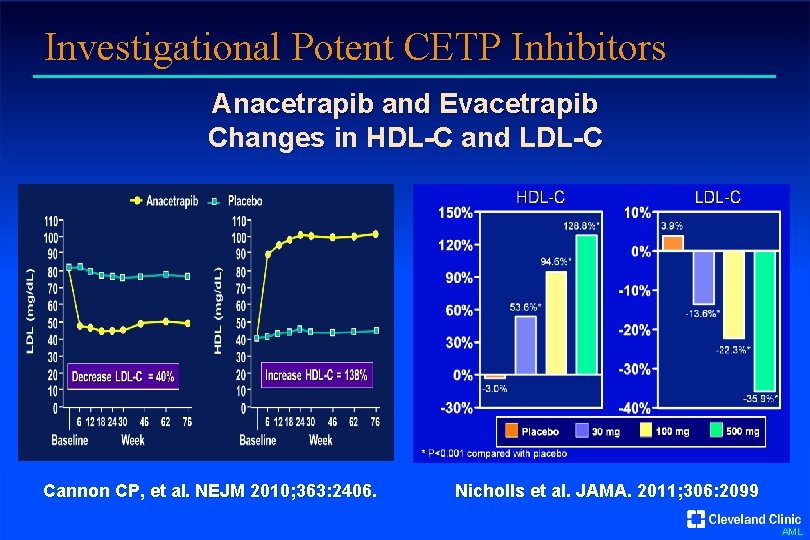
Investigational Potent CETP Inhibitors Anacetrapib and Evacetrapib Changes in HDL-C and LDL-C Cannon CP, et al. NEJM 2010; 363: 2406. Nicholls et al. JAMA. 2011; 306: 2099 AML
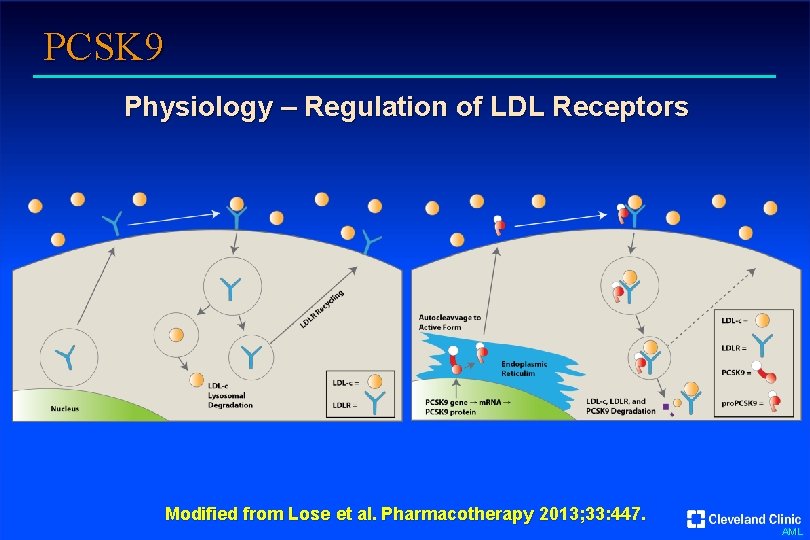
PCSK 9 Physiology – Regulation of LDL Receptors Modified from Lose et al. Pharmacotherapy 2013; 33: 447. AML
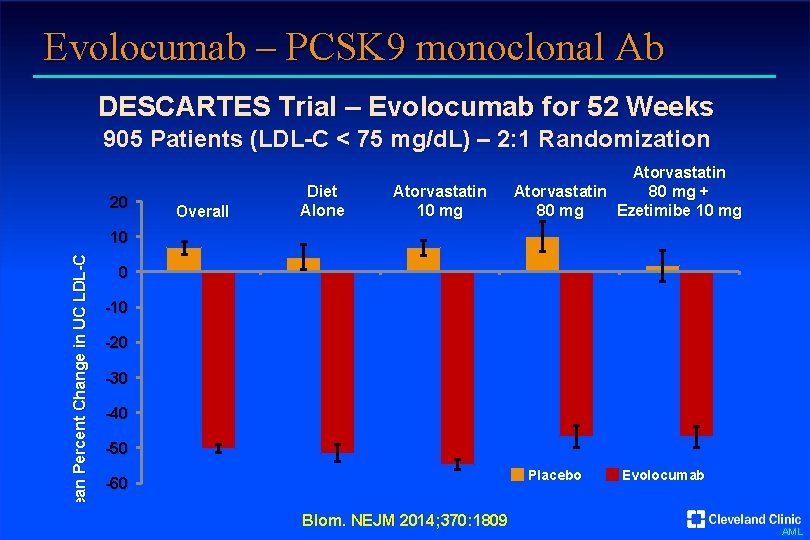
Evolocumab – PCSK 9 monoclonal Ab DESCARTES Trial – Evolocumab for 52 Weeks 905 Patients (LDL-C < 75 mg/d. L) – 2: 1 Randomization 20 Overall Diet Alone Atorvastatin 10 mg Atorvastatin 80 mg + Atorvastatin Ezetimibe 10 mg 80 mg Mean Percent Change in UC LDL-C 10 0 -10 -20 -30 -40 -50 Placebo -60 Blom. NEJM 2014; 370: 1809 Evolocumab AML
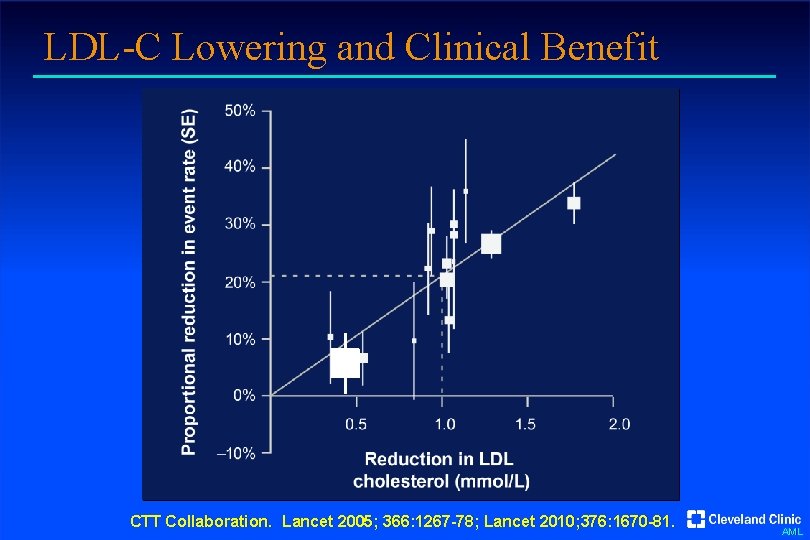
LDL-C Lowering and Clinical Benefit CTT Collaboration. Lancet 2005; 366: 1267 -78; Lancet 2010; 376: 1670 -81. AML

CETP and PCSK 9 Inhibition The Science Behind Clinical Development § Animal models § Human genetic variations – GOF (“gain of function”) or LOF (“loss of function”) § Associations with plasma mass or activity AML
