CRT 2016 The Case for Bivalirudin Roxana Mehran

CRT 2016 The Case for Bivalirudin Roxana Mehran, MD Professor of Medicine (Cardiology) and Health Evidence Policy Director of Interventional Cardiovascular Research and Clinical Trials The Icahn School of Medicine at Mount Sinai, New York, NY Chief Scientific Officer Cardiovascular Research Foundation, New York, NY

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. These relationships may lead to bias in my presentation. Affiliation/Financial Relationship Company • Grant/Research Support • The Medicines Co. , BMS, • Advisory Board • Janssen (J+J), • Consulting Fees/Honoraria • Abbott Vascular, (Institutional) Astra Zeneca, Lilly/Daiichi Sankyo Astra. Zeneca, Boston Scientific, Covidien, CSL Behring, Janssen (J+J), Merck,

Adjunct Pharmacotherapy for PCI ¡ Antiplatelet agents ¡ Anticoagulants • Unfractionated heparin • Enoxaparin • Bivalirudin • Aspirin • Thienopyridines • Cyclo-pentyl-triazolopyrimidines • Fondaparinux • Warfarin • Dabigatran • GP IIb/IIIa inhibitors • Cilastozol • Rivaroxaban • Apixaban ¡ Thrombolytic therapy ¡ Optimal medical therapy ¡ Adenosine • Beta blockers ¡ Vasodilators • ACE inhibitors ¡ Pressors • ARBs ¡ Agents to prevent RCN • Statins
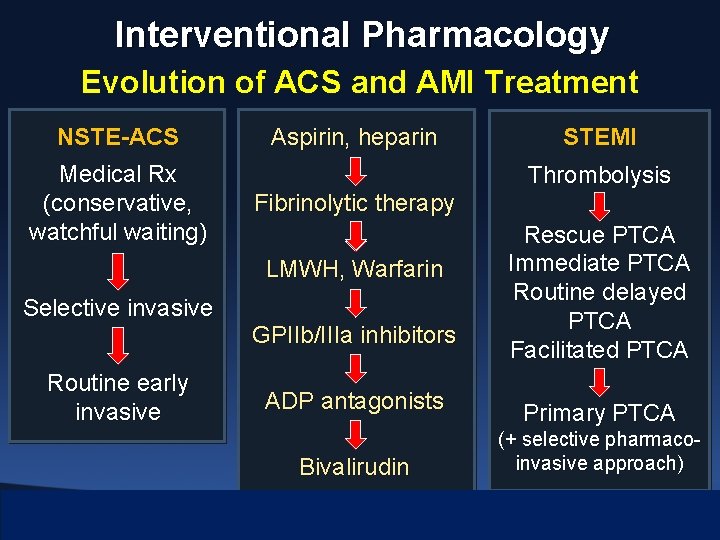
Interventional Pharmacology Evolution of ACS and AMI Treatment NSTE-ACS Medical Rx (conservative, watchful waiting) Aspirin, heparin Thrombolysis Fibrinolytic therapy LMWH, Warfarin Selective invasive GPIIb/IIIa inhibitors Routine early invasive STEMI ADP antagonists Bivalirudin Rescue PTCA Immediate PTCA Routine delayed PTCA Facilitated PTCA Primary PTCA (+ selective pharmacoinvasive approach)

