CRT 2013 Washington DC USA Feb 23 26

CRT 2013 Washington, DC, USA, Feb 23 -26, 2013 Three-Year Results from SYMPLICITY HTN-1 and SYMPLICITY HTN-2: Lessons Learned Horst Sievert, Stefan Bertog, Ilona Hofmann, Laura Vaskelyte, on behalf of the Symplicity HNT-1 and -2 Investigators Cardio. Vascular Center Frankfurt - CVC Frankfurt, Germany

Horst Sievert, MD Consulting: Access. Closure, Inc. , AGA Medical Corporation, Ardian, Inc. , Arstasis, Inc. , Atritech Atrium Medical Corporation, Avinger, Inc. , Bard Peripheral Vascular, Inc. , Boston Scientific Corporation, Bridgepoint, Cardio. Kinetix Inc. , Cardio. MEMS, Inc. , Coherex, Inc. , Contego, CSI, Endo. Cross, Endotex Interventional Systems, Epitek, Evalve, Inc. and ev 3, Inc.

Horst Sievert, MD Consulting: Flow. Cardia, Inc. , Gore, Guidant, Lumen Biomedical, Inc. , HLT, Kensey Nash Corporation, Kyoto Medical, Lifetech, Lutonix, Inc. , Medinol, Medtronic, Inc. , NDCNMT Medical, Inc. , OAS, Occlutech Osprey Medical, Inc. , Ovalis, Inc. , Pathway Medical Technologies, Inc. , Pendra. Care International B. V. , Pfm Medical, Inc. , Rox Medical, Recor, Sadra Medical, Sorin Biomedica Cardio S. R. L. , and Spectranetics Corporation, Trireme, Trivascular and Viacor, Inc.

Horst Sievert, MD Honoraria: Veryan, Ardian, Inc. , Atritech, Atrium Medical Corporation, Boston Scientific Corporation, Cardio. Kinetix Inc. , Cardio. MEMS, Inc. , Coherex, Inc. , Contego, Epitek, Evalve, Inc. , Guidant, Gore, Kyoto Medical, Lutonix, Inc. , Medinol, Medtronic, Inc. , Pfm Medical, Inc. , and Spectranetics Corporation.

Horst Sievert, MD Honoraria: Viacor, Inc. , HLT, Lifetech, Recor, Trivascular, Veryan, CVRx, GDS and In. Seal Medical.

Symplicity HTN-1 Lancet. 2009; 373: 1275 -1281 Initial Cohort – Reported in the Lancet, 2009: -First-in-man, non-randomized -Cohort of 45 patients with resistant HTN (SBP ≥ 160 mm. Hg on ≥ 3 anti-HTN drugs, including a diuretic; e. GFR ≥ 45 m. L/min) - 12 -month data Hypertension. 2011; 57: 911 -917. Expanded Cohort – initially reported in Hypertension, 2011, updated -n=153 -24 and 36 -month follow-up
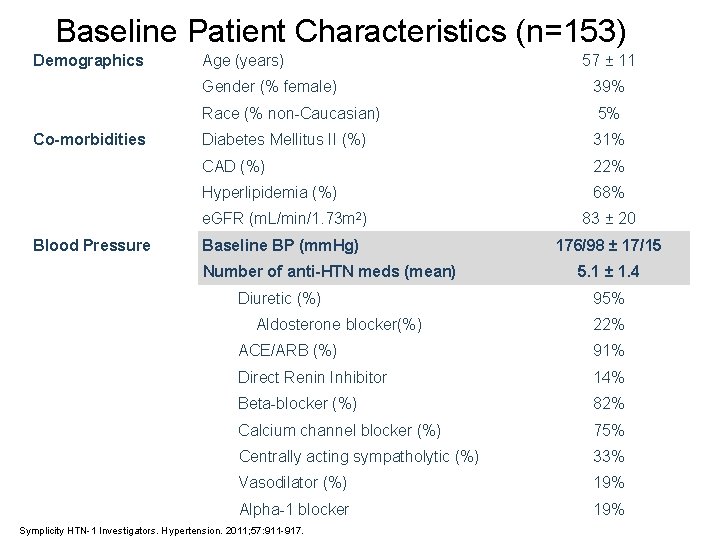
Baseline Patient Characteristics (n=153) Demographics Co-morbidities Blood Pressure Age (years) 57 ± 11 Gender (% female) 39% Race (% non-Caucasian) 5% Diabetes Mellitus II (%) 31% CAD (%) 22% Hyperlipidemia (%) 68% e. GFR (m. L/min/1. 73 m 2) 83 ± 20 Baseline BP (mm. Hg) 176/98 ± 17/15 Number of anti-HTN meds (mean) Diuretic (%) Aldosterone blocker(%) 5. 1 ± 1. 4 95% 22% ACE/ARB (%) 91% Direct Renin Inhibitor 14% Beta-blocker (%) 82% Calcium channel blocker (%) 75% Centrally acting sympatholytic (%) 33% Vasodilator (%) 19% Alpha-1 blocker 19% Symplicity HTN-1 Investigators. Hypertension. 2011; 57: 911 -917.

