CRT 2014 Washington DC Feb 22 25 2014

CRT 2014 Washington DC, Feb 22 -25, 2014 Renal Denervation with a Percutaneous Bipolar Radiofrequency Balloon Catheter in Patients with Resistant Hypertension: 6 -Month Efficacy and Safety Results from the REDUCE-HTN Trial Horst Sievert On behalf of the REDUCE-HTN Investigators Cardio. Vascular Center Frankfurt CVC Frankfurt, Germany

Horst Sievert, MD Consulting: Access. Closure, Inc. , AGA Medical Corporation, Ardian, Inc. , Arstasis, Inc. , Atritech Atrium Medical Corporation, Avinger, Inc. , Bard Peripheral Vascular, Inc. , Boston Scientific Corporation, Bridgepoint, Cardio. Kinetix Inc. , Cardio. MEMS, Inc. , Coherex, Inc. , Contego, CSI, Endo. Cross, Endotex Interventional Systems, Epitek, Evalve, Inc. , ev 3, Inc.

Horst Sievert, MD Consulting: Flow. Cardia, Inc. , Gore, Guidant, Lumen Biomedical, Inc. , HLT, Kensey Nash Corporation, Kyoto Medical, Lifetech, Lutonix, Inc. , Medinol, Medtronic, Inc. , NDCNMT Medical, Inc. , OAS, Occlutech Osprey Medical, Inc. , Ovalis, Inc. , Pathway Medical Technologies, Inc. , Pendra. Care International B. V. , Pfm Medical, Inc. , Rox Medical, Recor, Sadra Medical, Sorin Biomedica Cardio S. R. L. , and Spectranetics Corporation, Trireme, Trivascular, Viacor, Inc.

Horst Sievert, MD Honoraria: Viacor, Inc. , HLT, Lifetech, Recor, Trivascular, Veryan, CVRx, GDS, In. Seal Medical.

Renal Denervation for Resistant Hypertension • More than 25% of adults have hypertension 1 -4 • Between 5%-30% of these patients have elevated blood pressure despite aggressive medical treatment 5, 6 • It has been shown that renal denervation reduces blood pressure in patients with resistant hypertension 5 1. Kearney et al. Lancet 2005; 365: 217 -23 2. Lloyd-Jones et al. Circulation 2010; 121: e 46–e 215 3. Wolf-Meier et al. JAMA 2003; 289: 2363– 2369 4. Sekikawa and Hayakawa. J Hum Hypertens 2004; 18: 911 -912 5. Mancia et al. J Hypertens 2013; 31(7): 1281 -1357 6. Persell. Hypertension 2011; 57(6): 1076 -1080

Vessix Renal Denervation System • Balloon-based technology • 4 - 7 mm diameters • Bipolar radiofrequency electrodes • 30 second treatment time • All electrodes are activated simultaneously • Bipolar energy delivery, ~1 Watt • Temperature-controlled algorithm ensures energy delivery at 68°C • One button operation • Approved in Europe (CE mark) and Australia
REDUCE-HTN Clinical Sites Institution Parcelsus Medical University Allgemein Affentliches Krankenhaus der Stad Linz Onze-Lieve-Vrouwziekenhuis, (OLVZ) Monash Heart Southern Health Cardio. Vasculares Centrum (CVC) Frankfurt George Pompidou Hospital The Prince Charles Hospital Cardiology Center Geneva university Hospitals Auckland City Hospital Flinders Medical Centre Erasmus Medical Center Vascular Center Berlin Main Tanus Kliniken St. Vincent's Zentrum für Gefäßmedizin Academic Medical Center Cliniques Universitaires Saint Luc Clinique Pasteur German Heart Centre Universitäres Herz- und Gefäßzentrum Hamburg Zentralklinik Bad Berka Gmb. H Royal Adelaide Hospital Mercy Angiography - Auckland Principal Investigator Prof. Dr. Uta C. Hoppe Prof. Dr. Clemens Steinwender Dr. Eric Wyffels Prof. Dr. Ian Meredith Prof. Dr. med Horst Sievert Prof. Dr. Michel Azizi Associate Prof. Darren Walters Dr. Georg Ehret Dr. Mark Webster Dr. Ajay Sinhal Dr. Joost Daemen Dr. Ralf Langhoff Prof. Dr. med. Nicolaus Reifart Dr. David Muller Prof. Dierk Scheinert Prof. Robbert-Jan de Winter Prof. dr. Alexandre Persu Prof. Jean Fajadet Prof. Dr. med. Ilka Ott Prof. Dr. med. Joachim Schofer Dr. med. Ahmed Farah Prof. Steven Worthley Dr. John Ormiston

