CRT 2016 Washington DC February 2016 Heparin Should

CRT 2016, Washington, DC. February 2016 Heparin Should be the First-line Therapy for Patients with ACS/AMI Heart and Vascular Institute A. Michael Lincoff, M. D. Director, C 5 Research (Cleveland Clinic Coordinating Center for Clinical Research) Vice Chairman for Clinical Research, Lerner Research Institute Vice Chairman of Cardiovascular Medicine Professor of Medicine AML

Speaker Disclosure – A. Michael Lincoff, MD Relationships with Industry Research Funding: Astra. Zeneca, CSL Behring, Eli Lilly, Esperion, Pfizer, Roche Consultant: Abbott, Amgen, Sarepta, Sermonix, St. Jude Medical Supervisory – C 5 Research Director: Amgen, Atricure, Cardiovascular Systems, Centocor, Covidien, Edwards Lifesciences, Early Sense, Ethicon, Janssen, Juventas, Medicines Company, Medtronic, Novartis, Orexigen, Stemedica, Takeda AML

Paradigm for Antithrombotic Drugs More Bleeding Goal of New Rx Less Thrombosis More AML
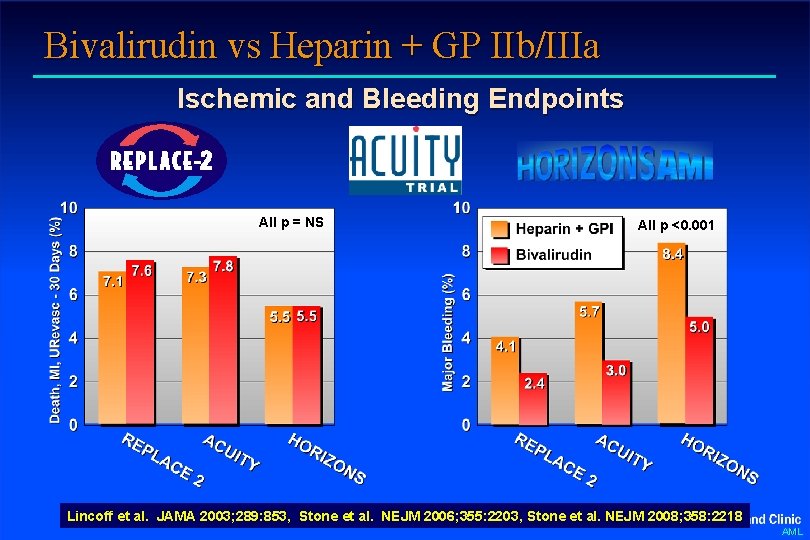
Bivalirudin vs Heparin + GP IIb/IIIa Ischemic and Bleeding Endpoints All p = NS All p <0. 001 Lincoff et al. JAMA 2003; 289: 853, Stone et al. NEJM 2006; 355: 2203, Stone et al. NEJM 2008; 358: 2218 AML
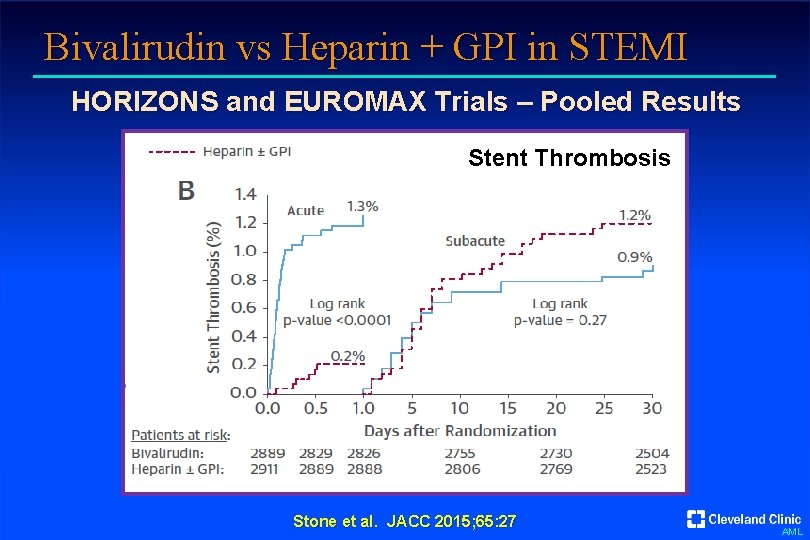
Bivalirudin vs Heparin + GPI in STEMI HORIZONS and EUROMAX Trials – Pooled Results Stent Thrombosis Stone et al. JACC 2015; 65: 27 AML

Heparin • Combine 5, 000 lbs intestines, 200 gallons water, 10 gallons chloroform, and 5 gallons toluene. Hold at 90°F for 17 hours. • Add 30 gallons acetic acid, 35 gallons ammonia, sodium hydroxide to adjust p. H, and 235 gallons water. Bring to a boil; then filter. • Add 200 gallons hot water to filtrate and allow to stand overnight, then skim off the fat. • Keep pancreatic extract at 100°F for three days, then bring to boil. • Filter solids and assay for heparin content. Kleiman NS and Weitz JI. J Inv Cardiol 2000; 12: 20 F. AML