Procedural Results • • Technical success in all pts Procedure time: 38 min Average of 4 ablations per artery Intravenous narcotics & sedatives used to manage pain during delivery of RF energy 8

Periprocedural Adverse Events • No catheter or generator malfunctions • No major complications • Minor complications 4/153: • 1 renal artery dissection during catheter delivery (prior to RF energy) • no sequelae • 3 access site complications • treated without further sequelae 9

Long-term Adverse Events • Renal CTA, MRA, or Duplex • No vascular abnormalities at any site of RF delivery • One progression of a pre-existing stenosis unrelated to RF treatment (stented without further sequelae) • One new moderate stenosis, no treatment • 3 deaths within the follow-up period; all unrelated to the device or therapy • No orthostatic or electrolyte disturbances • No change in renal function (∆ e. GFR) • 12 Months: -2. 9 m. L/min/1. 73 m 2 (n. s. ) 10
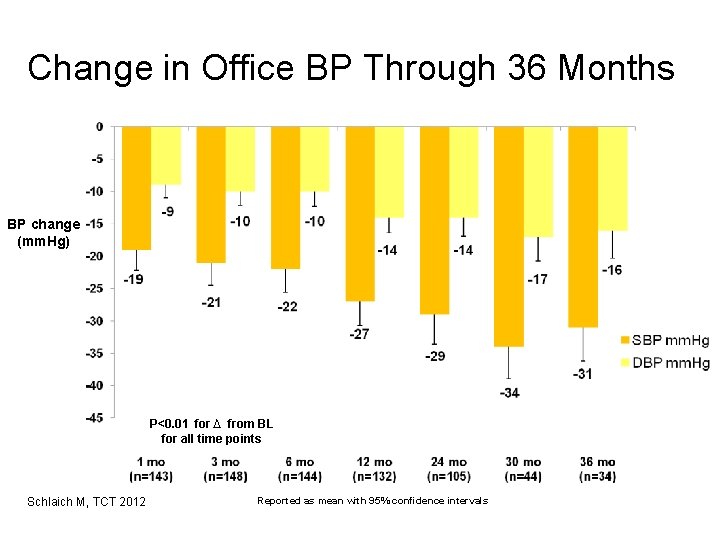
Change in Office BP Through 36 Months BP change (mm. Hg) P<0. 01 for ∆ from BL for all time points Schlaich M, TCT 2012 Reported as mean with 95% confidence intervals
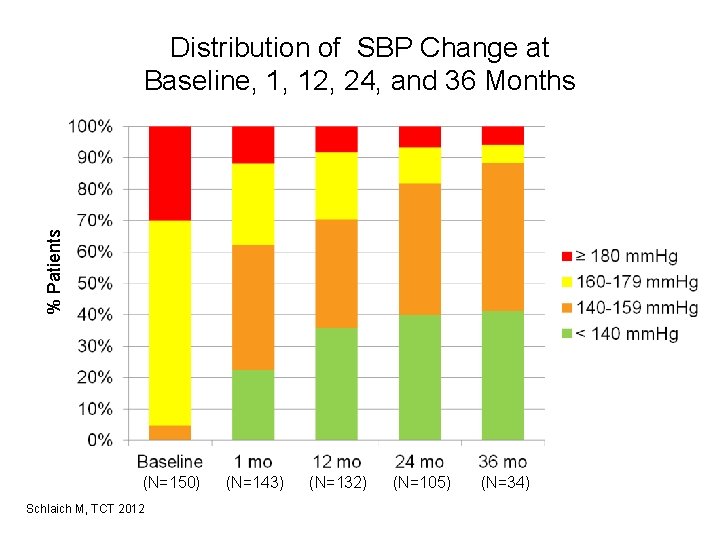
% Patients Distribution of SBP Change at Baseline, 1, 12, 24, and 36 Months (N=150) Schlaich M, TCT 2012 (N=143) (N=132) (N=105) (N=34)
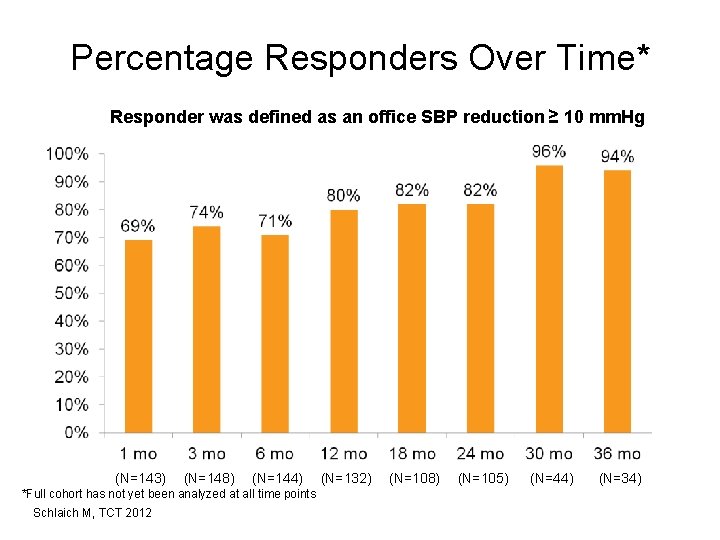
