CRT 2013 Washinton DC February 28 2013 Are

CRT 2013 Washinton DC, February 28, 2013 Are we ready to perform TAVI in Intermediate Risk Patients? Eberhard Grube MD FACC, FSCAI Heart Center University Bonn , Bonn, Germany Stanford University, School of Medicine, Stanford, California

Eberhard Grube, MD Consulting: Medtronic Core. Valve, Boston Scientific Corporation, Cordis Corporation, Johnson and Abbott Vascular Honoraria: Boston Scientific Corporation and Biosensors International Stocks, Stock Options, other ownership interest: Medtronic Core. Valve and Biosensors International Off-Label: Off-label use of stents and valve prosthesis

Better TAVR Outcomes in Lower Risk Patients N=420 patients (105 per quartile) Quartile 1 Quartile 4 81. 1 years 78. 9 years Logistic Euroscore, % 25. 4% 17. 8% STS-PROM, % 7. 13% 4. 8% Crude 30 day Mortality, % 11. 4% 3. 8% Age, years Lange JACC 2012; 59: 280– 7)

“Risk Creep” Favors TAVR Preferentially
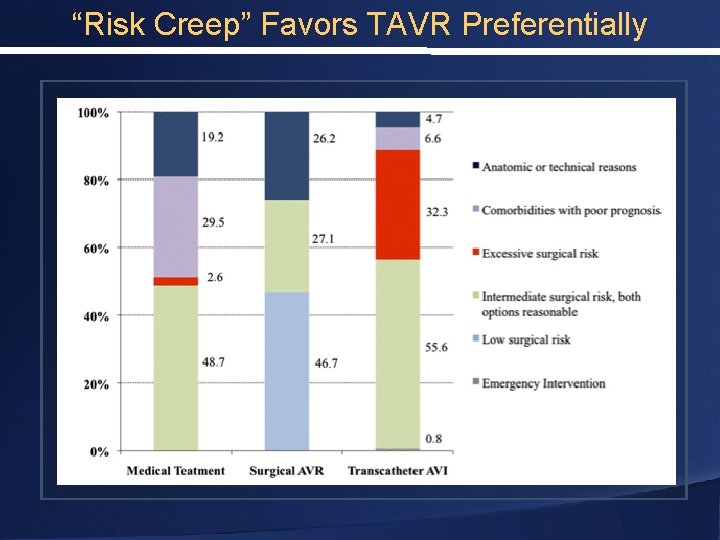
“Risk Creep” Favors TAVR Preferentially
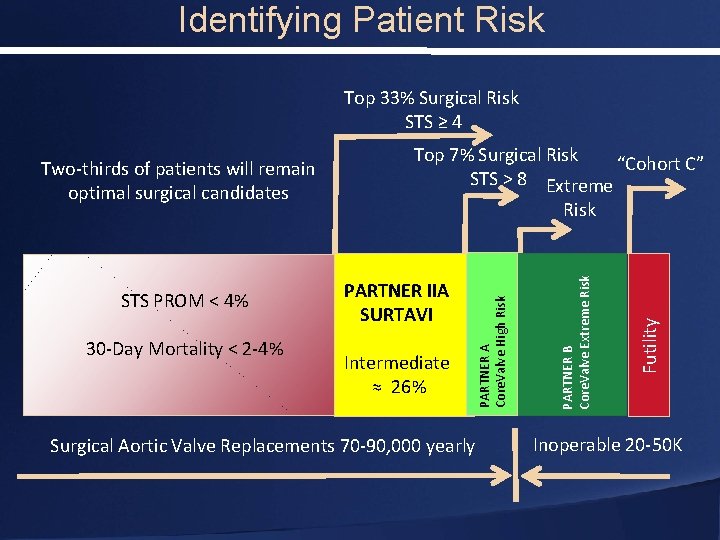
Identifying Patient Risk Top 33% Surgical Risk STS ≥ 4 PARTNER IIA SURTAVI Intermediate ≈ 26% Surgical Aortic Valve Replacements 70 -90, 000 yearly Futility 30 -Day Mortality < 2 -4% Risk PARTNER B Core. Valve Extreme Risk STS PROM < 4% Top 7% Surgical Risk “Cohort C” STS > 8 Extreme PARTNER A Core. Valve High Risk Two-thirds of patients will remain optimal surgical candidates Inoperable 20 -50 K
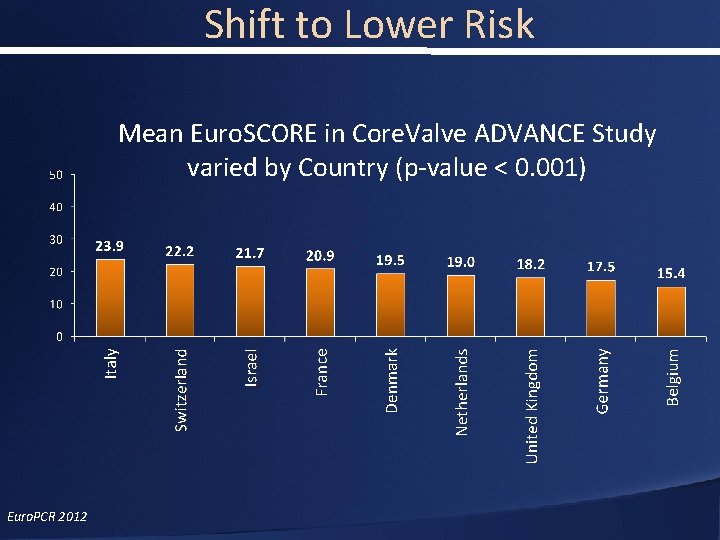
Shift to Lower Risk Mean Euro. SCORE in Core. Valve ADVANCE Study varied by Country (p-value < 0. 001) Euro. PCR 2012