REDUCE-HTN FIM + PMS Key Inclusion Criteria FIM (N=18) REDUCE-HTN FIM + PMS (N=146) PMS (N=128) • Office-based blood pressure (average of 3 readings): • First in Man (FIM): SBP ≥ 160 mm. Hg • Post-Market Study (PMS): SBP/DBP ≥ 160/90 mm. Hg • ≥ 3 antihypertensive drugs at maximally tolerated doses with stable regimen for at least 2 weeks prior to enrollment • Renal artery length: • FIM: ≥ 20 mm • PMS: ≥ 15 mm • Renal artery without significant stenosis (i. e. , baseline diameter stenosis <30%) • Main renal artery diameter of ≥ 3. 5 mm and ≤ 7. 0 mm for each kidney: • FIM: Single renal artery • PMS: Subjects with accessory renal arteries were enrolled

REDUCE-HTN FIM + PMS Study Endpoints • Efficacy • Reduction of office-based blood pressures at 6 months • Reduction of 24 -hour ambulatory blood pressure at 6 months • Safety Endpoints • Acute Safety • Renal artery dissection or perforation during the procedure that requires stenting or surgery • Renal artery infarction or embolus • Periprocedural cerebrovascular accident • Periprocedural myocardial infarction • Periprocedural sudden cardiac death • Long-term Safety • Renal stenosis requiring an intervention, documented by angiography • Duplex ultrasound required at 6 months to assess absence of flow limiting stenosis in the renal artery (core lab analysis) • Renal function (e. GFR) • Primary endpoint analysis includes 142 patients who have completed 6 months of follow-up (97% of enrolled patients)
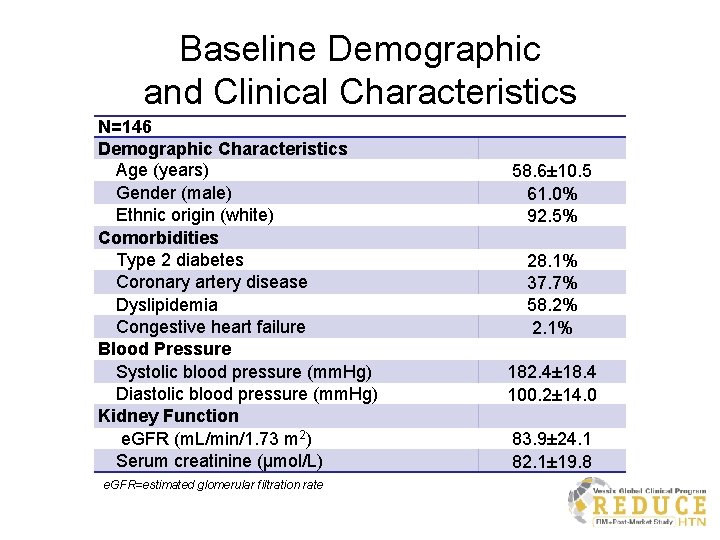
Baseline Demographic and Clinical Characteristics N=146 Demographic Characteristics Age (years) Gender (male) Ethnic origin (white) Comorbidities Type 2 diabetes Coronary artery disease Dyslipidemia Congestive heart failure Blood Pressure Systolic blood pressure (mm. Hg) Diastolic blood pressure (mm. Hg) Kidney Function e. GFR (m. L/min/1. 73 m 2) Serum creatinine (μmol/L) e. GFR=estimated glomerular filtration rate 58. 6± 10. 5 61. 0% 92. 5% 28. 1% 37. 7% 58. 2% 2. 1% 182. 4± 18. 4 100. 2± 14. 0 83. 9± 24. 1 82. 1± 19. 8

