CRT 2015 Washington DC February 22 nd 2015

CRT 2015 Washington DC, February 22 nd, 2015 Protection Devices for Stroke Prevention Does the Data Support Routine Use? Eberhard Grube MD, FACC, FSCAI University Hospital, Dept of Medicine II, Bonn, Germany Stanford University, Palo Alto, California, USA CRT 2015

Eberhard Grube, MD Physician Name Company/Relationship Eberhard Grube, MD Medtronic, Core. Valve: C, SB, AB, OF Direct Flow: C, SB, AB Mitralign: AB, SB, E Boston Scientific: C, SB, AB Biosensors: E, SB, C, AB Cordis: AB Abbott Vascular: AB In. Seal Medical: AB, E, Valtech: E, SB, Claret: SB Keystone: AB Shockwave: E, AB Key G – Grant and or Research Support E – Equity Interests S – Salary, AB – Advisory Board C – Consulting fees, Honoraria R – Royalty Income I – Intellectual Property Rights SB – Speaker’s Bureau O – Ownership OF – Other Financial Benefits‘ CRT 2015

Let Me Lay out my Position in Advance. 1. I am a TRUE believer in TAVI and its role in treating AS in defined patient groups 2. However, TAVI undoubtedly causes embolic showers to the Brain, which cause lesions: some ‘silent’, some more clinically obvious 