Glycoprotein IIb/IIIa Inhibitors • Inhibit final common pathway of platelet aggregation • Achieve >80 -90% platelet inhibition within 15 min of administration • Effective in preventing periprocedural ischemic complications with added to heparin for elective and urgent PCI ¡ EPISTENT, ESPRIT, PRISM-PLUS, PURSUIT trials • Enhance vessel patency and reduces ischemic complications when added to thrombolytic therapy in STEMI ¡ TIMI 14, GUSTO V, ASSENT 3 trials
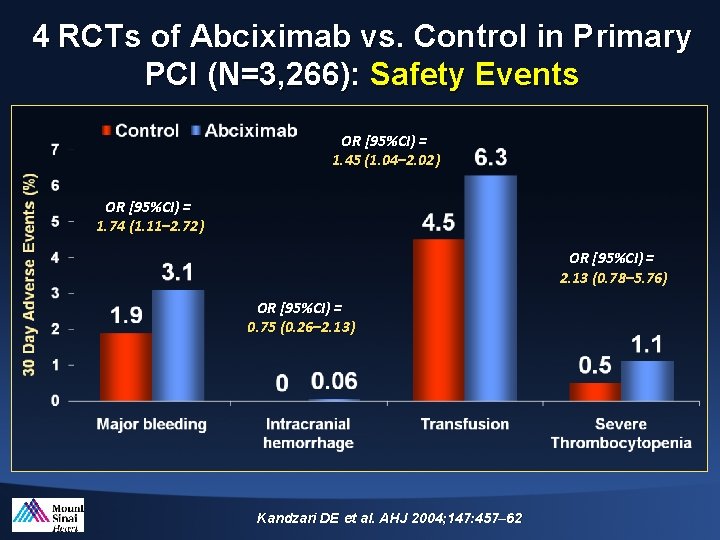
4 RCTs of Abciximab vs. Control in Primary PCI (N=3, 266): Safety Events OR [95%CI) = 1. 45 (1. 04– 2. 02) OR [95%CI) = 1. 74 (1. 11– 2. 72) OR [95%CI) = 2. 13 (0. 78– 5. 76) OR [95%CI) = 0. 75 (0. 26– 2. 13) Kandzari DE et al. AHJ 2004; 147: 457– 62
Antithrombin Choices for PCI, ACS and AMI LMW Heparin Unfractionated heparin Fondaparinux Bivalirudin

Limitations of Heparins Attribute UFH Enox Impact 30– 35% 40– 60% Unpredictable Action independent of AT No No Unpredictable Non-specific protease binding Yes Partial Unpredictable Variable PK-PD Yes Less Unpredictable Inhibits fibrin-bound thrombin No No Need dose Activates/aggregates platelets Yes +/- Need IIb/IIIa 60– 90’ 270’ Bleeding Yes Reduced Very bad Active moieties in substance T 0. 5 in minutes PF-4 complexing & risk of HIT
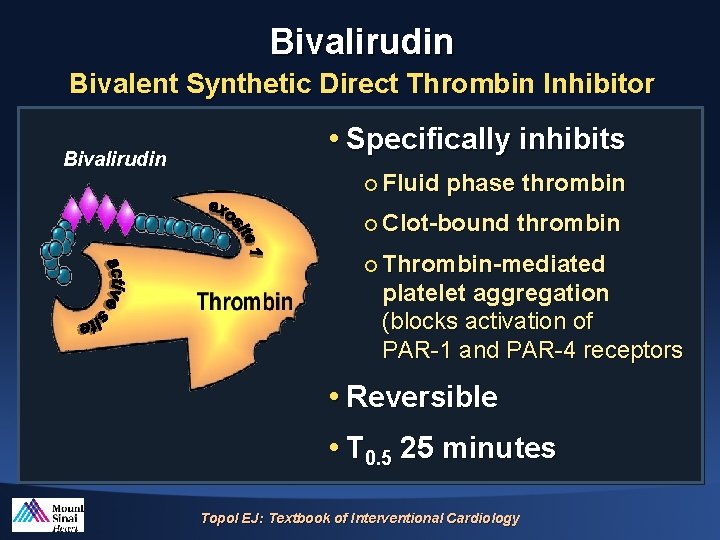
Bivalirudin Bivalent Synthetic Direct Thrombin Inhibitor Bivalirudin • Specifically inhibits ¡ Fluid phase thrombin ¡ Clot-bound thrombin ¡ Thrombin-mediated platelet aggregation (blocks activation of PAR-1 and PAR-4 receptors • Reversible • T 0. 5 25 minutes Topol EJ: Textbook of Interventional Cardiology

Large-scale Bivalirudin RCTs in ACS NSTEACS RCTs ACUITY (n=13, 819) ISAR-REACT 4 (n=1721) STEMI RCTs HORIZONS-AMI (n=3602) EUROMAX (n=2198) BRIGHT (n=2194) HEAT PPCI (n=1812) STEMI + NSTEMI RCTs MATRIX (n=7213) multicenter --- single center --- multicenter
NSTEACS: Pathophysiology Ruptured plaque with sub-occlusive thrombus
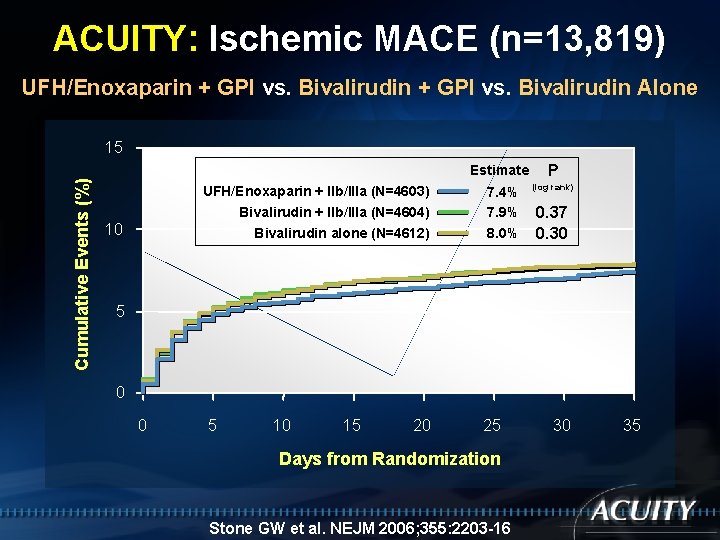