TAVI for Intermediate Patients • Munich and BERMUDA Triangle Studies • Beyond Risk Models • Intermediate Risk Trials
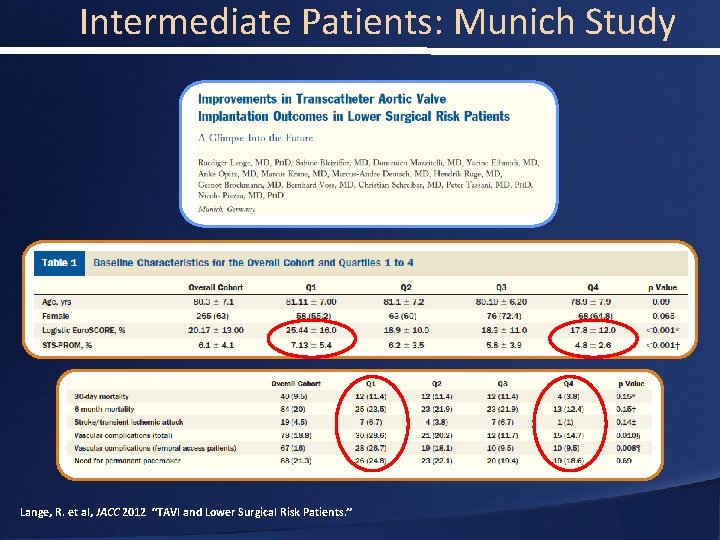
Intermediate Patients: Munich Study Lange, R. et al, JACC 2012 “TAVI and Lower Surgical Risk Patients. ”