Percentage Responders Over Time* Responder was defined as an office SBP reduction ≥ 10 mm. Hg (N=143) (N=148) (N=144) *Full cohort has not yet been analyzed at all time points Schlaich M, TCT 2012 (N=132) (N=108) (N=105) (N=44) (N=34)
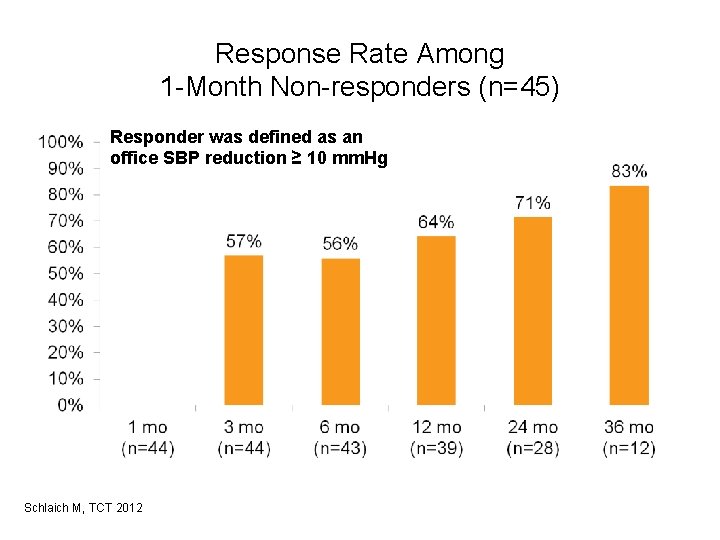
Response Rate Among 1 -Month Non-responders (n=45) Responder was defined as an office SBP reduction ≥ 10 mm. Hg Schlaich M, TCT 2012
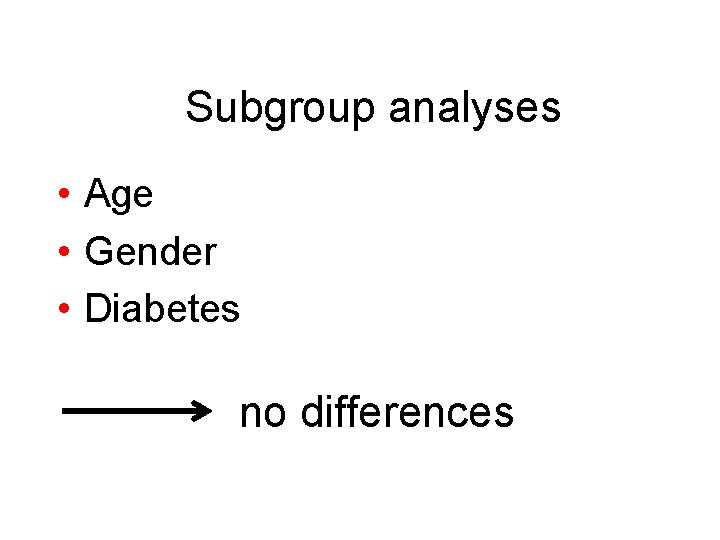
Subgroup analyses • Age • Gender • Diabetes no differences
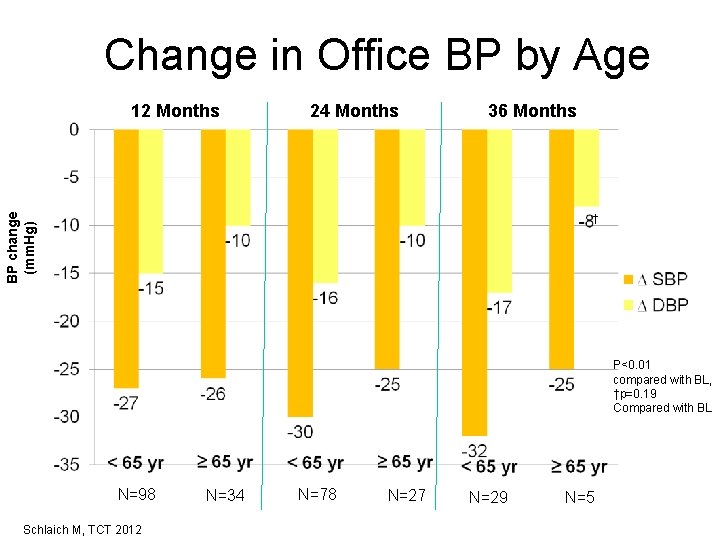
Change in Office BP by Age 24 Months 36 Months BP change (mm. Hg) 12 Months † P<0. 01 compared with BL, †p=0. 19 Compared with BL N=98 Schlaich M, TCT 2012 N=34 N=78 N=27 N=29 N=5
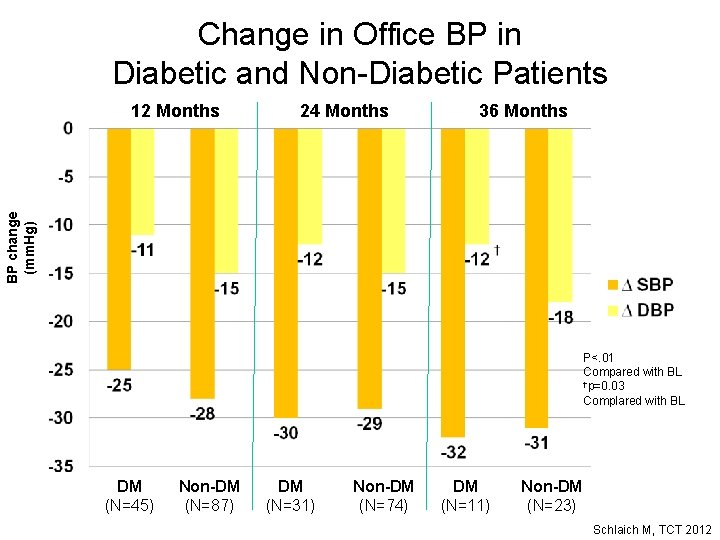
Change in Office BP in Diabetic and Non-Diabetic Patients 24 Months 36 Months BP change (mm. Hg) 12 Months P<. 01 Compared with BL †p=0. 03 Complared with BL DM (N=45) Non-DM (N=87) DM (N=31) Non-DM (N=74) DM (N=11) Non-DM (N=23) Schlaich M, TCT 2012
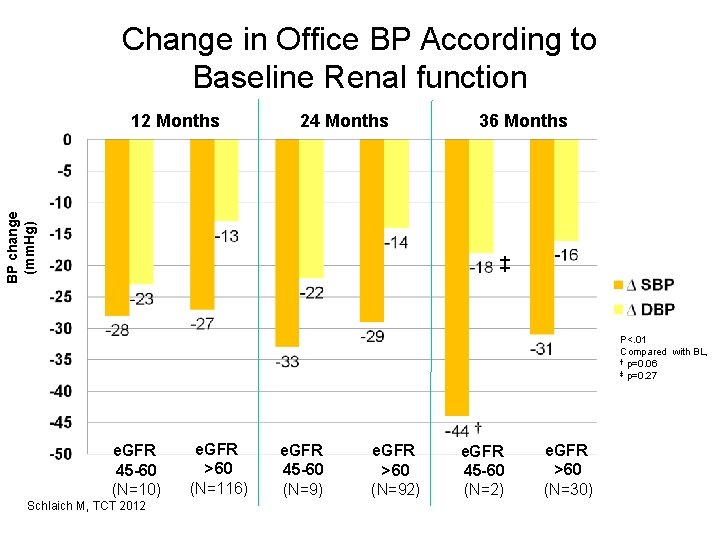
Change in Office BP According to Baseline Renal function 24 Months BP change (mm. Hg) 12 Months 36 Months ‡ P<. 01 Compared with BL, † p=0. 06 ‡ p=0. 27 e. GFR 45 -60 (N=10) Schlaich M, TCT 2012 e. GFR >60 (N=116) e. GFR 45 -60 (N=9) e. GFR >60 (N=92) e. GFR 45 -60 (N=2) e. GFR >60 (N=30)