ACUITY: Ischemic MACE (n=13, 819) UFH/Enoxaparin + GPI vs. Bivalirudin Alone Cumulative Events (%) 15 Estimate UFH/Enoxaparin + IIb/IIIa (N=4603) Bivalirudin + IIb/IIIa (N=4604) Bivalirudin alone (N=4612) 10 7. 4% 7. 9% 8. 0% P (log rank) 0. 37 0. 30 5 0 0 5 10 15 20 25 Days from Randomization Stone GW et al. NEJM 2006; 355: 2203 -16 30 35
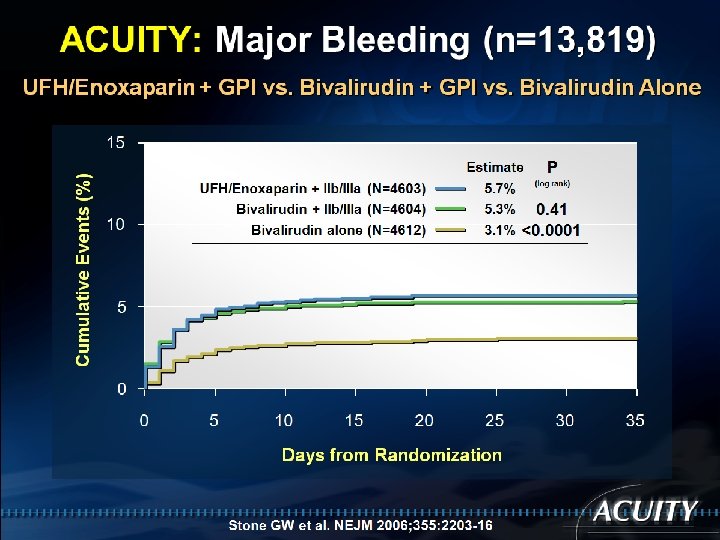
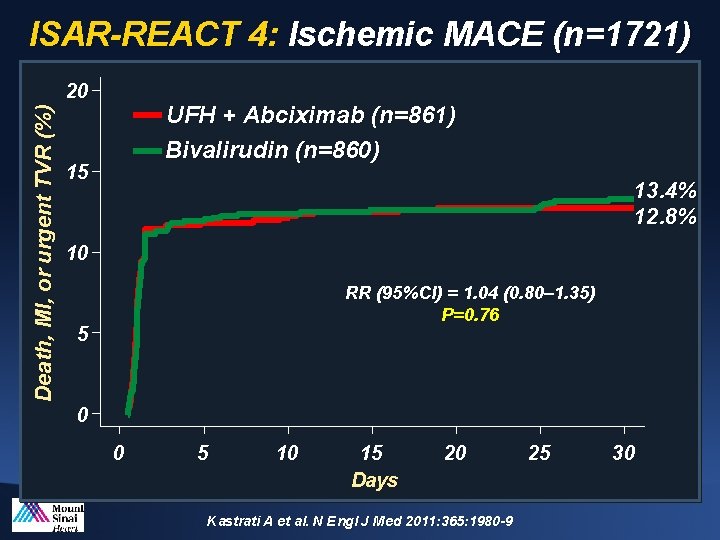
ISAR-REACT 4: Ischemic MACE (n=1721) Death, MI, or urgent TVR (%) 20 UFH + Abciximab (n=861) Bivalirudin (n=860) 15 13. 4% 12. 8% 10 RR (95%CI) = 1. 04 (0. 80– 1. 35) P=0. 76 5 0 0 5 10 15 Days 20 Kastrati A et al. N Engl J Med 2011: 365: 1980 -9 25 30
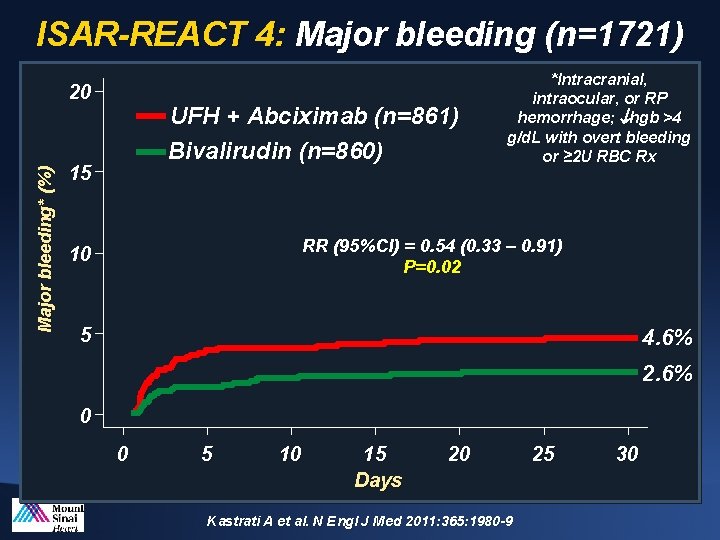
ISAR-REACT 4: Major bleeding (n=1721) Major bleeding* (%) 20 UFH + Abciximab (n=861) Bivalirudin (n=860) 15 *Intracranial, intraocular, or RP hemorrhage; hgb >4 g/d. L with overt bleeding or ≥ 2 U RBC Rx RR (95%CI) = 0. 54 (0. 33 – 0. 91) P=0. 02 10 5 4. 6% 2. 6% 0 0 5 10 15 Days 20 Kastrati A et al. N Engl J Med 2011: 365: 1980 -9 25 30
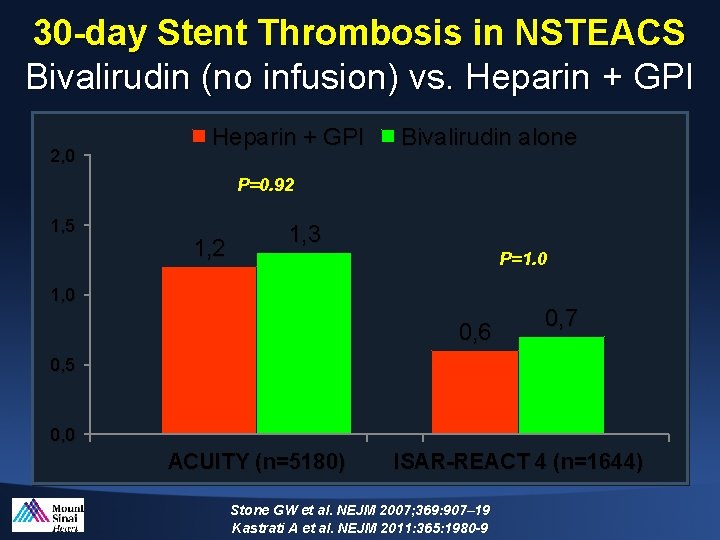
30 -day Stent Thrombosis in NSTEACS Bivalirudin (no infusion) vs. Heparin + GPI 2, 0 Heparin + GPI Bivalirudin alone P=0. 92 1, 5 1, 2 1, 3 P=1. 0 1, 0 0, 6 0, 7 0, 5 0, 0 ACUITY (n=5180) ISAR-REACT 4 (n=1644) Stone GW et al. NEJM 2007; 369: 907– 19 Kastrati A et al. NEJM 2011: 365: 1980 -9
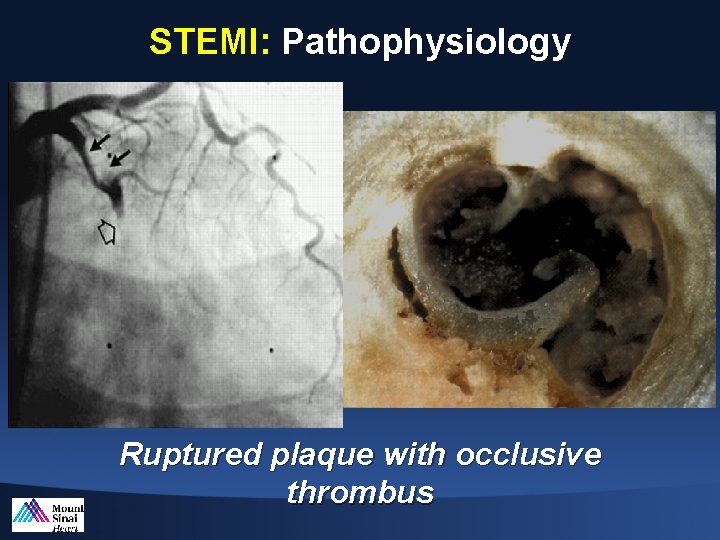
STEMI: Pathophysiology Ruptured plaque with occlusive thrombus
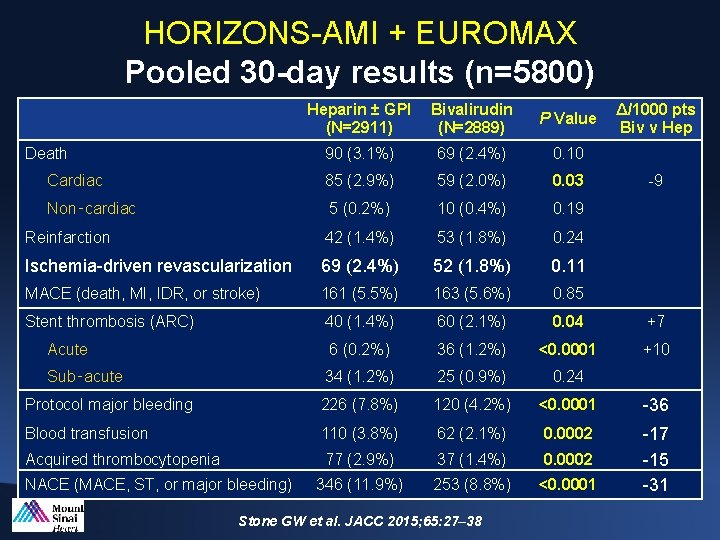
HORIZONS-AMI + EUROMAX Pooled 30 -day results (n=5800) Heparin ± GPI (N=2911) Bivalirudin (N=2889) P Value 90 (3. 1%) 69 (2. 4%) 0. 10 Cardiac 85 (2. 9%) 59 (2. 0%) 0. 03 Non‑cardiac 5 (0. 2%) 10 (0. 4%) 0. 19 Reinfarction 42 (1. 4%) 53 (1. 8%) 0. 24 Ischemia-driven revascularization 69 (2. 4%) 52 (1. 8%) 0. 11 MACE (death, MI, IDR, or stroke) 161 (5. 5%) 163 (5. 6%) 0. 85 Stent thrombosis (ARC) 40 (1. 