Bivalirudin shorter hospital stays. Bivalirudin is a synthetic version of hirudin, an anticoagulant in leech saliva. Researchers have been drooling lately over medical applications of the saliva of blood-suckers. Last month, Clinic researchers began testing a substance AML
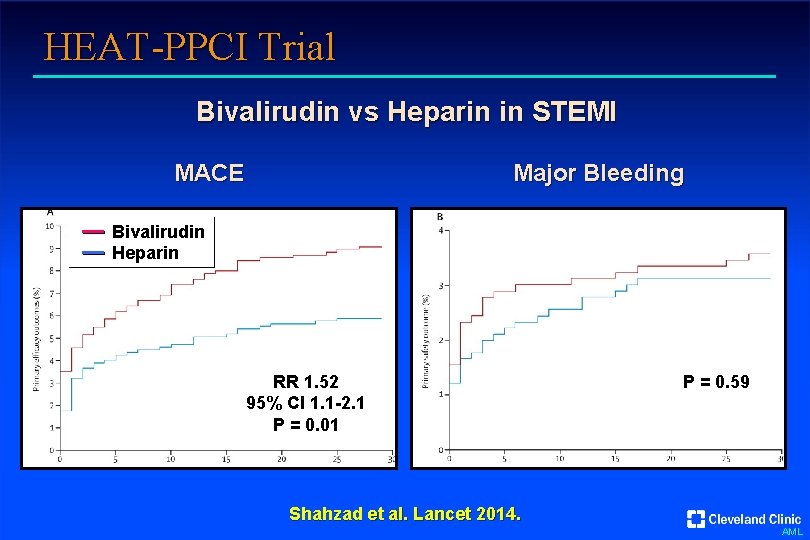
HEAT-PPCI Trial Bivalirudin vs Heparin in STEMI MACE Major Bleeding Bivalirudin Heparin RR 1. 52 95% CI 1. 1 -2. 1 P = 0. 01 P = 0. 59 Shahzad et al. Lancet 2014. AML
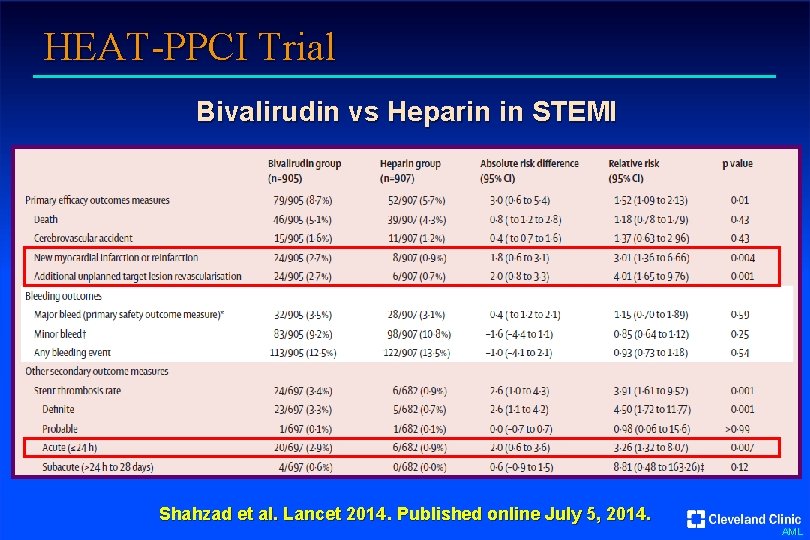
HEAT-PPCI Trial Bivalirudin vs Heparin in STEMI Shahzad et al. Lancet 2014. Published online July 5, 2014. AML
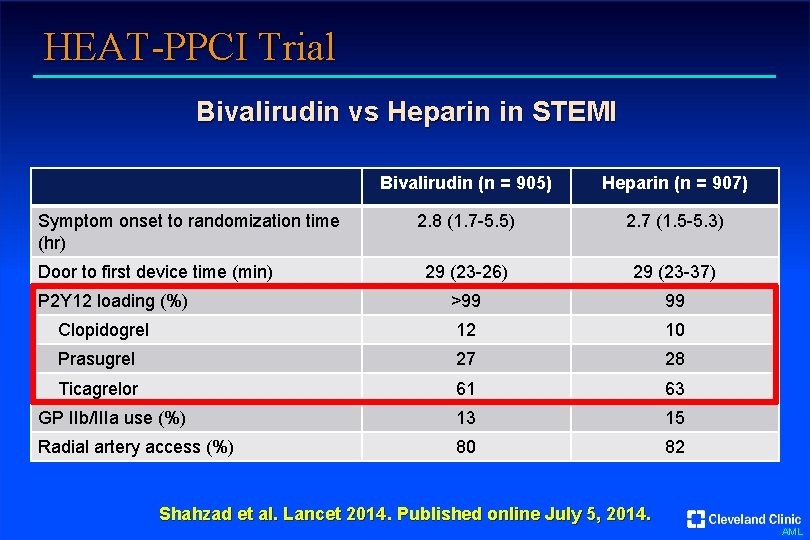
HEAT-PPCI Trial Bivalirudin vs Heparin in STEMI Bivalirudin (n = 905) Heparin (n = 907) 2. 8 (1. 7 -5. 5) 2. 7 (1. 5 -5. 3) 29 (23 -26) 29 (23 -37) >99 99 Clopidogrel 12 10 Prasugrel 27 28 Ticagrelor 61 63 GP IIb/IIIa use (%) 13 15 Radial artery access (%) 80 82 Symptom