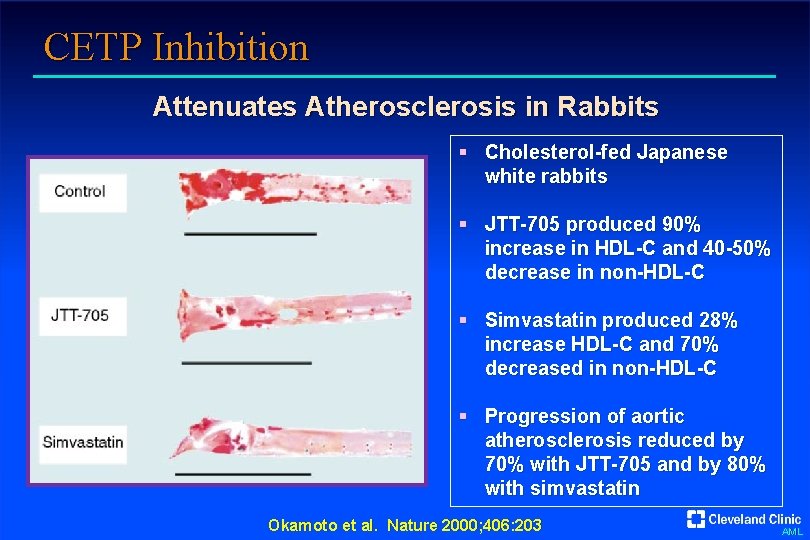
CETP Inhibition Attenuates Atherosclerosis in Rabbits § Cholesterol-fed Japanese white rabbits § JTT-705 produced 90% increase in HDL-C and 40 -50% decrease in non-HDL-C § Simvastatin produced 28% increase HDL-C and 70% decreased in non-HDL-C § Progression of aortic atherosclerosis reduced by 70% with JTT-705 and by 80% with simvastatin Okamoto et al. Nature 2000; 406: 203 AML
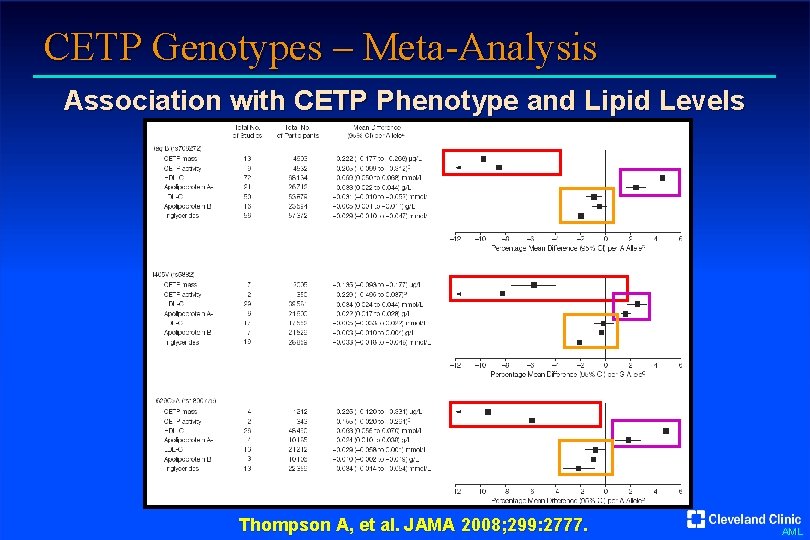
CETP Genotypes – Meta-Analysis Association with CETP Phenotype and Lipid Levels Thompson A, et al. JAMA 2008; 299: 2777. AML
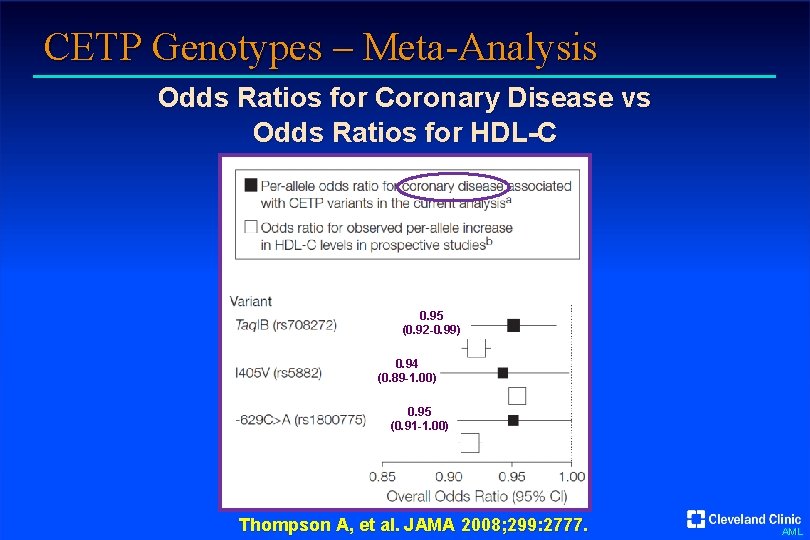
CETP Genotypes – Meta-Analysis Odds Ratios for Coronary Disease vs Odds Ratios for HDL-C 0. 95 (0. 92 -0. 99) 0. 94 (0. 89 -1. 00) 0. 95 (0. 91 -1. 00) Thompson A, et al. JAMA 2008; 299: 2777. AML
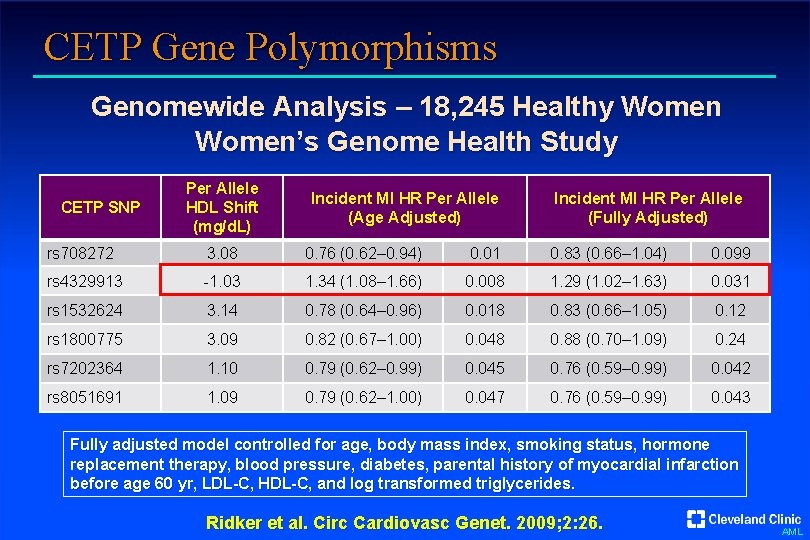