4%) 60 (2. 1%) 0. 04 +7 Acute 6 (0. 2%) 36 (1. 2%) <0. 0001 +10 Sub‑acute 34 (1. 2%) 25 (0. 9%) 0. 24 Protocol major bleeding 226 (7. 8%) 120 (4. 2%) <0. 0001 -36 Blood transfusion 110 (3. 8%) 62 (2. 1%) 0. 0002 Acquired thrombocytopenia 77 (2. 9%) 37 (1. 4%) 0. 0002 346 (11. 9%) 253 (8. 8%) <0. 0001 -17 -15 -31 Death NACE (MACE, ST, or major bleeding) Stone GW et al. JACC 2015; 65: 27– 38 Δ/1000 pts Biv v Hep -9
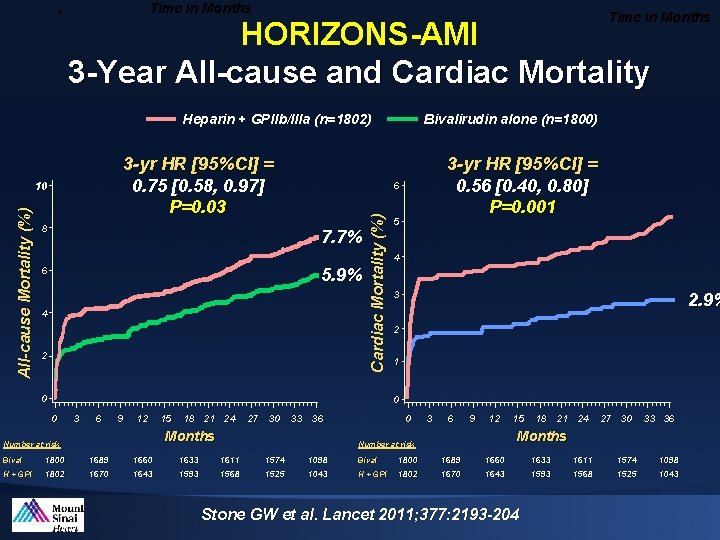
' Time in Months HORIZONS-AMI 3 -Year All-cause and Cardiac Mortality Heparin + GPIIb/IIIa (n=1802) 3 -yr HR [95%CI] = 0. 75 [0. 58, 0. 97] P=0. 03 8 7. 7% 5. 9% 6 4 2 0 3 6 9 12 1800 1802 15 18 21 24 27 30 33 36 Months Number at risk H + GPI 5 4 3 2. 9% 2 1 0 0 Bival 3 -yr HR [95%CI] = 0. 56 [0. 40, 0. 80] P=0. 001 6 Cardiac Mortality (%) All-cause Mortality (%) 10 Bivalirudin alone (n=1800) 1689 1670 1660 1643 1633 1593 0 3 6 9 12 1574 1525 1098 1043 Bival H + GPI 1800 1802 18 21 24 27 30 33 36 Months Number at risk 1611 1568 15 1689 1670 1660 1643 Stone GW et al. Lancet 2011; 377: 2193 -204 1633 1593 1611 1568 1574 1525 1098 1043
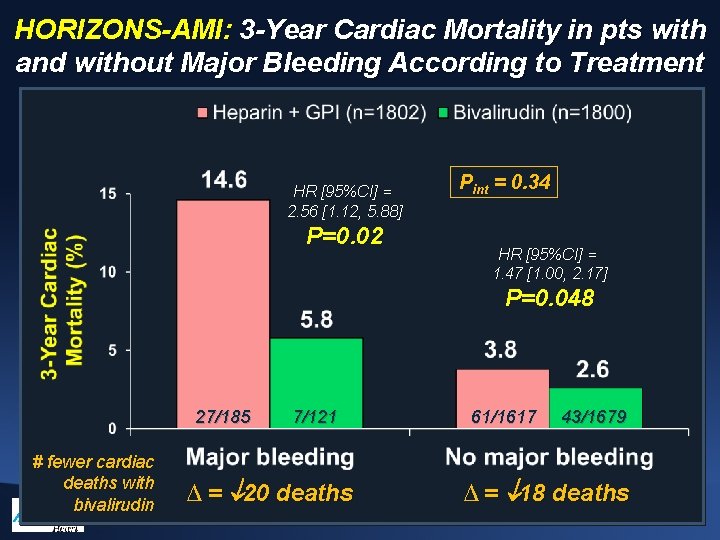
HORIZONS-AMI: 3 -Year Cardiac Mortality in pts with and without Major Bleeding According to Treatment HR [95%CI] = 2. 56 [1. 12, 5. 88] P=0. 02 Pint = 0. 34 HR [95%CI] = 1. 47 [1. 00, 2. 17] P=0. 048 27/185 # fewer cardiac deaths with bivalirudin 7/121 ∆ = 20 deaths 61/1617 43/1679 ∆ = 18 deaths

HEAT PPCI: Design and enrollment 1917 STEMI pts scheduled for emergency angiography at a single center between Feb 2012–Nov 2013* 29 (1. 5%) already randomized in the trial 59 (3. 0%) met one or more other exclusion criteria 1829 eligible for recruitment were randomized 1: 1 Assigned to Heparin* (n=914) Assigned to Bivalirudin (n=915) *70 U/kg Assigned to Heparin* (n=907) Assigned to Bivalirudin (n=905) Shahzad A et al. Lancet 2014 Exclusion Criteria • Active bleeding at presentation • Factors precluding oral DAPT • Intolerance or contraindication to trial medications • Previous enrolment in this trial 17 (1%) refused post procedure consent and were withdrawn