Vascular Complications Out to 36 months Stenosis (previously reported to 18 months) • One progression of a pre-existing stenosis unrelated to RF treatment (stented without further sequelae) • One new moderate stenosis which was not hemodynamically relevant , requiring no treatment No new reports of vascular complications* *Analysis includes data on all patients available through 36 months Schlaich M, TCT 2012 Symplicity HTN-1 Investigators. Hypertension. 2011; 57: 911 -917.

Other Adverse Events Out to 36 months* • Hypotension and Renal Failure (18 m) n=1 • Due to sepsis, resolved • Hypotension and Renal Failure (24 m) n=1 • Post-operative acute renal failure • resolved • Hypotension Episode (n = 1) • No treatment, resolved • 3 deaths, unrelated to procedure • Myocardial Infarction - After 3 -day visit • Sudden death (cardiac) - After 6 months • Cardio-respiratory arrest - After 18 months *Analysis includes data on all patients available through 36 months Symplicity HTN-1 Investigators. Hypertension. 2011; 57: 911 -917. Schlaich M – TCT 2012

Conclusions • The magnitude of blood pressure reduction is significant and sustained through 36 months • Increasing responder rates indicate: • Some subjects respond late to RDN • No loss of treatment effect out to 36 months • The treatment effect was consistent across subgroups (age, diabetes status, and baseline renal function) • No late adverse events related to RDN through 36 months



Symplicity HTN-2 Lancet. 2010; 376: 1903 -1909 • Study design: randomized, controlled, clinical trial • Patients: 106 patients randomized 1: 1 to treatment with renal denervation vs. control • Clinical Sites: 24 centers in Europe, Australia, & New Zealand Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909