3. Many TAVI studies under-report Stroke & few employ independent neurologists 4. Percentages do not always tell the whole story and Stroke is truly devastating 5. If cerebral protection can reduce or eliminate lesions in the brain, then it should become routine practice CRT 2015
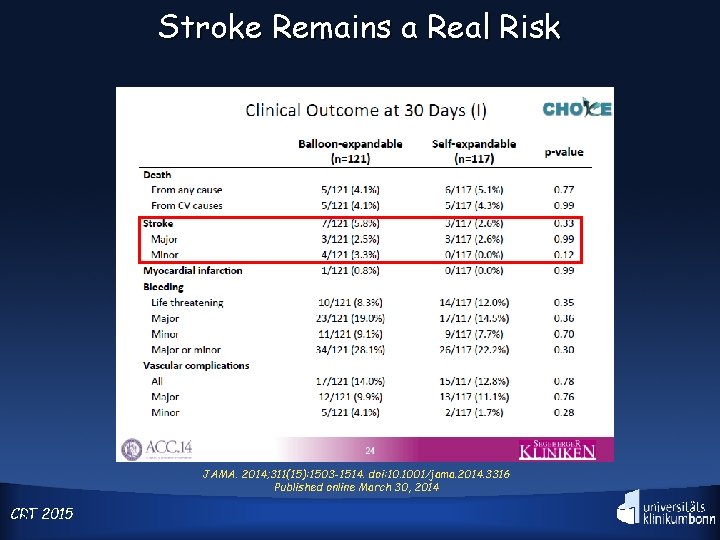
Stroke Remains a Real Risk JAMA. 2014; 311(15): 1503 -1514. doi: 10. 1001/jama. 2014. 3316 Published online March 30, 2014 CRT 3 2015
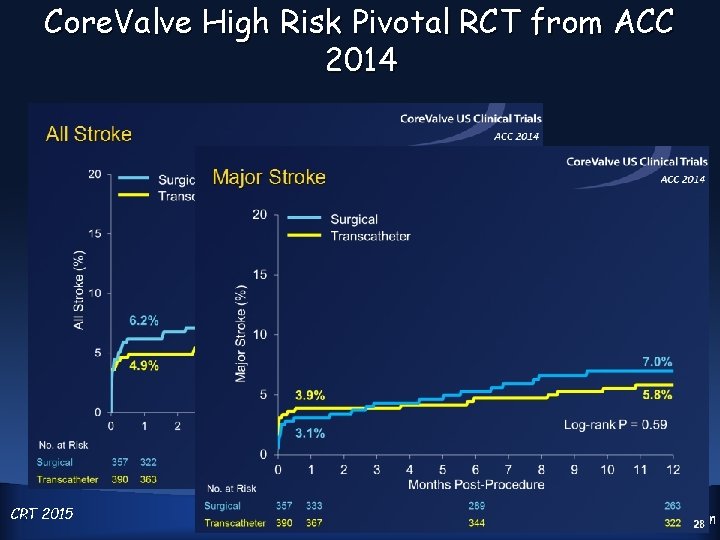
Core. Valve High Risk Pivotal RCT from ACC 2014 CRT 4 2015
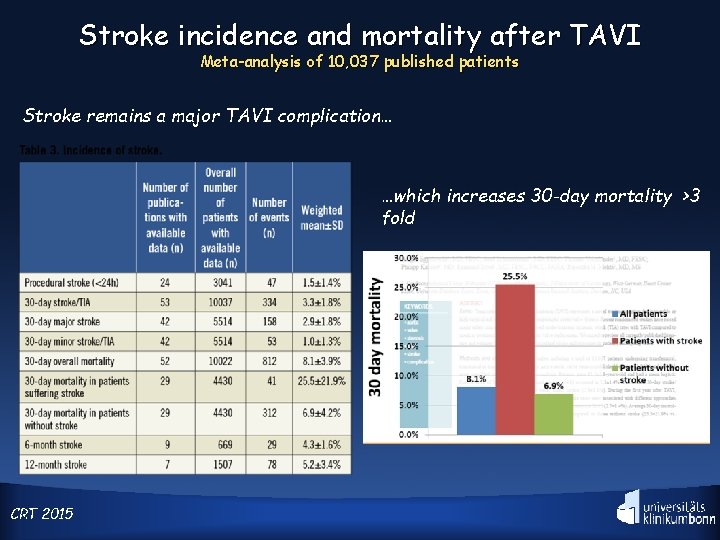
Stroke incidence and mortality after TAVI Meta-analysis of 10, 037 published patients Stroke remains a major TAVI complication… …which increases 30 -day mortality >3 fold CRT 5 2015
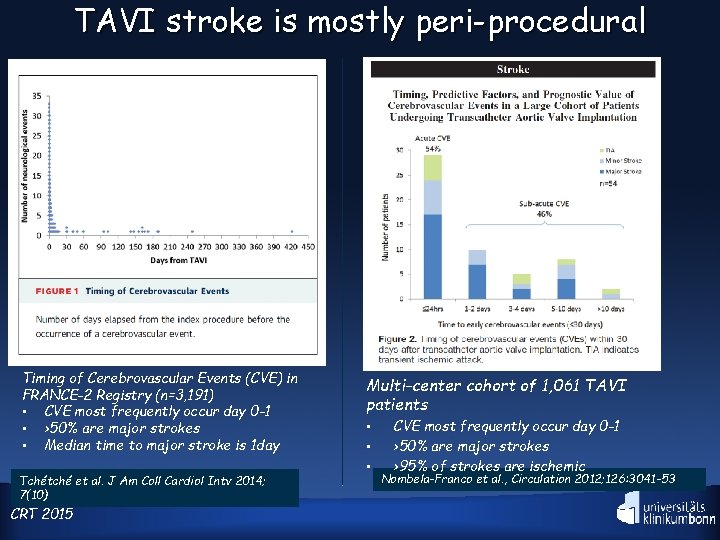
TAVI stroke is mostly peri-procedural Timing of Cerebrovascular Events (CVE) in FRANCE-2 Registry (n=3, 191) • CVE most frequently occur day 0 -1 • >50% are major strokes • Median time to major stroke is 1 day Tchétché et al. J Am Coll Cardiol Intv 2014; 7(10) CRT 2015 Multi-center cohort of 1, 061 TAVI patients • • • CVE most frequently occur day 0 -1 >50% are major strokes >95% of strokes are ischemic Nombela-Franco et al. , Circulation 2012; 126: 3041 -53