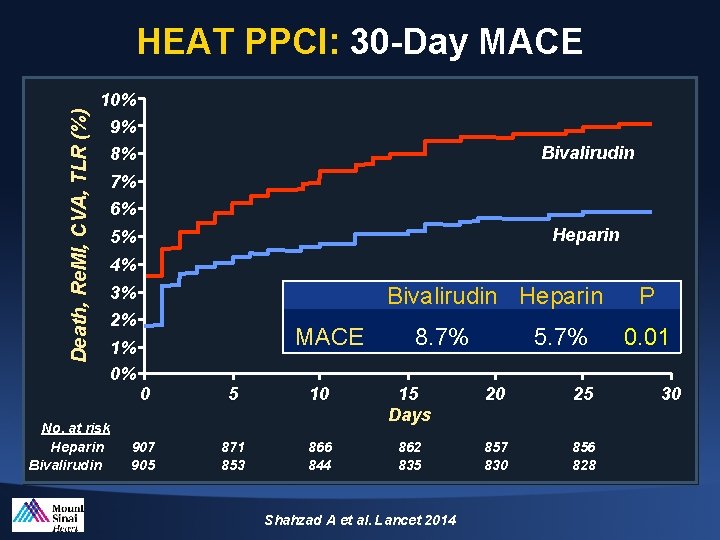
Death, Re. MI, CVA, TLR (%) HEAT PPCI: 30 -Day MACE 10% 9% Bivalirudin 8% 7% 6% Heparin 5% 4% 3% Bivalirudin Heparin 2% 1% 0% No. at risk Heparin Bivalirudin MACE 8. 7% 5. 7% 0 5 10 15 Days 20 25 907 905 871 853 866 844 862 835 857 830 856 828 Shahzad A et al. Lancet 2014 P 0. 01 30
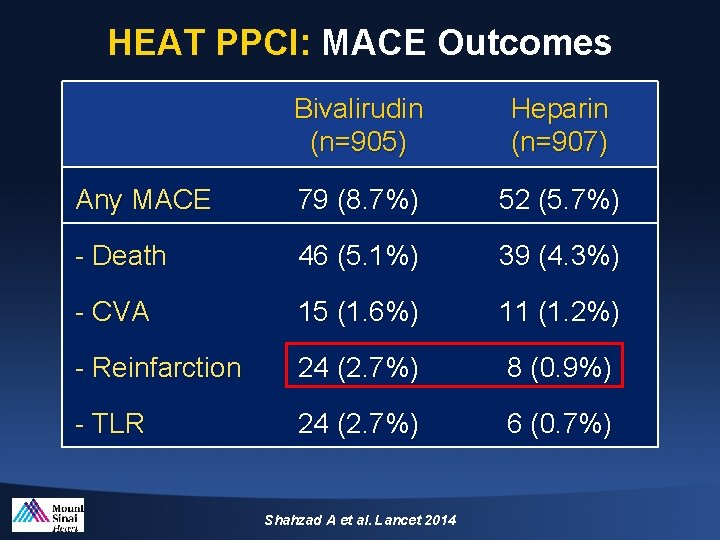
HEAT PPCI: MACE Outcomes Bivalirudin (n=905) Heparin (n=907) Any MACE 79 (8. 7%) 52 (5. 7%) - Death 46 (5. 1%) 39 (4. 3%) - CVA 15 (1. 6%) 11 (1. 2%) - Reinfarction 24 (2. 7%) 8 (0. 9%) - TLR 24 (2. 7%) 6 (0. 7%) Shahzad A et al. Lancet 2014

HEAT PPCI: Stent Thrombosis ARC definite or probable stent thrombosis Bivalirudin (n=905) Heparin (n=907) Definite or probable 24 (3. 4%) 6 (0. 9%) - Definite 23 (3. 3%) 5 (0. 7%) - Probable 1 (0. 1%) - Acute 20 (2. 9%) 6 (0. 9%) - Subacute 4 (0. 6%) 0 (0%) Shahzad A et al. Lancet 2014
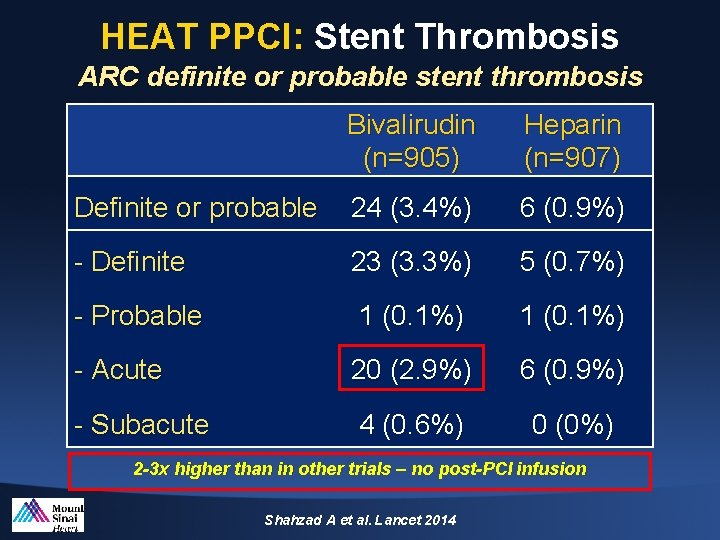
HEAT PPCI: Stent Thrombosis ARC definite or probable stent thrombosis Bivalirudin (n=905) Heparin (n=907) Definite or probable 24 (3. 4%) 6 (0. 9%) - Definite 23 (3. 3%) 5 (0. 7%) - Probable 1 (0. 1%) - Acute 20 (2. 9%) 6 (0. 9%) - Subacute 4 (0. 6%) 0 (0%) 2 -3 x higher than in other trials – no post-PCI infusion Shahzad A et al. Lancet 2014
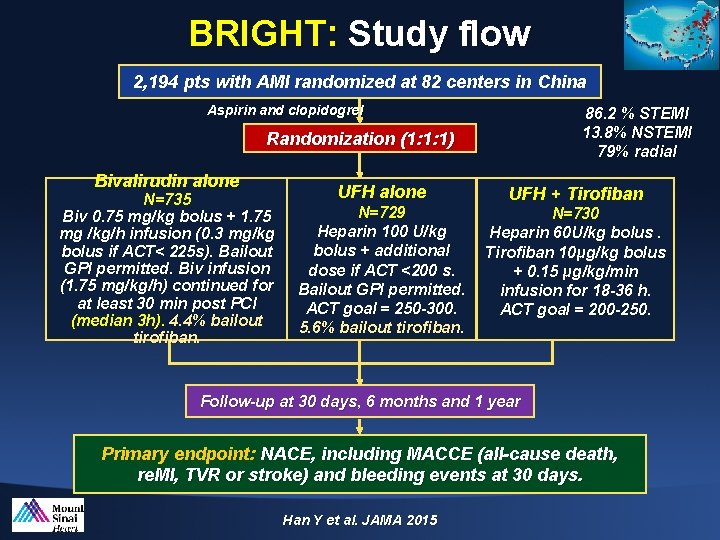
BRIGHT: Study flow 2, 194 pts with AMI randomized at 82 centers in China Aspirin and clopidogrel 86. 2 % STEMI 13. 8% NSTEMI 79% radial Randomization (1: 1: 1) Bivalirudin alone N=735 Biv 0. 75 mg/kg bolus + 1. 75 mg /kg/h infusion (0. 3 mg/kg bolus if ACT< 225 s). Bailout GPI permitted. Biv infusion (1. 