Symplicity HTN-2 Trial • Treatment-resistant HTN population • BL OBP 178/97 mm. Hg • 49 RDN, 51 Control • Age 58 years • BMI 31 kg/m² • 40% with Diabetes • e. GFR 77* • Avg # meds 5. 2 • RDN and Control groups generally well-matched *MDRD, ml/min/1. 73 m 2 Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909. Inclusion Criteria: - Office SBP ≥ 160 mm. Hg (≥ 150 mm. Hg with type II diabetes mellitus) Stable drug regimen of 3+ more anti-HTN medications Age 18 -85 years Exclusion Criteria: - - Hemodynamically or anatomically significant renal artery abnormalities or prior renal artery intervention e. GFR < 45 m. L/min/1. 73 m 2 (MDRD formula) Type 1 diabetes mellitus Contraindication to MRI Stenotic valvular heart disease for which reduction of BP would be hazardous MI, unstable angina, or CVA in the prior 6 months
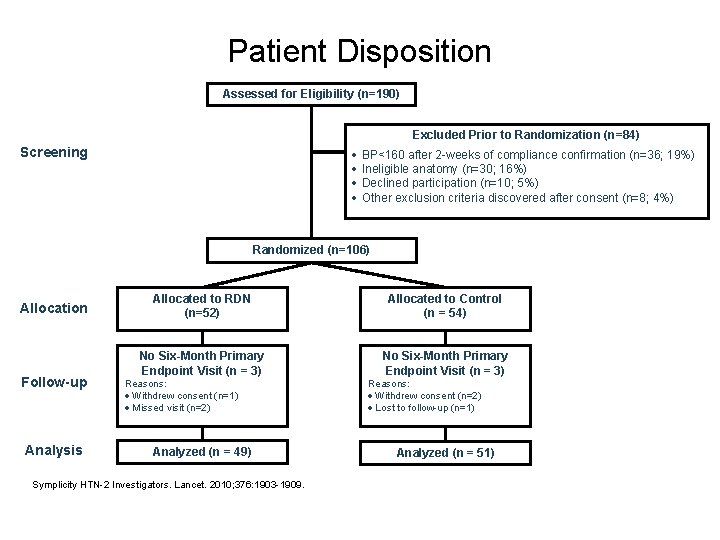
Patient Disposition Assessed for Eligibility (n=190) Excluded Prior to Randomization (n=84) Screening · BP<160 after 2 -weeks of compliance confirmation (n=36; 19%) · Ineligible anatomy (n=30; 16%) · Declined participation (n=10; 5%) · Other exclusion criteria discovered after consent (n=8; 4%) Randomized (n=106) Allocation Follow-up Analysis Allocated to RDN (n=52) Allocated to Control (n = 54) No Six-Month Primary Endpoint Visit (n = 3) Reasons: · Withdrew consent (n=1) · Missed visit (n=2) Analyzed (n = 49) Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909. Reasons: · Withdrew consent (n=2) · Lost to follow-up (n=1) Analyzed (n = 51)