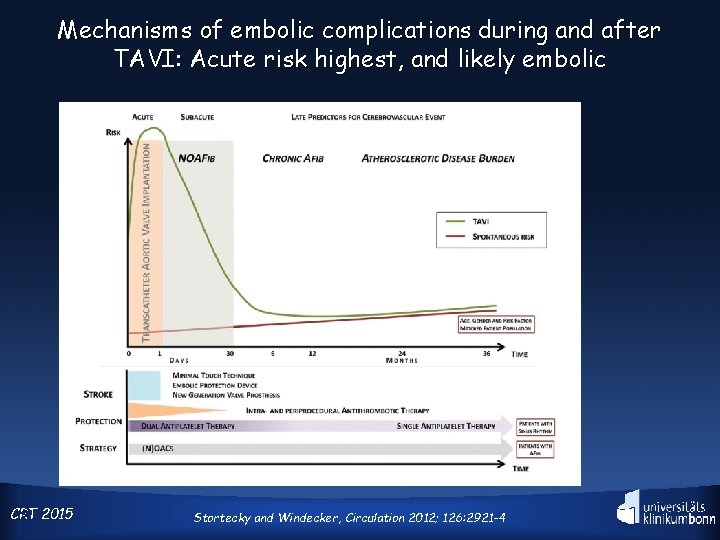
Mechanisms of embolic complications during and after TAVI: Acute risk highest, and likely embolic CRT 9 2015 Stortecky and Windecker, Circulation 2012; 126: 2921 -4
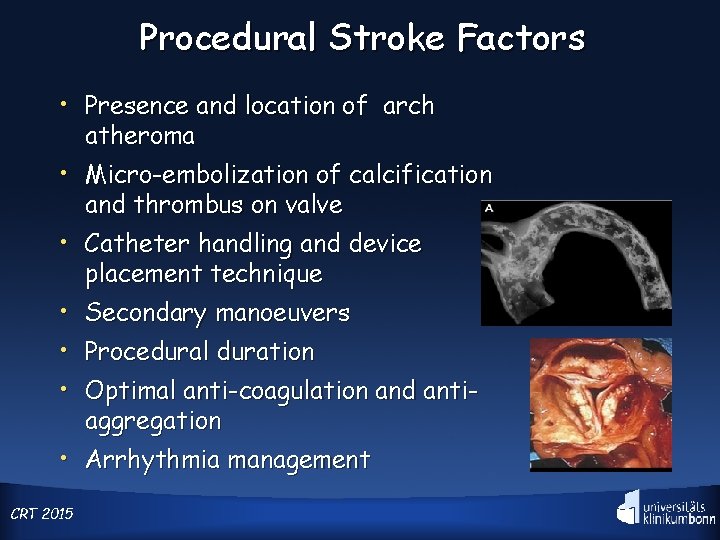
Procedural Stroke Factors • Presence and location of arch atheroma • Micro-embolization of calcification and thrombus on valve • Catheter handling and device • • • placement technique Secondary manoeuvers Procedural duration Optimal anti-coagulation and antiaggregation • Arrhythmia management CRT 2015
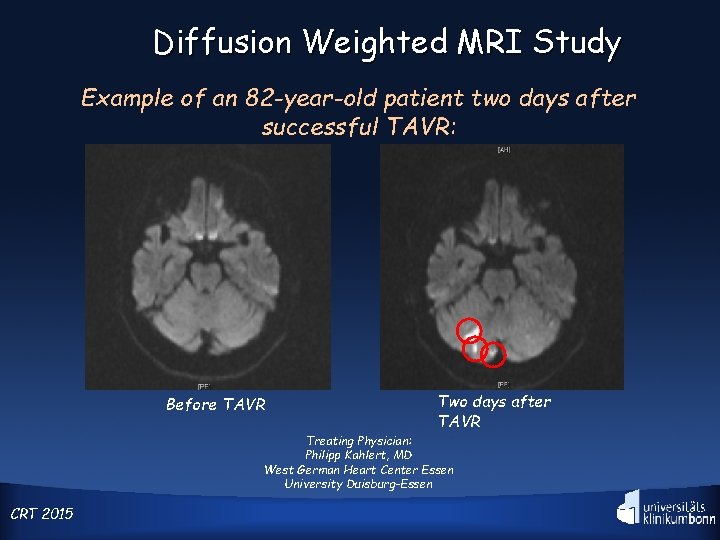
Diffusion Weighted MRI Study Example of an 82 -year-old patient two days after successful TAVR: Before TAVR Two days after TAVR Treating Physician: Philipp Kahlert, MD West German Heart Center Essen University Duisburg-Essen CRT 2015
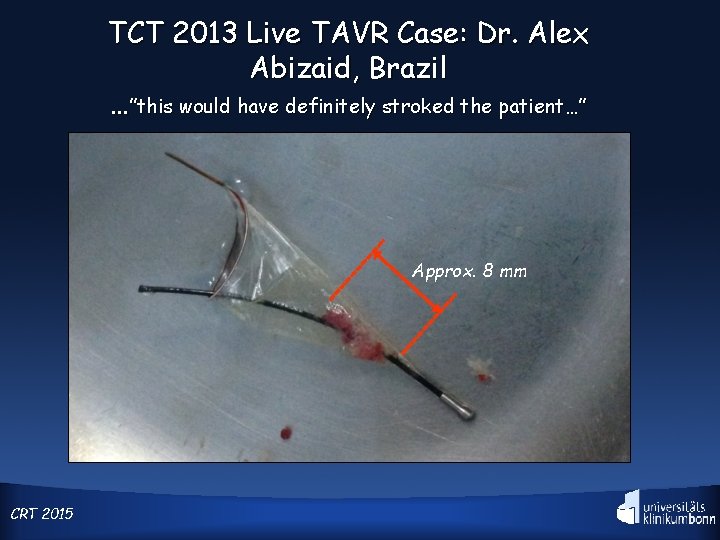
TCT 2013 Live TAVR Case: Dr. Alex Abizaid, Brazil …”this would have definitely stroked the patient…” Approx. 8 mm CRT 2015