75 mg/kg/h) continued for at least 30 min post PCI (median 3 h). 4. 4% bailout tirofiban. UFH alone N=729 Heparin 100 U/kg bolus + additional dose if ACT <200 s. Bailout GPI permitted. ACT goal = 250 -300. 5. 6% bailout tirofiban. UFH + Tirofiban N=730 Heparin 60 U/kg bolus. Tirofiban 10μg/kg bolus + 0. 15 μg/kg/min infusion for 18 -36 h. ACT goal = 200 -250. Follow-up at 30 days, 6 months and 1 year Primary endpoint: NACE, including MACCE (all-cause death, re. MI, TVR or stroke) and bleeding events at 30 days. Han Y et al. JAMA 2015
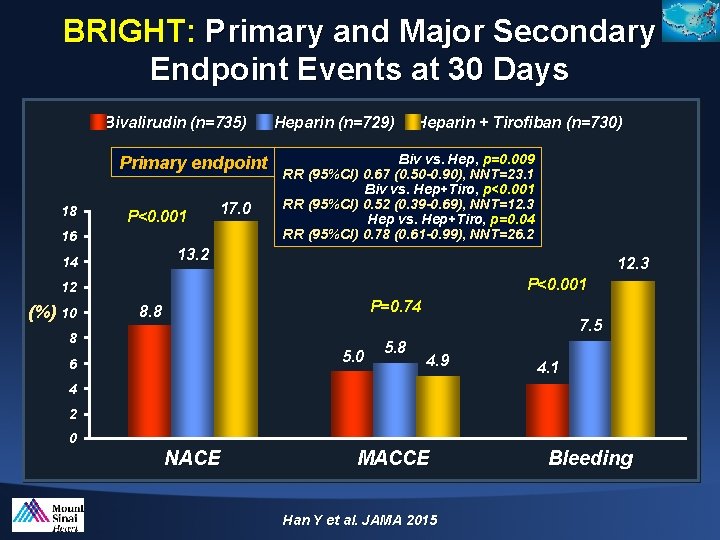
BRIGHT: Primary and Major Secondary Endpoint Events at 30 Days Bivalirudin (n=735) Primary endpoint 18 P<0. 001 17. 0 16 Heparin (n=729) Heparin + Tirofiban (n=730) Biv vs. Hep, p=0. 009 RR (95%CI) 0. 67 (0. 50 -0. 90), NNT=23. 1 Biv vs. Hep+Tiro, p<0. 001 RR (95%CI) 0. 52 (0. 39 -0. 69), NNT=12. 3 Hep vs. Hep+Tiro, p=0. 04 RR (95%CI) 0. 78 (0. 61 -0. 99), NNT=26. 2 13. 2 14 12. 3 P<0. 001 12 (%) 10 P=0. 74 8. 8 7. 5 8 5. 0 6 5. 8 4. 9 4. 1 4 2 0 NACE MACCE Han Y et al. JAMA 2015 Bleeding
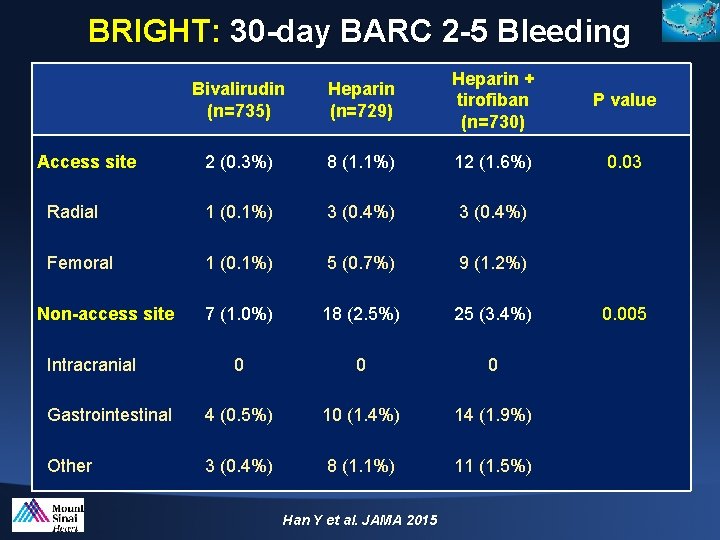
BRIGHT: 30 -day BARC 2 -5 Bleeding Bivalirudin (n=735) Heparin (n=729) Heparin + tirofiban (n=730) P value 2 (0. 3%) 8 (1. 1%) 12 (1. 6%) 0. 03 Radial 1 (0. 1%) 3 (0. 4%) Femoral 1 (0. 1%) 5 (0. 7%) 9 (1. 2%) 7 (1. 0%) 18 (2. 5%) 25 (3. 4%) 0 0 0 Gastrointestinal 4 (0. 5%) 10 (1. 4%) 14 (1. 9%) Other 3 (0. 4%) 8 (1. 1%) 11 (1. 5%) Access site Non-access site Intracranial Han Y et al. JAMA 2015 0. 005
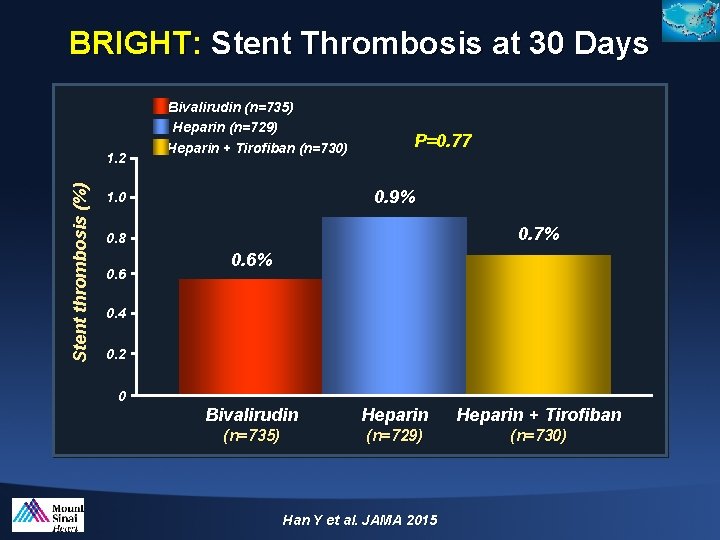
BRIGHT: Stent Thrombosis at 30 Days Stent thrombosis (%) 1. 2 Bivalirudin (n=735) Heparin (n=729) Heparin + Tirofiban (n=730) P=0. 77 0. 9% 1. 0 0. 7% 0. 8 0. 6% 0. 4 0. 2 0 Bivalirudin Heparin + Tirofiban (n=735) (n=729) (n=730) Han Y et al. JAMA 2015
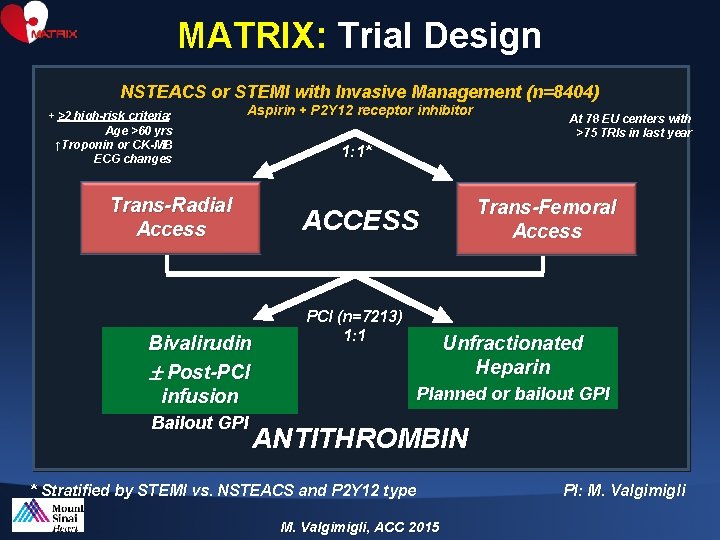