onset to randomization time (hr) Door to first device time (min) P 2 Y 12 loading (%) Shahzad et al. Lancet 2014. Published online July 5, 2014. AML
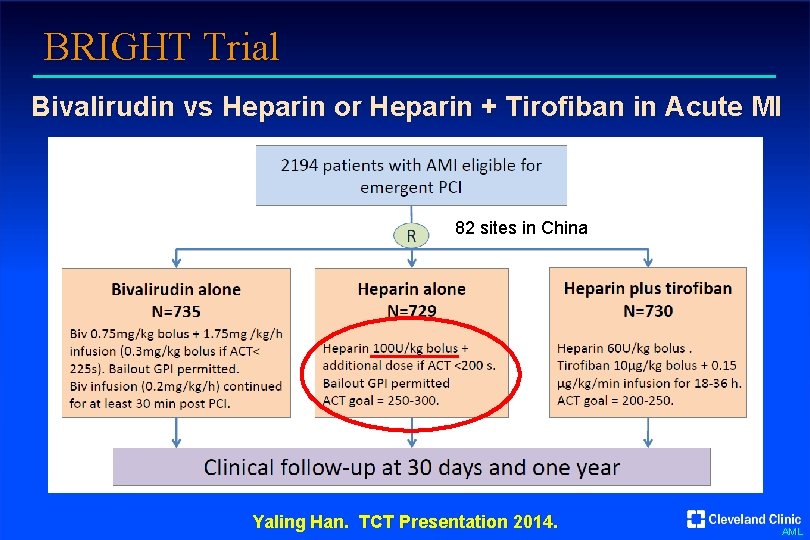
BRIGHT Trial Bivalirudin vs Heparin or Heparin + Tirofiban in Acute MI 82 sites in China Yaling Han. TCT Presentation 2014. AML
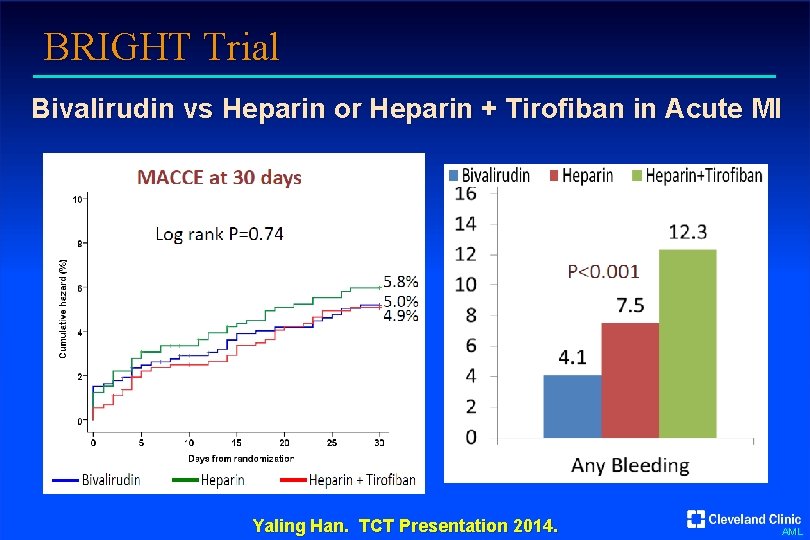
BRIGHT Trial Bivalirudin vs Heparin or Heparin + Tirofiban in Acute MI Yaling Han. TCT Presentation 2014. AML
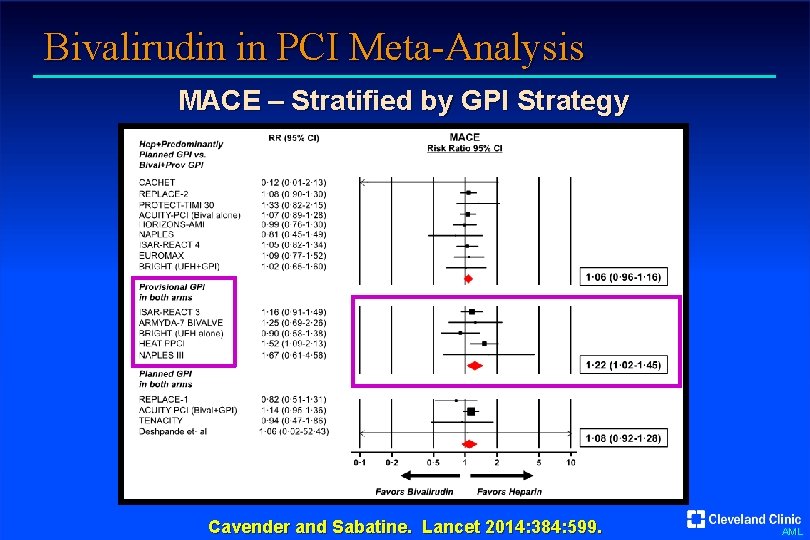
Bivalirudin in PCI Meta-Analysis MACE – Stratified by GPI Strategy Cavender and Sabatine. Lancet 2014: 384: 599. AML
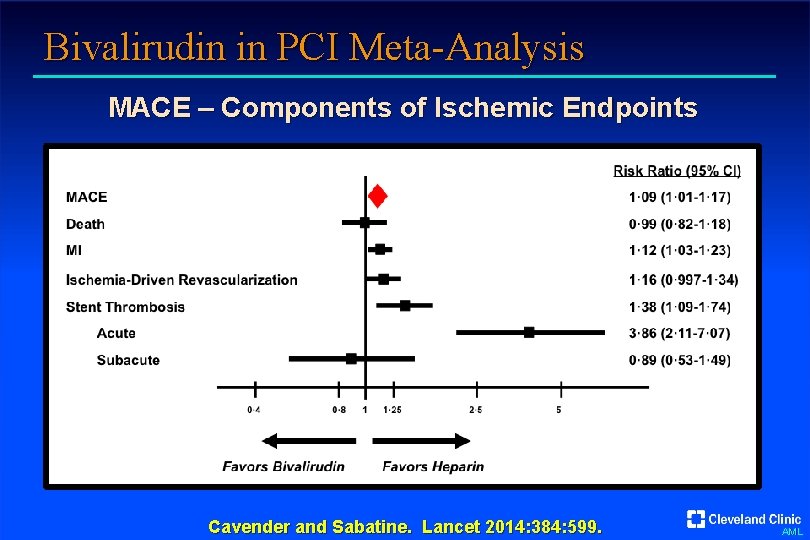
Bivalirudin in PCI Meta-Analysis MACE – Components of Ischemic Endpoints Cavender and Sabatine. Lancet 2014: 384: 599. AML
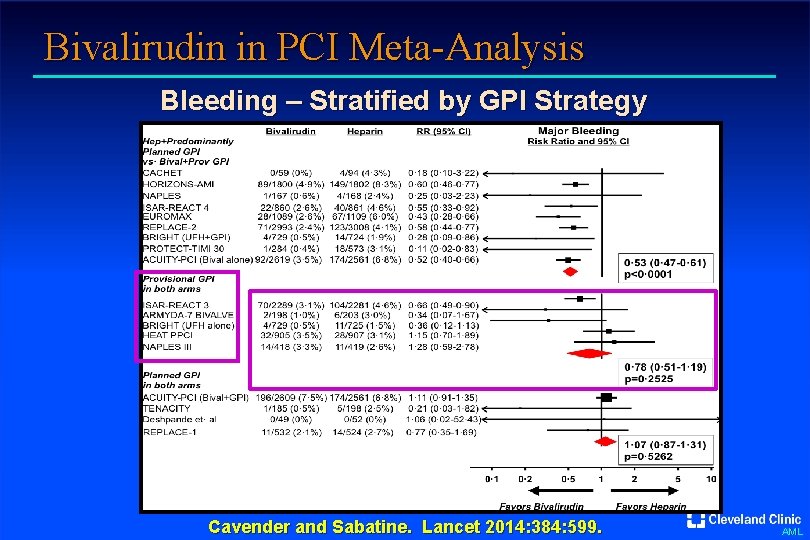
Bivalirudin in PCI Meta-Analysis Bleeding – Stratified by GPI Strategy Cavender and Sabatine. Lancet 2014: 384: 599. AML