Adverse events Symplicity HTN-2 • Renal artery dissection (n=1) - • One hospitalization prolonged due to hypotension (n=1) - • resolved Minor adverse events (full cohort) - • from injection of contrast into renal artery wall during dye angiography. stented without further consequences 1 femoral artery pseudoaneurysm treated with manual compression 1 postprocedural drop in BP resulting in a reduction in medication 1 urinary tract infection 1 prolonged hospitalization for evaluation of paraesthesias 1 back pain treated with pain medications and resolved after 1 month 6 -month renal imaging (n=43) showed no vascular abnormalities at any RF treatment site • 1 MRA indicates possible progression of a pre-existing stenosis unrelated to RF treatment (no furtherapy warranted) Esler M, ACC 2012 Esler M, ESC 2012
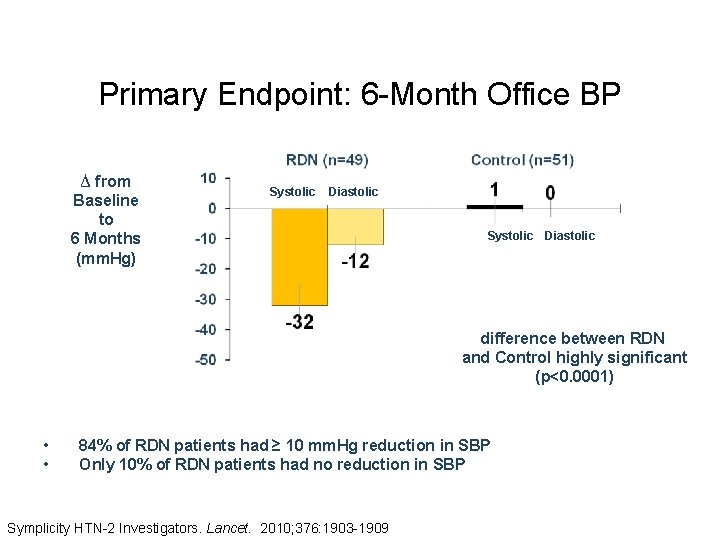
Primary Endpoint: 6 -Month Office BP ∆ from Baseline to 6 Months (mm. Hg) Systolic Diastolic difference between RDN and Control highly significant (p<0. 0001) • • 84% of RDN patients had ≥ 10 mm. Hg reduction in SBP Only 10% of RDN patients had no reduction in SBP Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909
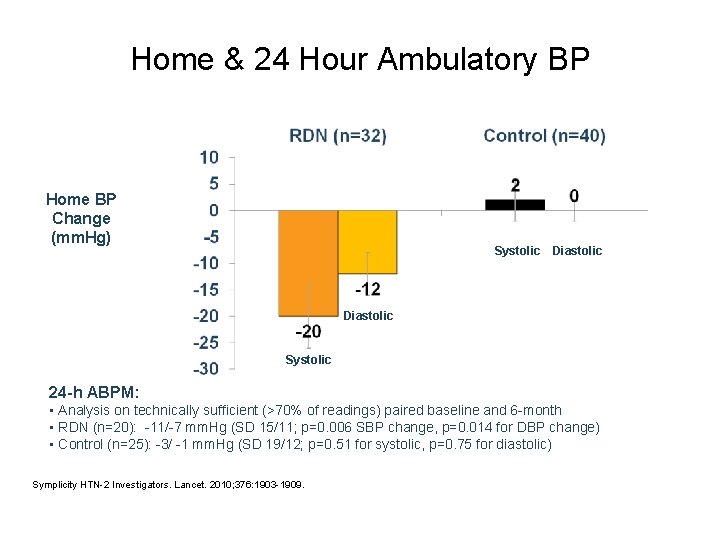
Home & 24 Hour Ambulatory BP Home BP Change BP (mm. Hg) Change Systolic (mm. Hg) Diastolic Systolic 24 -h ABPM: • Analysis on technically sufficient (>70% of readings) paired baseline and 6 -month • RDN (n=20): -11/-7 mm. Hg (SD 15/11; p=0. 006 SBP change, p=0. 014 for DBP change) • Control (n=25): -3/ -1 mm. Hg (SD 19/12; p=0. 51 for systolic, p=0. 75 for diastolic) Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909.
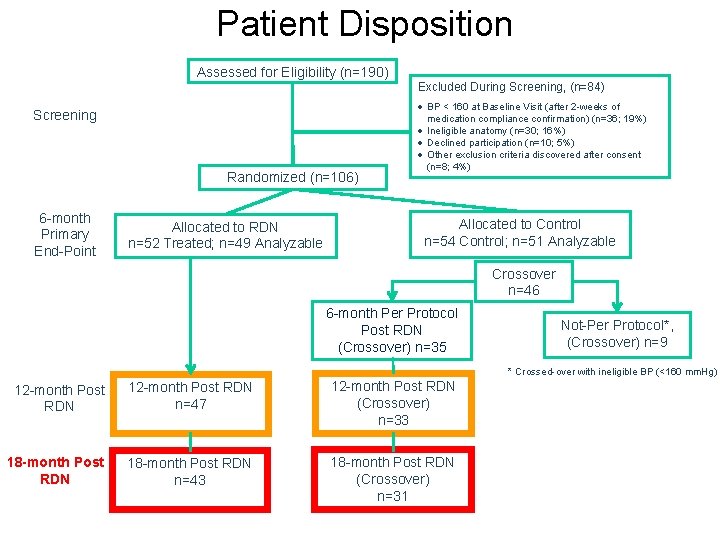