MATRIX: Trial Design NSTEACS or STEMI with Invasive Management (n=8404) + ≥ 2 high-risk criteria: Age >60 yrs ↑Troponin or CK-MB ECG changes Aspirin + P 2 Y 12 receptor inhibitor 1: 1* Trans-Radial Access Trans-Femoral Access ACCESS Bivalirudin Post-PCI infusion Bailout GPI At 78 EU centers with >75 TRIs in last year PCI (n=7213) 1: 1 Unfractionated Heparin Planned or bailout GPI ANTITHROMBIN * Stratified by STEMI vs. NSTEACS and P 2 Y 12 type M. Valgimigli, ACC 2015 PI: M. Valgimigli
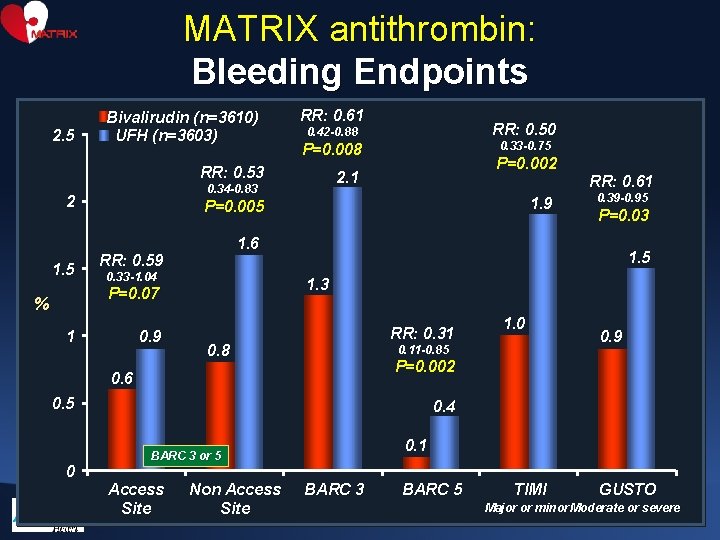
MATRIX antithrombin: Bleeding Endpoints 2. 5 Bivalirudin (n=3610) UFH (n=3603) RR: 0. 61 1. 5 P=0. 002 2. 1 0. 34 -0. 83 1. 9 P=0. 005 RR: 0. 59 0. 33 -1. 04 1 0. 9 0. 39 -0. 95 P=0. 03 1. 5 1. 3 RR: 0. 31 0. 8 1. 0 0. 11 -0. 85 0. 9 P=0. 002 0. 6 0. 5 0 RR: 0. 61 1. 6 P=0. 07 % 0. 33 -0. 75 P=0. 008 RR: 0. 53 2 RR: 0. 50 0. 42 -0. 88 0. 4 0. 1 BARC 3 or 5 Access Site Non Access Site BARC 3 BARC 5 TIMI GUSTO Major or minor Moderate or severe
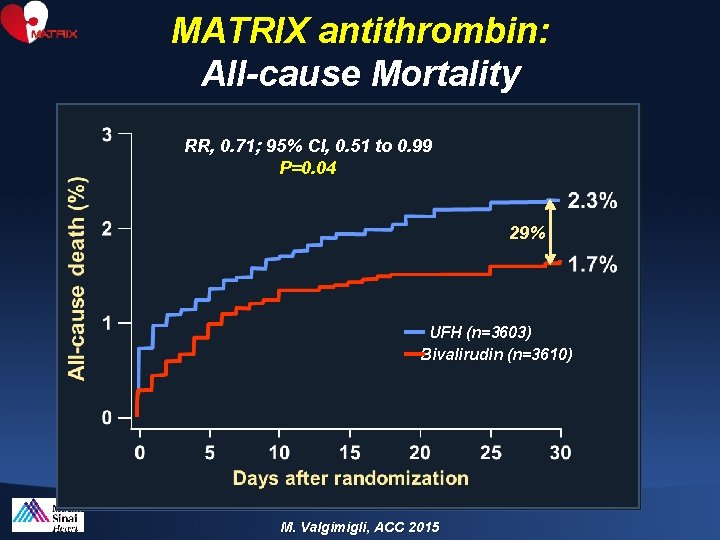
MATRIX antithrombin: All-cause Mortality RR, 0. 71; 95% CI, 0. 51 to 0. 99 P=0. 04 29% UFH (n=3603) Bivalirudin (n=3610) M. Valgimigli, ACC 2015

MATRIX antithrombin: Access Site Subgroup Analysis Hazard Ratio (95% CI) HR (95% CI) Biv vs. UFH P value for interaction Death Femoral 0. 66 (0. 42 -1. 03) Radial 0. 77 (0. 47 -1. 28) 0. 64 Bleeding Femoral 0. 50 (0. 32 -0. 79) Radial 0. 64 (0. 37 -1. 10) 2 UFH Better 1 0. 25 Bivalirudin Better M. Valgimigli, ACC 2015 0. 51
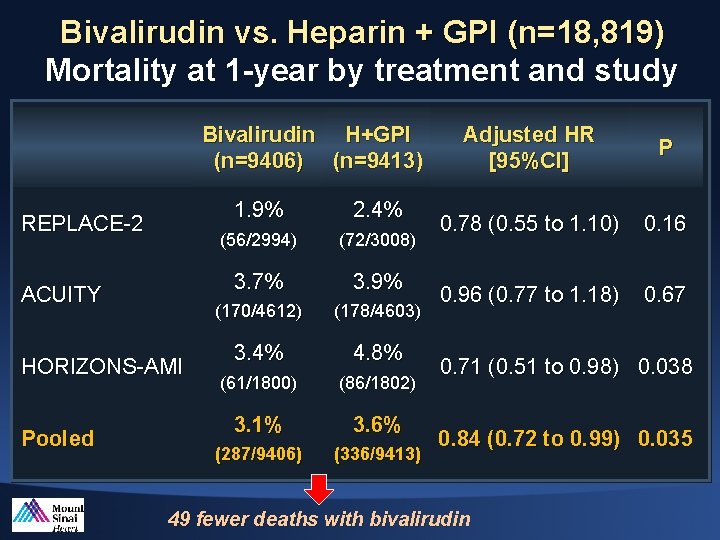
Bivalirudin vs. Heparin + GPI (n=18, 819) Mortality at 1 -year by treatment and study Bivalirudin H+GPI (n=9406) (n=9413) REPLACE-2 ACUITY HORIZONS-AMI Pooled 1. 9% 2. 4% (56/2994) (72/3008) 3. 7% 3. 9% (170/4612) (178/4603) 3. 4% 4. 8% (61/1800) (86/1802) 3. 1% 3. 6% (287/9406) (336/9413) Adjusted HR [95%CI] P 0. 78 (0. 55 to 1. 10) 0. 16 0. 96 (0. 77 to 1. 18) 0. 67 0. 71 (0. 51 to 0. 98) 0. 038 0. 84 (0. 72 to 0. 99) 0. 035 49 fewer deaths with bivalirudin