BERMUDA Triangle Study BERn – MUnich - rotter. DAm TVT 2011

BERMUDA Triangle Study 3666 patients enrolled TAVI – 782 (21. 3%) SAVR – 2884 (87. 7%) 2882 patients excluded based on propensity score TAVI – 390 (49. 9%) SAVR – 2492 (86. 4%) Propensity score matched patients n = 784 TAVI - 392 Lost to follow-up: 4 (1. 0%) 392 analysed SAVR- 392 Lost to follow-up: 15 (3. 8%) 392 analysed
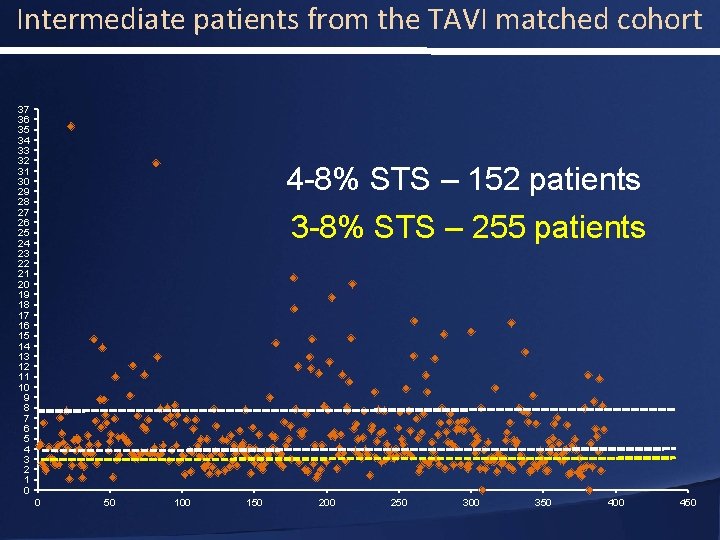
Intermediate patients from the TAVI matched cohort 37 36 35 34 33 32 31 30 29 28 27 26 25 24 23 22 21 20 19 18 17 16 15 14 13 12 11 10 9 8 7 6 5 4 3 2 1 0 4 -8% STS – 152 patients 3 -8% STS – 255 patients 0 50 100 150 200 250 300 350 400 450
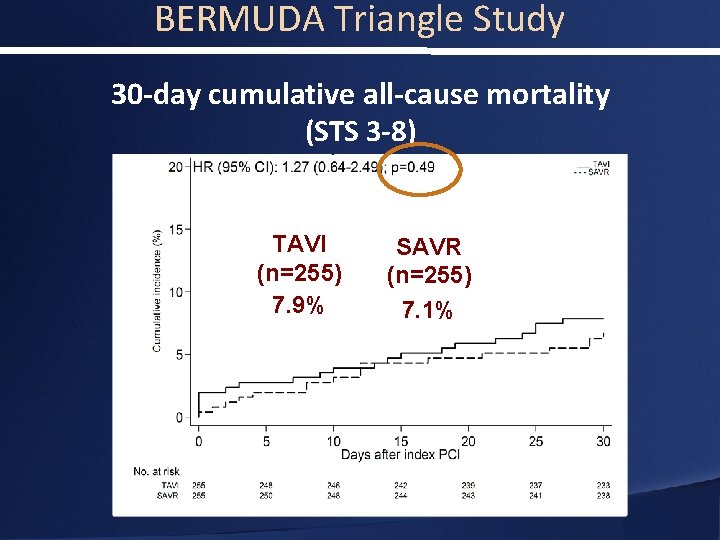
BERMUDA Triangle Study 30 -day cumulative all-cause mortality (STS 3 -8) TAVI (n=255) 7. 9% SAVR (n=255) 7. 1%
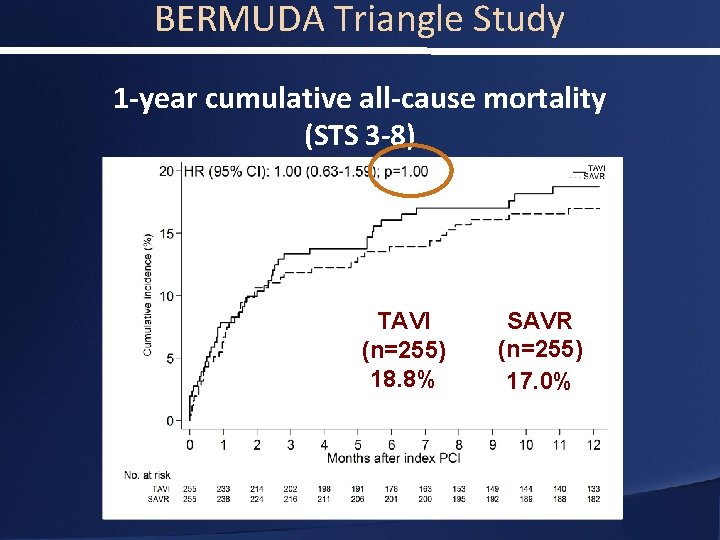
BERMUDA Triangle Study 1 -year cumulative all-cause mortality (STS 3 -8) TAVI (n=255) 18. 8% SAVR (n=255) 17. 0%