CETP Gene Polymorphisms Genomewide Analysis – 18, 245 Healthy Women’s Genome Health Study CETP SNP Per Allele HDL Shift (mg/d. L) Incident MI HR Per Allele (Age Adjusted) Incident MI HR Per Allele (Fully Adjusted) rs 708272 3. 08 0. 76 (0. 62– 0. 94) 0. 01 0. 83 (0. 66– 1. 04) 0. 099 rs 4329913 -1. 03 1. 34 (1. 08– 1. 66) 0. 008 1. 29 (1. 02– 1. 63) 0. 031 rs 1532624 3. 14 0. 78 (0. 64– 0. 96) 0. 018 0. 83 (0. 66– 1. 05) 0. 12 rs 1800775 3. 09 0. 82 (0. 67– 1. 00) 0. 048 0. 88 (0. 70– 1. 09) 0. 24 rs 7202364 1. 10 0. 79 (0. 62– 0. 99) 0. 045 0. 76 (0. 59– 0. 99) 0. 042 rs 8051691 1. 09 0. 79 (0. 62– 1. 00) 0. 047 0. 76 (0. 59– 0. 99) 0. 043 Fully adjusted model controlled for age, body mass index, smoking status, hormone replacement therapy, blood pressure, diabetes, parental history of myocardial infarction before age 60 yr, LDL-C, HDL-C, and log transformed triglycerides. Ridker et al. Circ Cardiovasc Genet. 2009; 2: 26. AML