Patient Disposition Assessed for Eligibility (n=190) Screening Randomized (n=106) 6 -month Primary End-Point Allocated to RDN n=52 Treated; n=49 Analyzable Excluded During Screening, (n=84) · BP < 160 at Baseline Visit (after 2 -weeks of medication compliance confirmation) (n=36; 19%) · Ineligible anatomy (n=30; 16%) · Declined participation (n=10; 5%) · Other exclusion criteria discovered after consent (n=8; 4%) Allocated to Control n=54 Control; n=51 Analyzable Crossover n=46 6 -month Per Protocol Post RDN (Crossover) n=35 Not-Per Protocol*, (Crossover) n=9 * Crossed-over with ineligible BP (<160 mm. Hg) 12 -month Post RDN n=47 12 -month Post RDN (Crossover) n=33 18 -month Post RDN n=43 18 -month Post RDN (Crossover) n=31
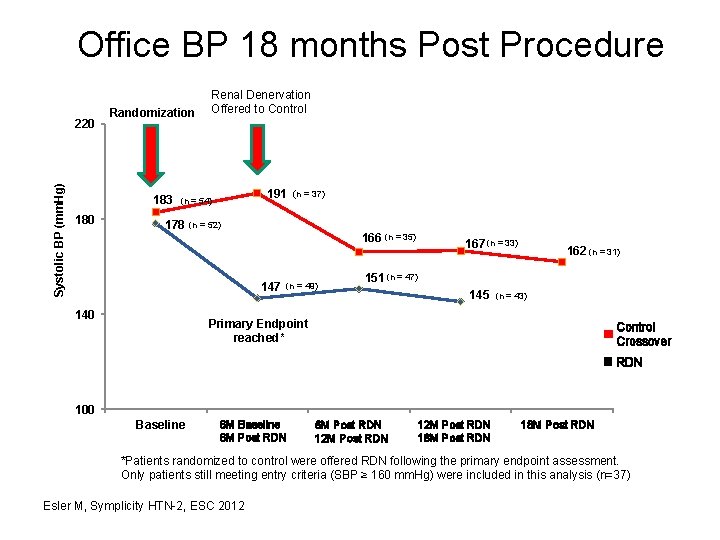
Office BP 18 months Post Procedure Systolic BP (mm. Hg) 220 Randomization 183 180 Renal Denervation Offered to Control 191 (n = 37) (n = 54) 178 (n = 52) 166 147 140 (n = 49) (n = 35) 167 (n = 33) 162 (n = 31) 151 (n = 47) 145 (n = 43) Primary Endpoint reached* Control Crossover RDN 100 Baseline 6 M Post RDN 12 M Post RDN 18 M Post RDN *Patients randomized to control were offered RDN following the primary endpoint assessment. Only patients still meeting entry criteria (SBP ≥ 160 mm. Hg) were included in this analysis (n=37) Esler M, Symplicity HTN-2, ESC 2012
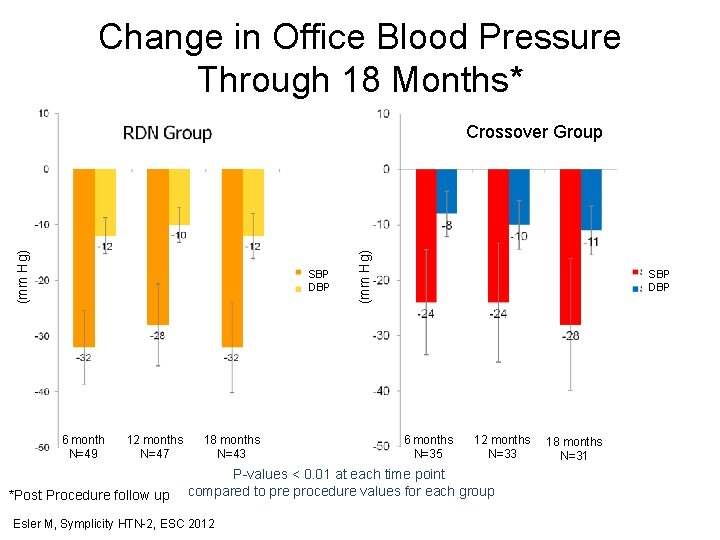
Change in Office Blood Pressure Through 18 Months* SBP DBP 6 month N=49 12 months N=47 *Post Procedure follow up 18 months N=43 (mm Hg) Crossover Group SBP DBP 6 months N=35 12 months N=33 P-values < 0. 01 at each time point compared to pre procedure values for each group Esler M, Symplicity HTN-2, ESC 2012 18 months N=31
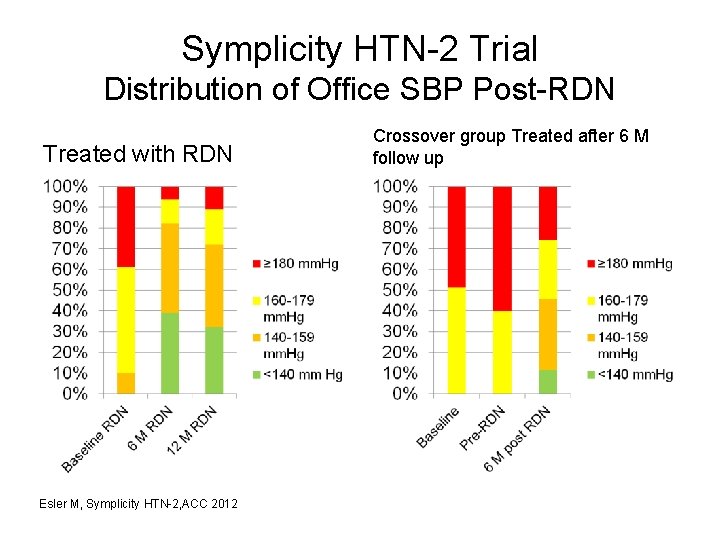