BERMUDA Triangle Study Conclusions • Patients with severe aortic stenosis and an intermediate surgical risk have a similar overall mortality at 30 days and 1 years irrespective of the treatment (TAVI or SAVR) • It appears that there is a difference in clinical outcomes between men and women • The efficacy of TAVI in intermediate risk populations and the differences in outcomes between men and women warrants further evaluation in the context of randomized control trials

TAVI for Intermediate Patients • BERMUDA Triangle Study • Beyond Risk Models • Intermediate Risk Trials
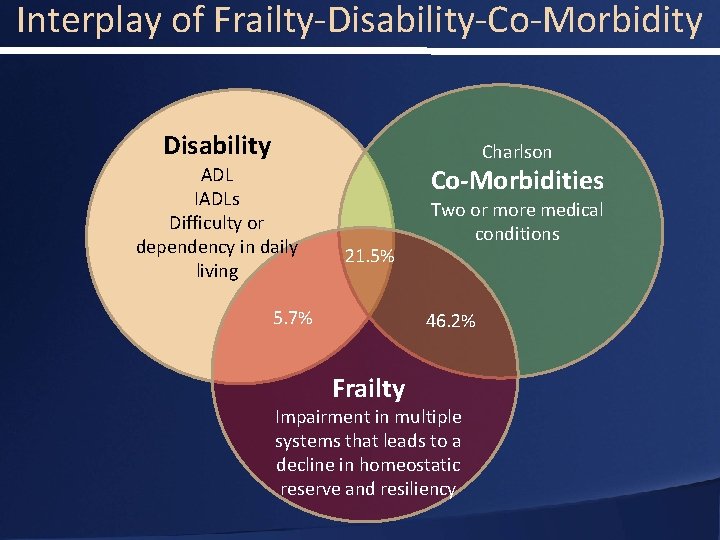
Interplay of Frailty-Disability-Co-Morbidity Disability ADL IADLs Difficulty or dependency in daily living Charlson Co-Morbidities 21. 5% 5. 7% Two or more medical conditions 46. 2% Frailty Impairment in multiple systems that leads to a decline in homeostatic reserve and resiliency

Variables Included in Various Risk Models • Frailty is not represented in any of the commonly used risk scores • STS has begun collecting 5 -meter gait speed as a measure of frailty • Euro. SCORE II has added “Poor mobility” defined as severe impairment of mobility secondary to musculoskeletal or neurological dysfunction Dewey J Thor CV Surgery 2008; 135: 180

Interplay of Frailty-Disability-Co-Morbidity • Improved Frailty Assessments are Needed for: Utility v. Futility • Current risk stratification methods are not sufficient to stratify frailty • Complete assessment includes Frailty Disability Co-Morbidity • Important interactions with pulmonary disease and cognitive function • We still have work to do to to find the risk index

TAVI for Intermediate Patients • BERMUDA Triangle Study • Beyond Risk Models • Intermediate Risk Trials