Baseline Antihypertensive Medications N=146 Number of antihypertensive medications per patient ≥ 5 Medications 4 Medications 3 Medications Percentage of Patients Receiving: ACE Inhibitors or ARBs β Blockers Diuretics Calcium Channel Blockers Centrally Acting Sympatholytic α-1 Blockers Aldosterone Antagonist Nitrate and Other Vasodilators Direct Renin Inhibitors Other Antihypertensive ACE=angiotensin-converting enzyme; ARB=angiotensin-receptor blocker 5. 3± 1. 9 61. 6% 21. 9% 13. 0% 93. 2% 78. 1% 76. 7% 75. 3% 39. 7% 34. 2% 29. 5% 15. 1% 11. 0% 2. 7%
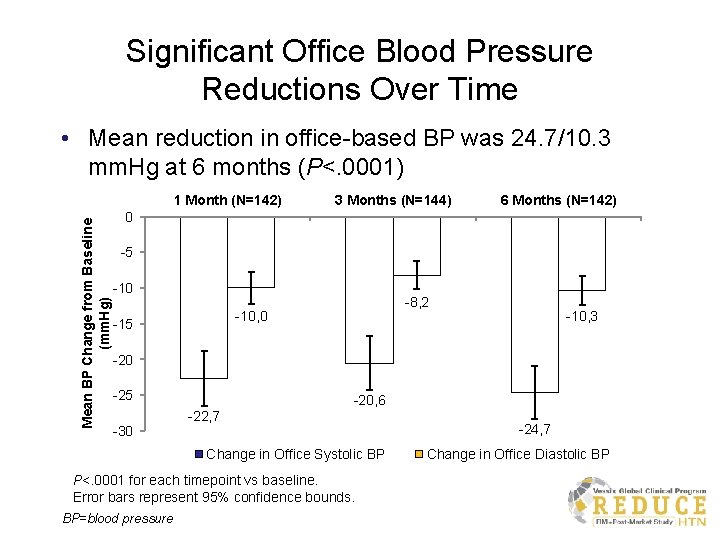
Significant Office Blood Pressure Reductions Over Time • Mean reduction in office-based BP was 24. 7/10. 3 mm. Hg at 6 months (P<. 0001) Mean BP Change from Baseline (mm. Hg) 1 Month (N=142) 3 Months (N=144) 6 Months (N=142) 0 -5 -10 -8, 2 -10, 0 -15 -10, 3 -20 -25 -30 -20, 6 -22, 7 Change in Office Systolic BP P<. 0001 for each timepoint vs baseline. Error bars represent 95% confidence bounds. BP=blood pressure -24, 7 Change in Office Diastolic BP
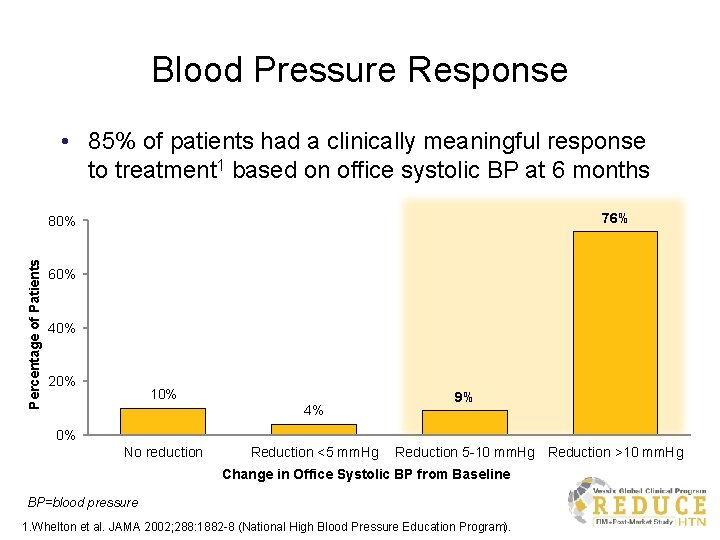