Clinical Need—Stroke, Silent and Apparent A closer look at the patients reveals a looming risk : >70% of TAVR patients have ischemic brain lesions when examined by DW-MRI CRT 2015
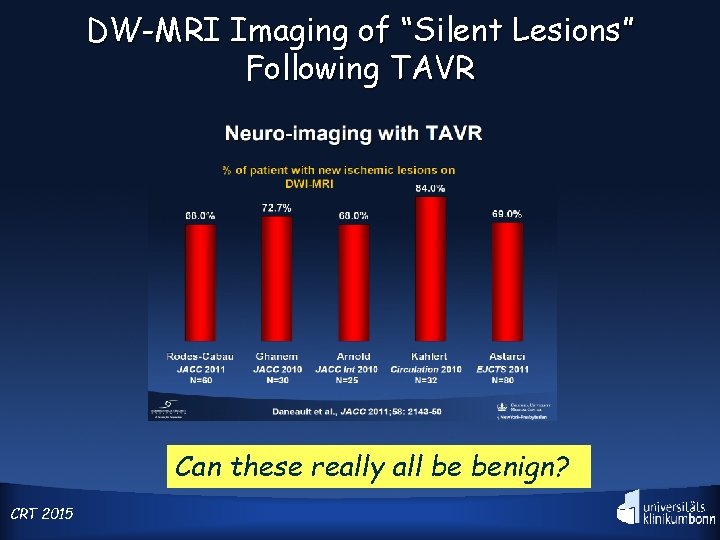
DW-MRI Imaging of “Silent Lesions” Following TAVR Can these really all be benign? CRT 2015