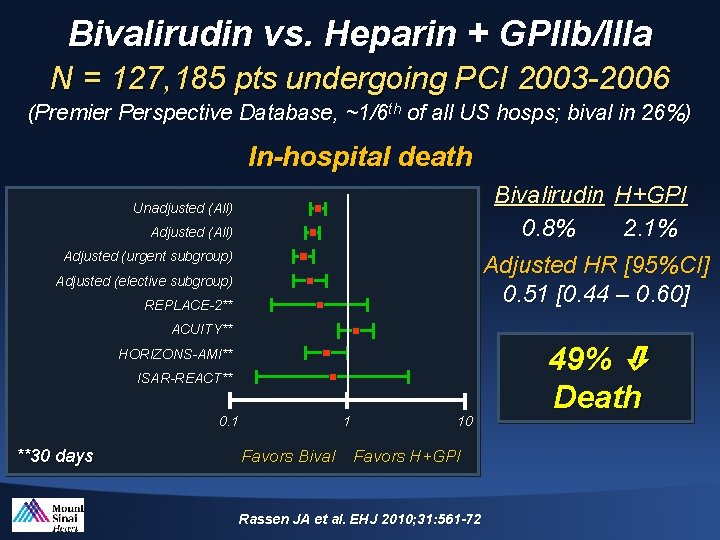
Bivalirudin vs. Heparin + GPIIb/IIIa N = 127, 185 pts undergoing PCI 2003 -2006 (Premier Perspective Database, ~1/6 th of all US hosps; bival in 26%) In-hospital death Bivalirudin H+GPI 0. 8% 2. 1% Unadjusted (All) Adjusted (urgent subgroup) Adjusted HR [95%CI] 0. 51 [0. 44 – 0. 60] Adjusted (elective subgroup) REPLACE-2** ACUITY** HORIZONS-AMI** ISAR-REACT** 0. 1 **30 days 1 Favors Bival 10 Favors H+GPI Rassen JA et al. EHJ 2010; 31: 561 -72 49% Death
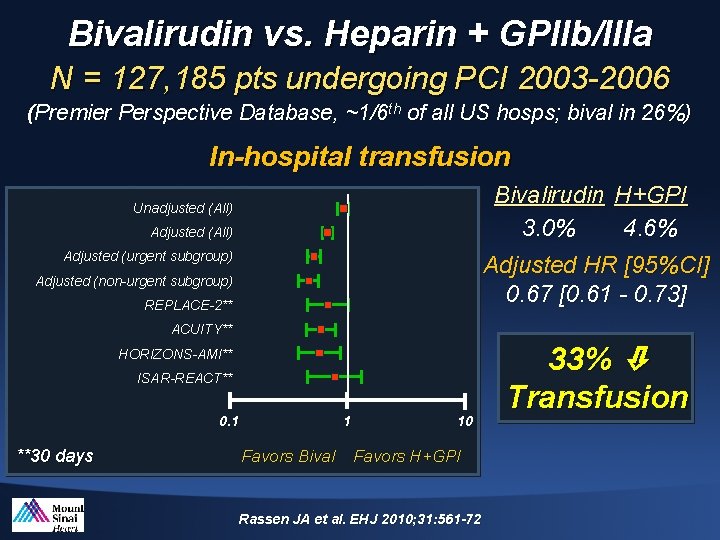
Bivalirudin vs. Heparin + GPIIb/IIIa N = 127, 185 pts undergoing PCI 2003 -2006 (Premier Perspective Database, ~1/6 th of all US hosps; bival in 26%) In-hospital transfusion Bivalirudin H+GPI 3. 0% 4. 6% Unadjusted (All) Adjusted (urgent subgroup) Adjusted HR [95%CI] 0. 67 [0. 61 - 0. 73] Adjusted (non-urgent subgroup) REPLACE-2** ACUITY** HORIZONS-AMI** ISAR-REACT** 0. 1 **30 days 1 Favors Bival 10 Favors H+GPI Rassen JA et al. EHJ 2010; 31: 561 -72 33% Transfusion

Conclusions • Randomized Clinical Trials incorporating Bivalirudin in PPCI have included over 10, 000 patients. • Most have shown a consistent benefit of reduced bleeding, increased early ST, trend toward lower mortality, and reduction in overall NACE • Antithrombotic/antiplatelet regimens, as well as access site (more radial) have evolved and evidence continues to accumulate regarding the importance of bleeding avoidance • Heat PPCI did show contradictory results to current evidence in a single center. This should be studied in a larger prospective study with Bival v. heparin mono-therapy • Until then, the case for Bivalirudin as the antithrombotic choice for PPCI (with close attention to ACT, prolonged infusion to avoid acute ST) remains.
- Slides: 37