Blood Pressure Response • 85% of patients had a clinically meaningful response to treatment 1 based on office systolic BP at 6 months 76% Percentage of Patients 80% 60% 40% 20% 10% 4% 9% 0% No reduction Reduction <5 mm. Hg Reduction 5 -10 mm. Hg Reduction >10 mm. Hg Change in Office Systolic BP from Baseline BP=blood pressure 1. Whelton et al. JAMA 2002; 288: 1882 -8 (National High Blood Pressure Education Program).
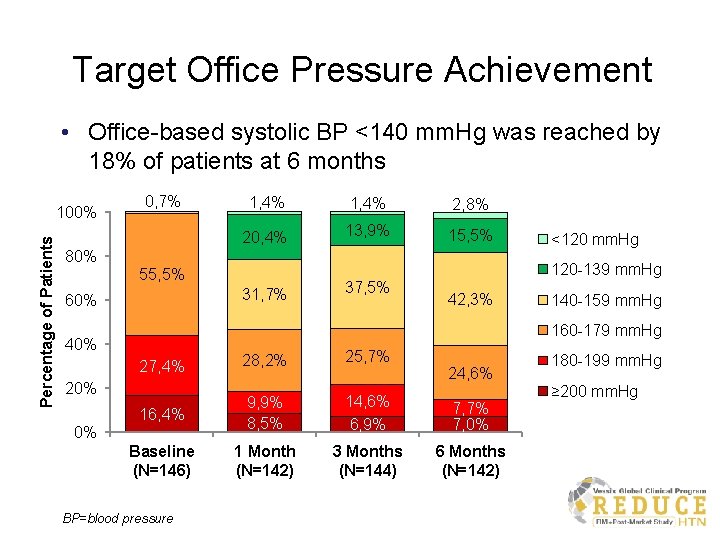
Target Office Pressure Achievement • Office-based systolic BP <140 mm. Hg was reached by 18% of patients at 6 months Percentage of Patients 100% 0, 7% 1, 4% 2, 8% 20, 4% 13, 9% 15, 5% 80% 55, 5% 31, 7% 60% 37, 5% <120 mm. Hg 120 -139 mm. Hg 42, 3% 140 -159 mm. Hg 160 -179 mm. Hg 40% 27, 4% 28, 2% 25, 7% 16, 4% 9, 9% 8, 5% 14, 6% 6, 9% 7, 7% 7, 0% Baseline (N=146) 1 Month (N=142) 3 Months (N=144) 6 Months (N=142) 20% 0% BP=blood pressure 24, 6% 180 -199 mm. Hg ≥ 200 mm. Hg
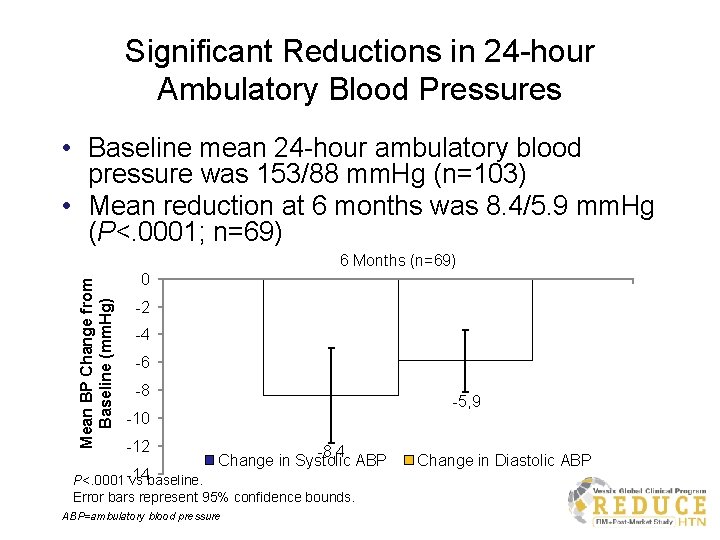
Significant Reductions in 24 -hour Ambulatory Blood Pressures • Baseline mean 24 -hour ambulatory blood pressure was 153/88 mm. Hg (n=103) • Mean reduction at 6 months was 8. 4/5. 9 mm. Hg (P<. 0001; n=69) Mean BP Change from Baseline (mm. Hg) 6 Months (n=69) 0 -2 -4 -6 -8 -5, 9 -10 -12 P<. 0001 -14 vs baseline. -8, 4 Change in Systolic ABP Error bars represent 95% confidence bounds. ABP=ambulatory blood pressure Change in Diastolic ABP