CETP Mass or Concentration Studies NOT supportive of inhibition, showing reduced risk of CV events with higher CETP levels Khera et al. 1 3218 pts in PROVE-IT MI/death in highest vs lowest quartile: HR 0. 81 (0. 67 -0. 98), p = 0. 027 Ritsch et al. 2 3256 pts in LURIC study CV death in highest vs lowest quartile: HR 0. 72 (0. 55 -0. 95), p = 0. 021 1 AJC 2010; 106: 451 2 Circ 2010; 121: 366 3 EHJ 2007; 28: 1012 Borggreve et al. 3 111 cases/116 controls in PREVEND Study CV events above vs below median (only pts with TGs below median) HR 0. 46 (0. 24 -0. 90), p = 0. 03 AML

ACCELERATE Trial Termination – October 12, 2015 “The Data Monitoring Committee (DMC) met on October 7, 2015 to conduct a periodic safety data review to evaluate the need for early termination of the study for adverse safety or lack of efficacy (futility). . . … evidence has emerged to support a recommendation to stop the study as the totality of evidence suggests that evacetrapib is unlikely to be superior to placebo at study end. ” ACC LBCT Presentation – Sunday April 3, 2016; 8: 30 am What to look for: • • What were the effects on HDL-C? What were the effect on LDL-C? What were 1 o endpoint events rates (power)? What were rates of components of 1 o endpoint? AML