MATRIX Trial – Bivalirudin vs UFH in ACS STEMI or NSTE-ACS with Invasive Management Aspirin + P 2 Y 12 Inhibitor Radial Bivalirudin* Access 1: 1 Antithrombin 1: 1 Femoral Unfractionated Heparin 70 -100 U/Kg w/o GPI 50 -70 U/Kg w/ GPI + Bailout GPI + Planned or Bailout GPI (N = 3610 Pts) (N = 3603 Pts) *47. 7% underwent prolonged Bivalirudin infusion post-PCI, of which 36% received 1. 75 mg/kg-min regimen N Engl J Med 2015; 373: 997 -1009 AML
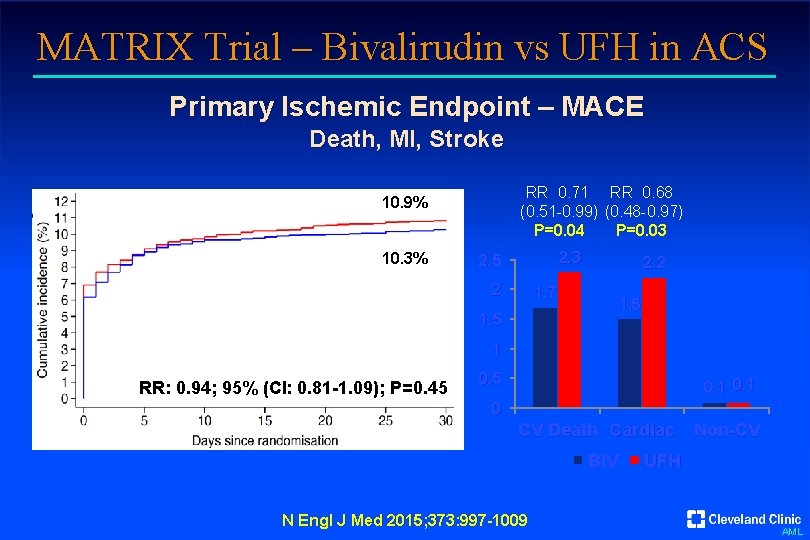
MATRIX Trial – Bivalirudin vs UFH in ACS Primary Ischemic Endpoint – MACE Death, MI, Stroke RR 0. 71 RR 0. 68 (0. 51 -0. 99) (0. 48 -0. 97) P=0. 04 P=0. 03 10. 9% 10. 3% 2. 3 2. 5 2 2. 2 1. 7 1. 5 1 RR: 0. 94; 95% (CI: 0. 81 -1. 09); P=0. 45 0. 1 0 CV Death Cardiac Non-CV BIV N Engl J Med 2015; 373: 997 -1009 UFH AML

MATRIX Trial – Bivalirudin vs UFH in ACS Bleeding Endpoints Bivalirudin UFH 2. 5 2 1. 5 P=0. 07 RR: 0. 59 0. 33 -1. 04 0. 9 1 P=0. 005 RR: 0. 53 0. 34 -0. 83 1. 