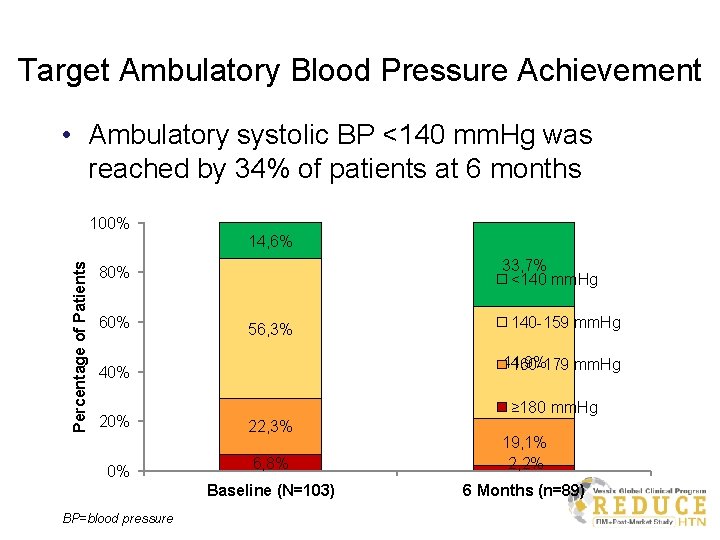
Target Ambulatory Blood Pressure Achievement • Ambulatory systolic BP <140 mm. Hg was reached by 34% of patients at 6 months 100% Percentage of Patients 14, 6% 33, 7% <140 mm. Hg 80% 60% 56, 3% 44, 9% 160 -179 mm. Hg 40% 20% 0% BP=blood pressure 140 -159 mm. Hg ≥ 180 mm. Hg 22, 3% 6, 8% 19, 1% 2, 2% Baseline (N=103) 6 Months (n=89)
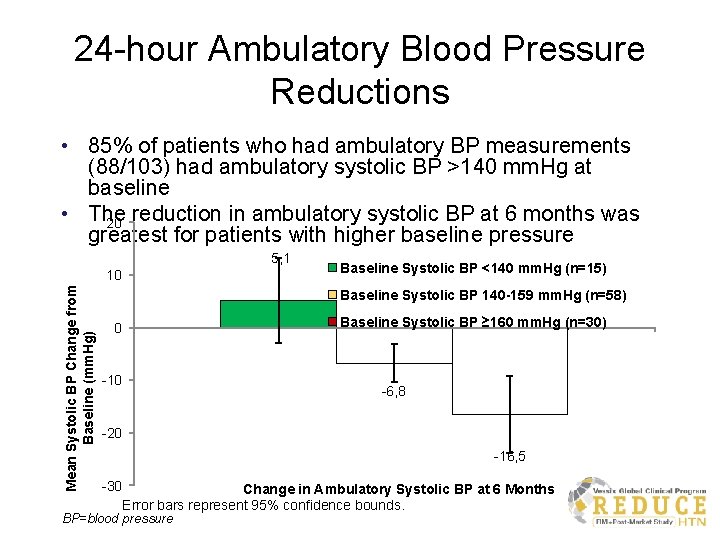
24 -hour Ambulatory Blood Pressure Reductions • 85% of patients who had ambulatory BP measurements (88/103) had ambulatory systolic BP >140 mm. Hg at baseline • The 20 reduction in ambulatory systolic BP at 6 months was greatest for patients with higher baseline pressure 5, 1 Mean Systolic BP Change from Baseline (mm. Hg) 10 Baseline Systolic BP <140 mm. Hg (n=15) Baseline Systolic BP 140 -159 mm. Hg (n=58) Baseline Systolic BP ≥ 160 mm. Hg (n=30) 0 -10 -6, 8 -20 -16, 5 -30 Change in Ambulatory Systolic BP at 6 Months Error bars represent 95% confidence bounds. BP=blood pressure
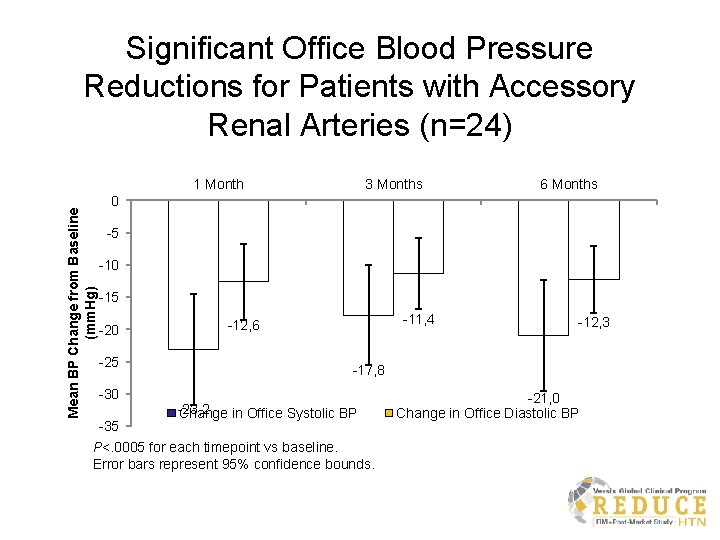
Significant Office Blood Pressure Reductions for Patients with Accessory Renal Arteries (n=24) Mean BP Change from Baseline (mm. Hg) 1 Month 3 Months 6 Months 0 -5 -10 -15 -20 -25 -30 -35 -11, 4 -12, 6 -17, 8 -23, 2 Change in Office Systolic BP P<. 0005 for each timepoint vs baseline. Error bars represent 95% confidence bounds. BP=blood pressure -12, 3 -21, 0 Change in Office Diastolic BP