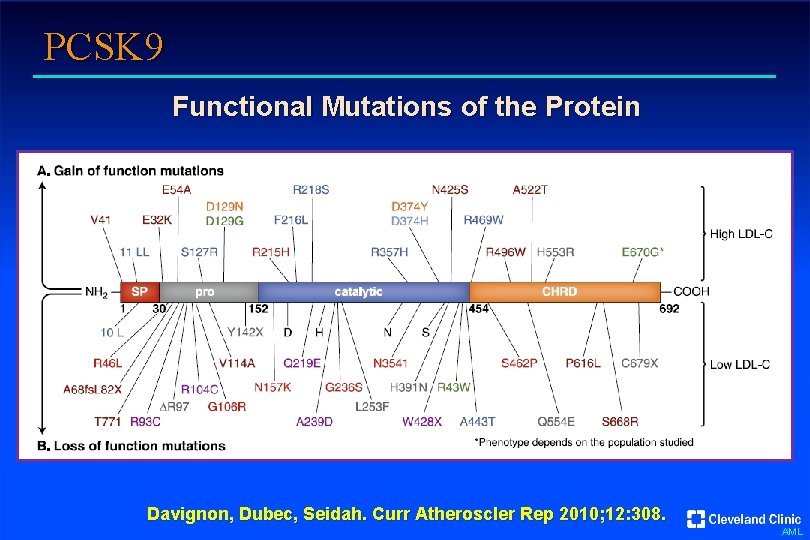
PCSK 9 Functional Mutations of the Protein Davignon, Dubec, Seidah. Curr Atheroscler Rep 2010; 12: 308. AML
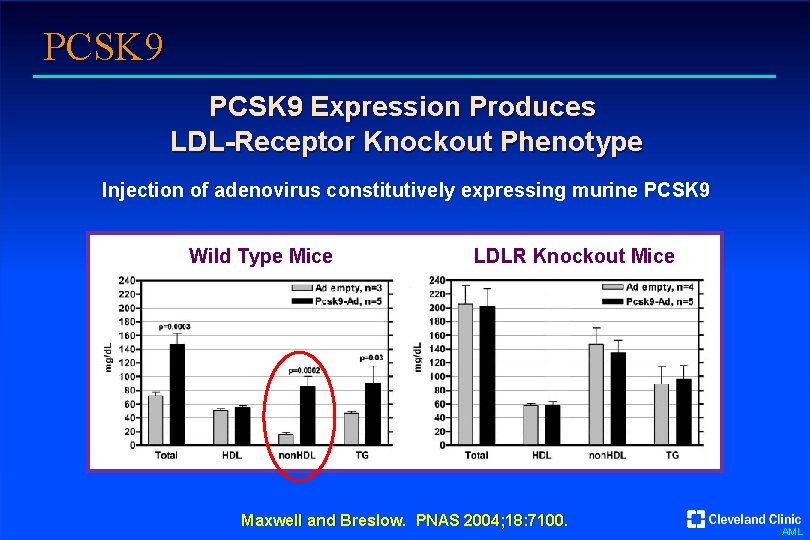
PCSK 9 Expression Produces LDL-Receptor Knockout Phenotype Injection of adenovirus constitutively expressing murine PCSK 9 Wild Type Mice LDLR Knockout Mice Maxwell and Breslow. PNAS 2004; 18: 7100. AML
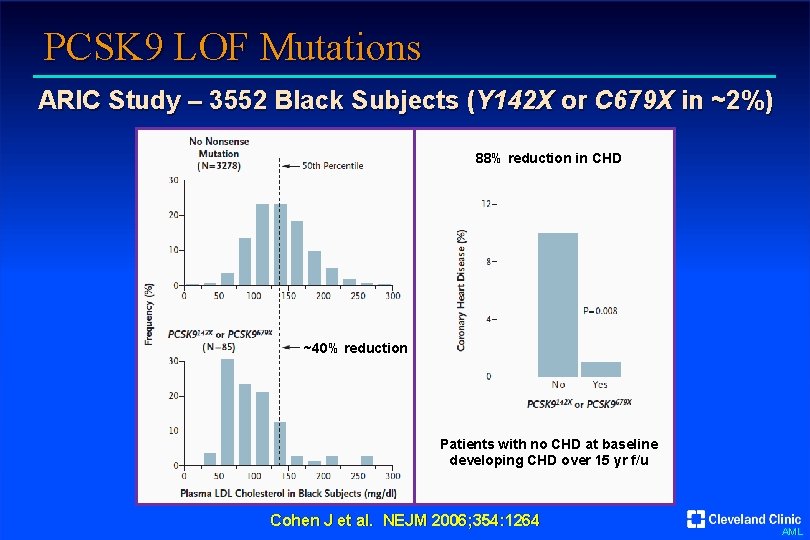
PCSK 9 LOF Mutations ARIC Study – 3552 Black Subjects (Y 142 X or C 679 X in ~2%) 88% reduction in CHD ~40% reduction Patients with no CHD at baseline developing CHD over 15 yr f/u Cohen J et al. NEJM 2006; 354: 1264 AML
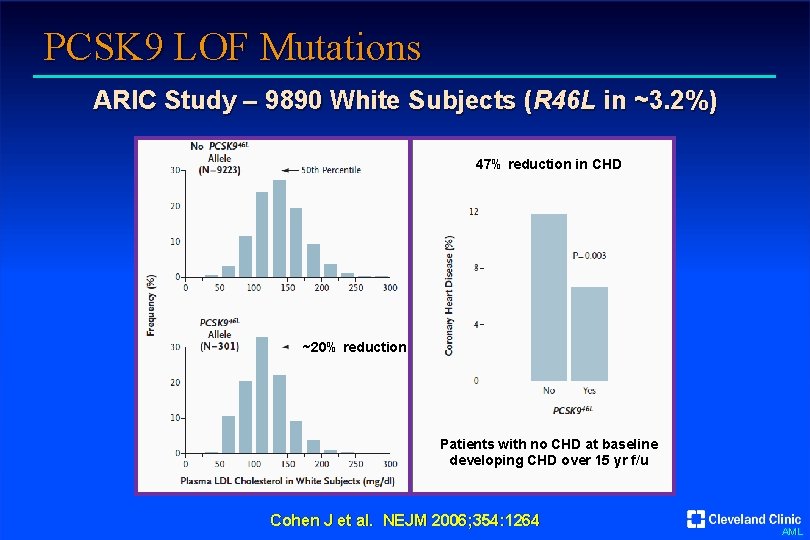
PCSK 9 LOF Mutations ARIC Study – 9890 White Subjects (R 46 L in ~3. 2%) 47% reduction in CHD ~20% reduction Patients with no CHD at baseline developing CHD over 15 yr f/u Cohen J et al. NEJM 2006; 354: 1264 AML
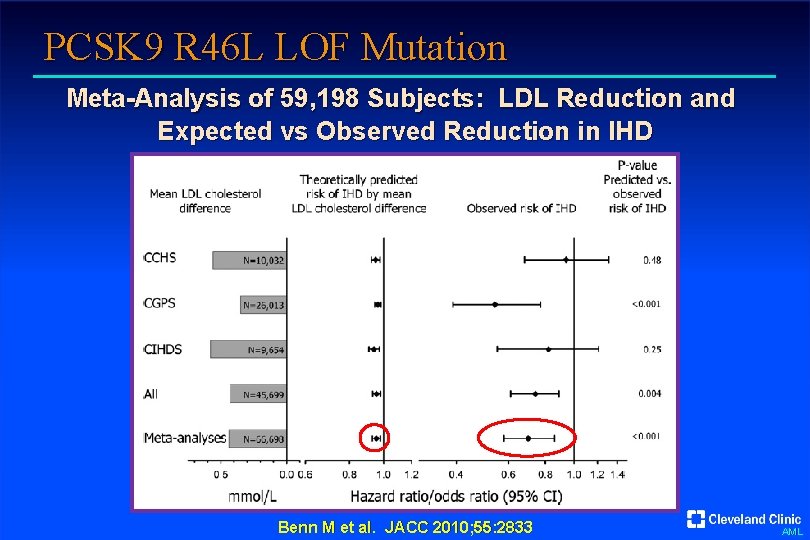
PCSK 9 R 46 L LOF Mutation Meta-Analysis of 59, 198 Subjects: LDL Reduction and Expected vs Observed Reduction in IHD Benn M et al. JACC 2010; 55: 2833 AML

CETP and PCSK 9 Inhibition Summary § CETP inhibition may enhance reverse cholesterol transport to liver. Most genotypes causing reductions in CETP mass or activity (5 -10%) are associated with modest increases in HDL-C (3 -5%) and small decreases in CV risk (3 -5%). § CETP inhibitors in current trials increase HDL-C by ~100% and reduce LDL-C by ~40%. The CVOT of the potent CETPi evacetrapib showed no risk reduction; another CVOT of anacetrapib remains underway. § PCSK 9 is a key modulator of LDL-C levels via degradation of the LDL receptor. Loss-of-function genetic variants consistently associated with reduced levels of LDL-C and reduced risk of CV disease. § PCSK 9 antibodies in current CVOTs reduce LDL-C by ~60% beyond by statins: very promising potential to further improve CV outcomes. AML

Heart and Vascular Institute AML
- Slides: 23