Patient Selection Presented By Prof. Patrick Serruys, MD on behalf of
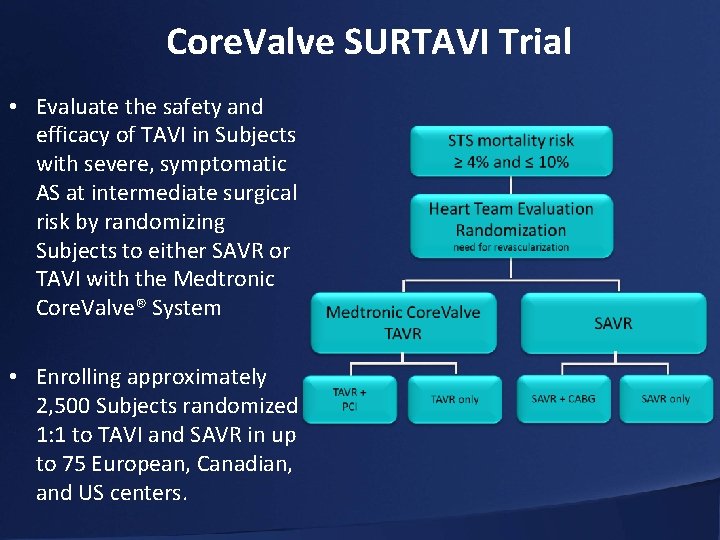
Core. Valve SURTAVI Trial • Evaluate the safety and efficacy of TAVI in Subjects with severe, symptomatic AS at intermediate surgical risk by randomizing Subjects to either SAVR or TAVI with the Medtronic Core. Valve® System • Enrolling approximately 2, 500 Subjects randomized 1: 1 to TAVI and SAVR in up to 75 European, Canadian, and US centers.
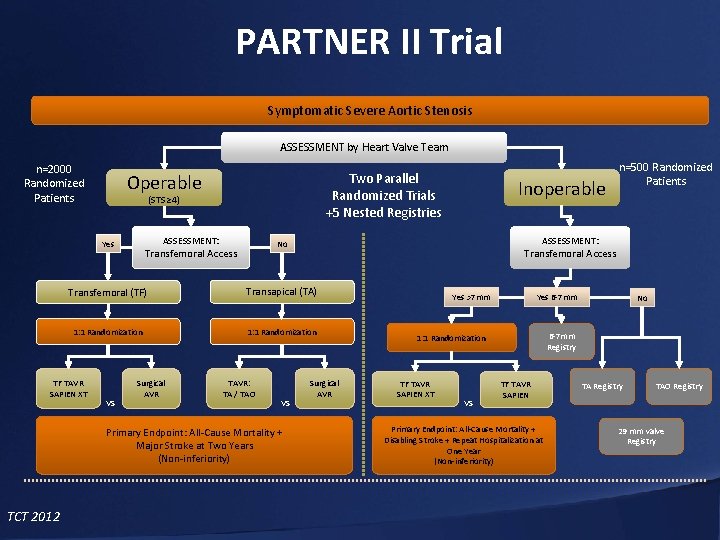
PARTNER II Trial Symptomatic Severe Aortic Stenosis ASSESSMENT by Heart Valve Team n=2000 Randomized Patients Operable Two Parallel Randomized Trials +5 Nested Registries (STS ≥ 4) ASSESSMENT: Yes Transfemoral Access Transapical (TA) 1: 1 Randomization VS Surgical AVR TAVR: TA / TAO VS Primary Endpoint: All-Cause Mortality + Major Stroke at Two Years (Non-inferiority) TCT 2012 ASSESSMENT: No Transfemoral Access Transfemoral (TF) TF TAVR SAPIEN XT Inoperable n=500 Randomized Patients Surgical AVR Yes 6 -7 mm Yes >7 mm 6 -7 mm Registry 1: 1 Randomization TF TAVR SAPIEN XT VS No TF TAVR SAPIEN Primary Endpoint: All-Cause Mortality + Disabling Stroke + Repeat Hospitalization at One Year (Non-inferiority) TA Registry TAO Registry 29 mm valve Registry

Conclusions • Although we have reasons to believe that Intermediate Risk patients will profit from TAVI outcome, we will need randomized data (Partner II, SURTAVI) to definitely extend and recommend TAVI in Intermediate Risk patients • Future risk models/scores will need to contemplate frailty, disability and co-morbidities • Additional “issues” need to be resolved such as procedural complications, durability of valves etc…
- Slides: 24