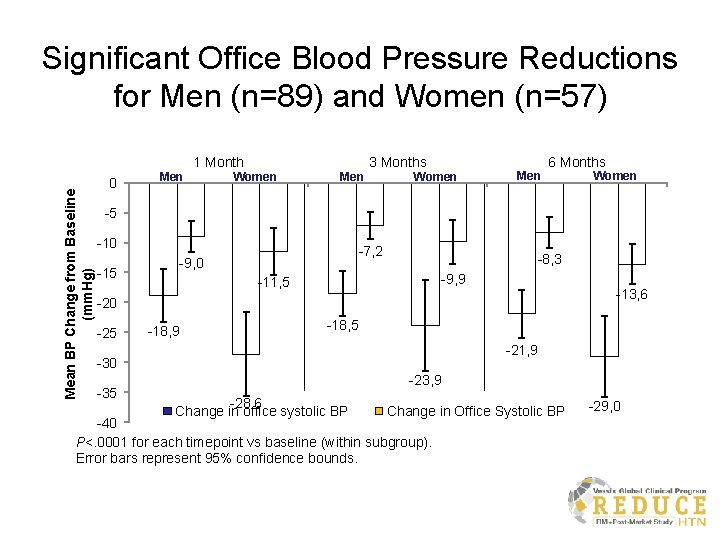
Significant Office Blood Pressure Reductions for Men (n=89) and Women (n=57) Mean BP Change from Baseline (mm. Hg) 1 Month 0 Men 3 Months Women Men 6 Months Women -5 -10 -15 -7, 2 -9, 0 -8, 3 -9, 9 -11, 5 -13, 6 -20 -25 -18, 9 -18, 5 -21, 9 -30 -35 -23, 9 -28, 6 Change in office systolic BP Change in Office Systolic BP -40 P<. 0001 for each timepoint vs baseline (within subgroup). Error bars represent 95% confidence bounds. -29, 0
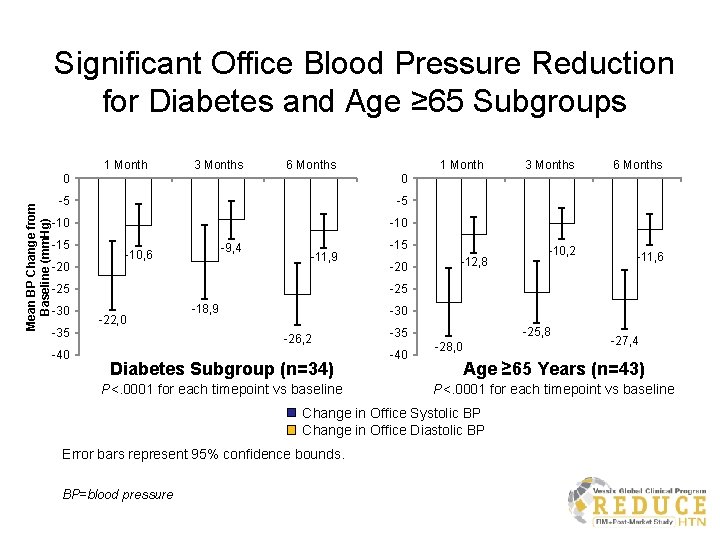
Significant Office Blood Pressure Reduction for Diabetes and Age ≥ 65 Subgroups Mean BP Change from Baseline (mm. Hg) 1 Month 3 Months 6 Months 1 Month 0 0 -5 -5 -10 -15 -20 -9, 4 -10, 6 -11, 9 -25 -30 -20 -12, 8 -10, 2 6 Months -11, 6 -25 -22, 0 -35 -40 -15 3 Months -18, 9 -30 -26, 2 Diabetes Subgroup (n=34) P<. 0001 for each timepoint vs baseline -25, 8 -35 -40 -28, 0 Age ≥ 65 Years (n=43) P<. 0001 for each timepoint vs baseline Change in Office Systolic BP Change in Office Diastolic BP Error bars represent 95% confidence bounds. BP=blood pressure -27, 4