TAVI Expanding to Healthier Patients CRT 2015

Second Generation ‘Repositionable’ TAVR Devices Still Require Finesse All patients were assessed by a neurologist before and after TAVR CRT 21 2015
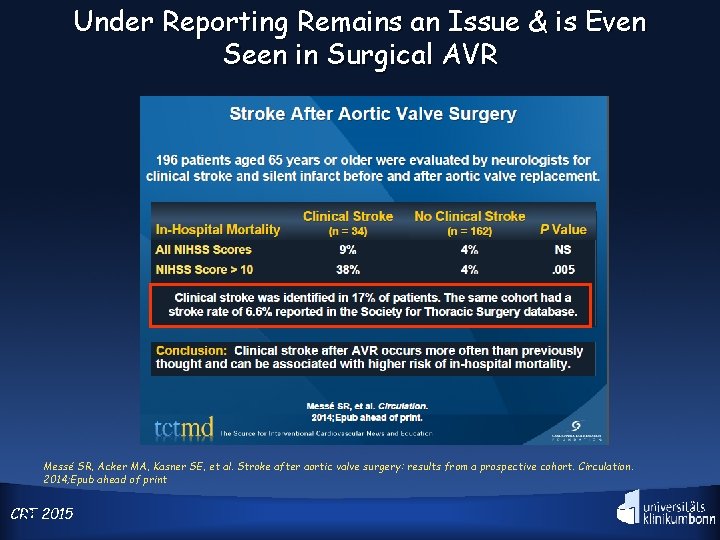
Under Reporting Remains an Issue & is Even Seen in Surgical AVR Messé SR, Acker MA, Kasner SE, et al. Stroke after aortic valve surgery: results from a prospective cohort. Circulation. 2014; Epub ahead of print CRT 22 2015
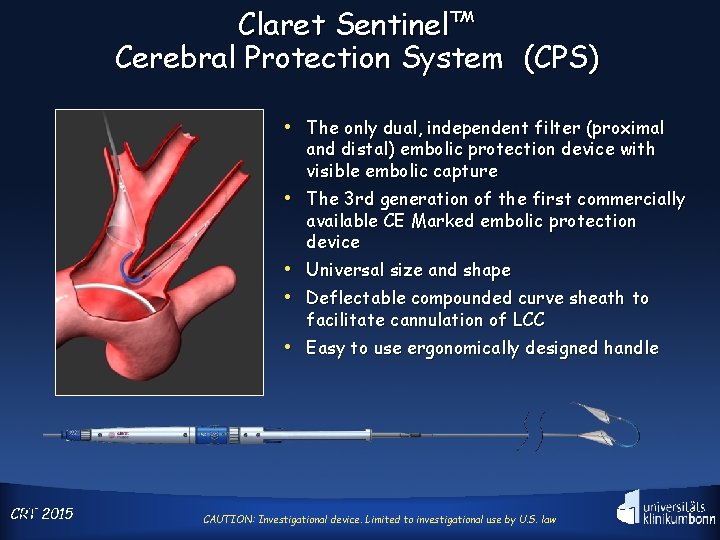
Claret Sentinel™ Cerebral Protection System (CPS) • The only dual, independent filter (proximal and distal) embolic protection device with visible embolic capture • The 3 rd generation of the first commercially available CE Marked embolic protection device • Universal size and shape • Deflectable compounded curve sheath to facilitate cannulation of LCC • Easy to use ergonomically designed handle 25 2015 CRT CAUTION: Investigational device. Limited to investigational use by U. S. law
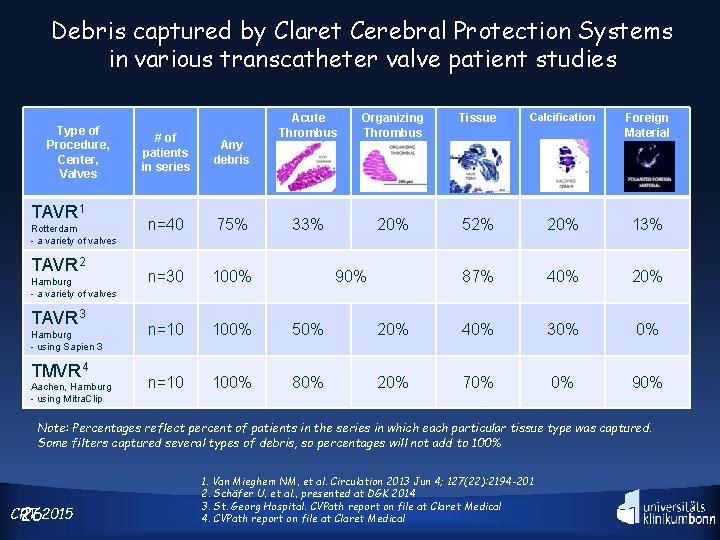
Debris captured by Claret Cerebral Protection Systems in various transcatheter valve patient studies Type of Procedure, Center, Valves TAVR 1 Rotterdam - a variety of valves TAVR 2 Hamburg - a variety of valves TAVR 3 Hamburg - using Sapien 3 TMVR 4 Aachen, Hamburg - using Mitra. Clip Acute Thrombus Organizing Thrombus Tissue Calcification Foreign Material 33% 20% 52% 20% 13% 87% 40% 20% # of patients in series Any debris n=40 75% n=30 100% n=10 100% 50% 20% 40% 30% 0% n=10 100% 80% 20% 70% 0% 90% Note: Percentages reflect percent of patients in the series in which each particular tissue type was captured. Some filters captured several types of debris, so percentages will not add to 100% CRT 262015 1. Van Mieghem NM, et al. Circulation 2013 Jun 4; 127(22): 2194 -201 2. Schäfer U, et al. , presented at DGK 2014 3. St. Georg Hospital. CVPath report on file at Claret Medical 4. CVPath report on file at Claret Medical

Cerebral Protection Reduces Periprocedural Strokes During Carotid Angioplasty & Stenting Why should this be different in TAVR? Garg et al: J Endovasc Ther. 2009; 16: 412 -427 CRT 2015

The world’s first RCT of cerebral embolic protection in TAVI was presented in the opening session of TCT ‘ 14 late-breaking clinical trials 28 2015 CRT