6 P=0. 002 RR: 0. 50 0. 33 -0. 75 1. 9 P=0. 027 RR: 0. 61 0. 39 -0. 95 1. 3 0. 8 0. 6 0. 5 BARC 3 or 5 0 P=0. 008 RR: 0. 61 0. 42 -0. 88 2. 1 P=0. 0016 RR: 0. 31 0. 11 -0. 85 0. 4 1. 0 0. 9 0. 1 Access Site Non Access BARC 3 BARC 5 Site N Engl J Med 2015; 373: 997 -1009 TIMI GUSTO AML
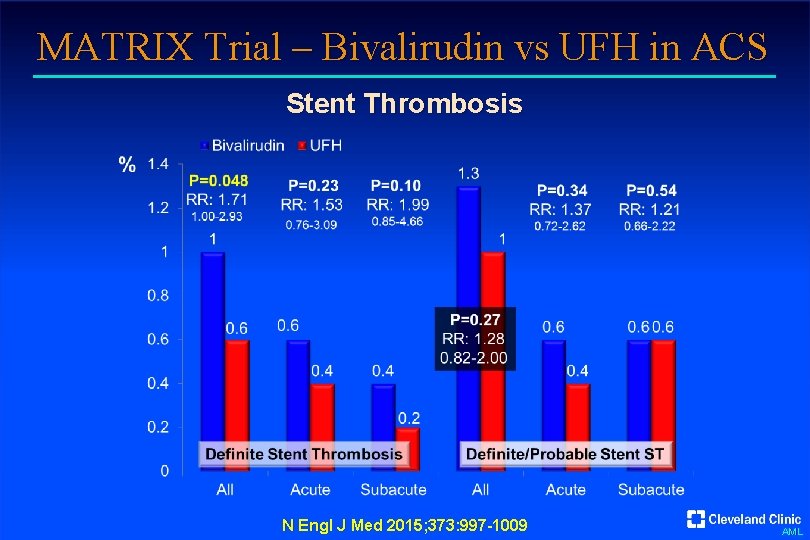
MATRIX Trial – Bivalirudin vs UFH in ACS Stent Thrombosis N Engl J Med 2015; 373: 997 -1009 AML
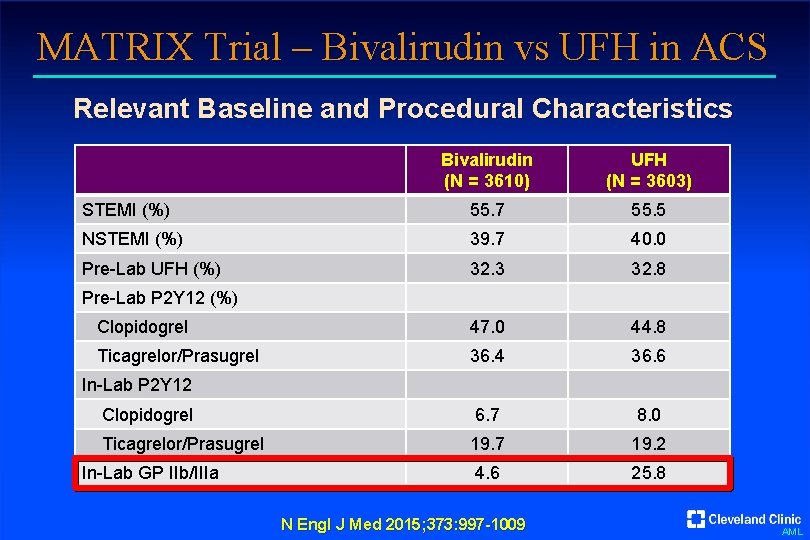
MATRIX Trial – Bivalirudin vs UFH in ACS Relevant Baseline and Procedural Characteristics Bivalirudin (N = 3610) UFH (N = 3603) STEMI (%) 55. 7 55. 5 NSTEMI (%) 39. 7 40. 0 Pre-Lab UFH (%) 32. 3 32. 8 Clopidogrel 47. 0 44. 8 Ticagrelor/Prasugrel 36. 4 36. 6 Clopidogrel 6. 7 8. 0 Ticagrelor/Prasugrel 19. 7 19. 2 4. 6 25. 8 Pre-Lab P 2 Y 12 (%) In-Lab P 2 Y 12 In-Lab GP IIb/IIIa N Engl J Med 2015; 373: 997 -1009 AML

Bivalirudin vs Heparin AML

Heart and Vascular Institute AML
- Slides: 22