Renal Function • Mean e. GFR changed -0. 9± 16. 4 m. L/min/1. 73 m 2 at 6 months • At 6 months, 10. 9% (15/138) of patients had a reduction in e. GFR >25% from baseline • Only 2 of these patients had kidney-related adverse events, and neither of them were determined to be related to the procedure by the Data Safety Monitoring Board: - Renal insufficiency at 3 months - Acute renal failure at 6 months e. GFR=estimated glomerular filtration rate

No Acute Safety Events and Low Rate of Procedure-Related SAEs • No patient experienced prespecified acute safety events • Low rate of procedure-related serious adverse eventsa (5. 5%) among the 146 patients - Hematoma (1) Bilateral flank pain (1) Vomiting (1) Access site infection (2) Pseudoaneurysm at access site (1) Femoral artery thrombus (1) Renal artery stenosis requiring treatment (1) • Baseline stenosis was 17% based on core lab assessment of angiogram; stenosis of 60% noted by angiography at 6 month follow-up; patient received percutaneous transluminal angioplasty/stent and continues to be monitored. • 6 -month patencyb 99. 2% by DUS (248/250 arteries) a. Serious adverse events (SAEs) defined according to International Organization for Standardization ISO 14155. defined as stenosis <60%. DUS=duplex ultrasound b. Patency

Conclusions • The Vessix System is a safe and effective method for the treatment of resistant hypertension • Significant office blood pressure reductions through 6 months • Significant reductions for patients with accessory renal arteries • Significant reductions for patients ≥ 65 years of age, those with diabetes, and both sexes • 85% of patients had an office systolic BP reduction of at least 5 mm. Hg at 6 months • Ambulatory systolic BP was below 140 mm. Hg at 6 months in 34% of the patients

Thank you!
- Slides: 24