CLEAN-TAVI shows positive outcome Claret Montage Cerebral Protection System significantly reduces new cerebral lesion number and volume at 2 & 7 days, as measured by DW-MRI. CRT 292015
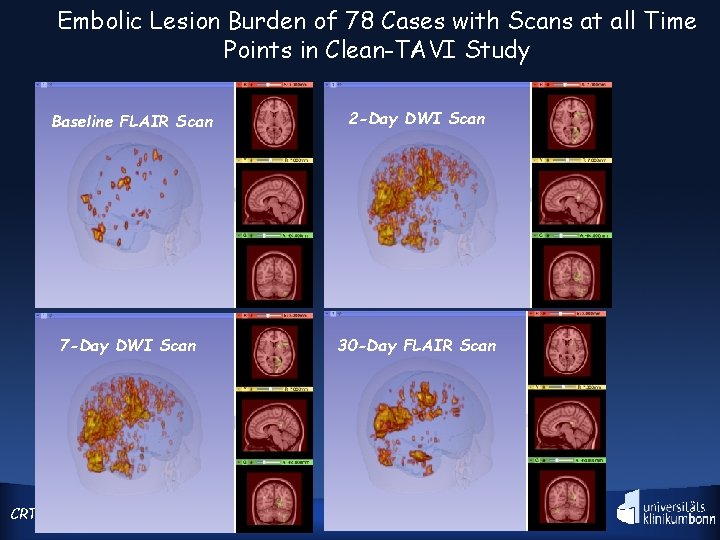
Embolic Lesion Burden of 78 Cases with Scans at all Time Points in Clean-TAVI Study Baseline FLAIR Scan 2 -Day DWI Scan 7 -Day DWI Scan 30 -Day FLAIR Scan CRT 2015

CLEAN-TAVI sets the stage for SENTINEL study SENTINEL is a US multi-center randomized trial of the Claret Sentinel. TM Cerebral Protection System in TAVR. CRT 322015

Sentinel IDE Study – Study Sites Study Site Principal Investigator Columbia University Medical Center Susheel Kodali, MD (Study Co-PI) Cleveland Clinic Samir Kapadia, MD (Study Co-PI) Morton Plant Hospital Joshua Rovin, MD Emory University Hospital Vinod Thourani, MD University of Texas Health Science Center Richard Smalling, MD Hospital of the University of Pennsylvania Joseph Bavaria, MD Washington Hospital Center Gus Pichard, MD Cedars-Sinai Health Systems Raj Makkar, MD Henry Ford Heart and Vascular Institute Mayra Guerrero, MD University of Washington Medical Center Mark Reisman, MD NY Presbyterian - Weill Cornell Medical Center S. Chiu Wong, MD University of Virginia Medical Center D. Scott Lim, MD Barnes Jewish Hospital/Washington University Alan Zajarias, MD Mount Sinai Samir Sharma, MD CRT 35 2015
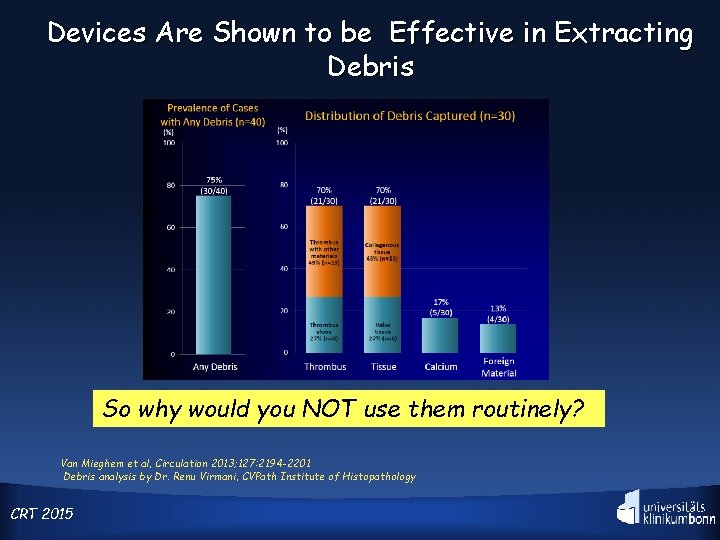
Devices Are Shown to be Effective in Extracting Debris So why would you NOT use them routinely? Van Mieghem et al, Circulation 2013; 127: 2194 -2201 Debris analysis by Dr. Renu Virmani, CVPath Institute of Histopathology CRT 2015

Accepted Wisdoms are Sometimes Flawed… In-Stent Restenosis is NOT Benign Neither are ‘silent’ lesions in the brain CRT 2015 Abu-Own et al: doi: 10. 1136/heartjnl-2011 -300198. 36

My Conclusion • • Devices are Improving Procedural Techniques are improving Patient Selection is Improving Adjunctive Pharma Therapy is improving BUT: Embolic Stroke still remains an issue in TAVR and is truly devastating!!! Cerebral Protection will (and should) become Standard of Care CRT 2015
- Slides: 27