Symplicity HTN-2 Trial Distribution of Office SBP Post-RDN Treated with RDN Esler M, Symplicity HTN-2, ACC 2012 Crossover group Treated after 6 M follow up
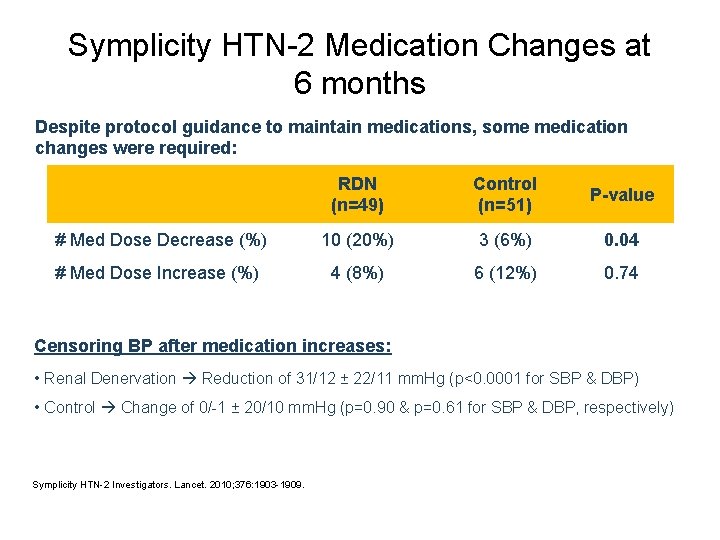
Symplicity HTN-2 Medication Changes at 6 months Despite protocol guidance to maintain medications, some medication changes were required: RDN (n=49) Control (n=51) P-value # Med Dose Decrease (%) 10 (20%) 3 (6%) 0. 04 # Med Dose Increase (%) 4 (8%) 6 (12%) 0. 74 Censoring BP after medication increases: • Renal Denervation Reduction of 31/12 ± 22/11 mm. Hg (p<0. 0001 for SBP & DBP) • Control Change of 0/-1 ± 20/10 mm. Hg (p=0. 90 & p=0. 61 for SBP & DBP, respectively) Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909.
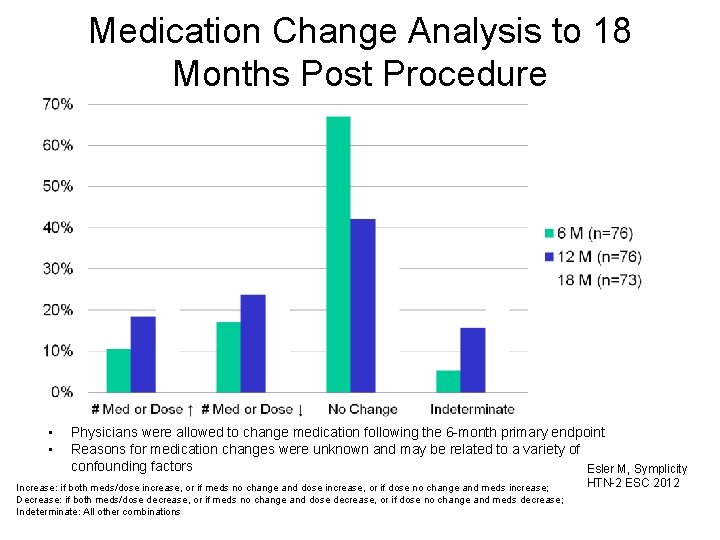
Medication Change Analysis to 18 Months Post Procedure • • Physicians were allowed to change medication following the 6 -month primary endpoint Reasons for medication changes were unknown and may be related to a variety of confounding factors Esler M, Symplicity Increase: if both meds/dose increase, or if meds no change and dose increase, or if dose no change and meds increase; Decrease: if both meds/dose decrease, or if meds no change and dose decrease, or if dose no change and meds decrease; Indeterminate: All other combinations HTN-2 ESC 2012
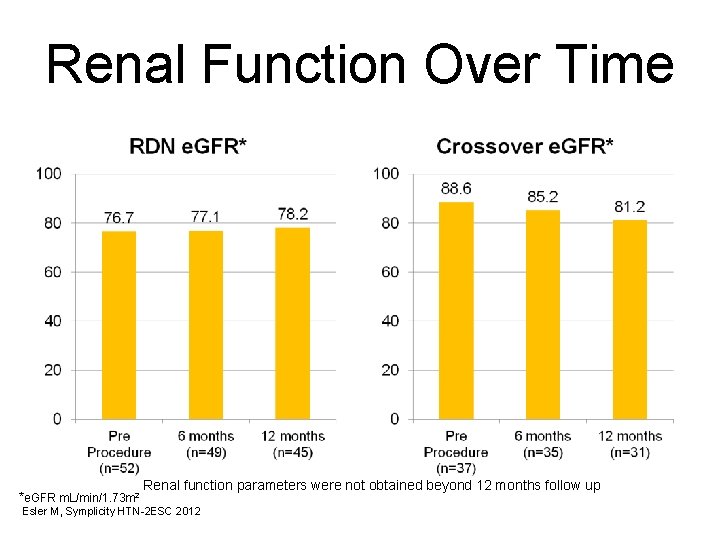
Renal Function Over Time *e. GFR m. L/min/1. 73 m 2 Renal function parameters were not obtained beyond 12 months follow up Esler M, Symplicity HTN-2 ESC 2012
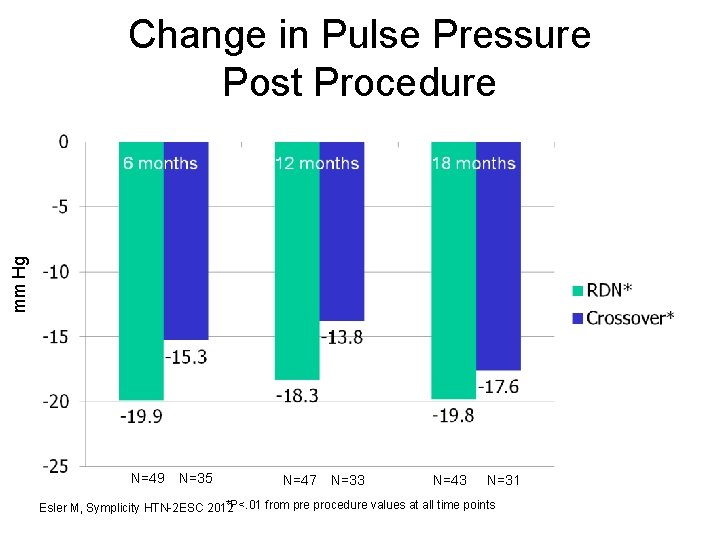
mm Hg Change in Pulse Pressure Post Procedure N=49 N=35 N=47 N=33 N=43 N=31 *P<. 01 from pre procedure values at all time points Esler M, Symplicity HTN-2 ESC 2012
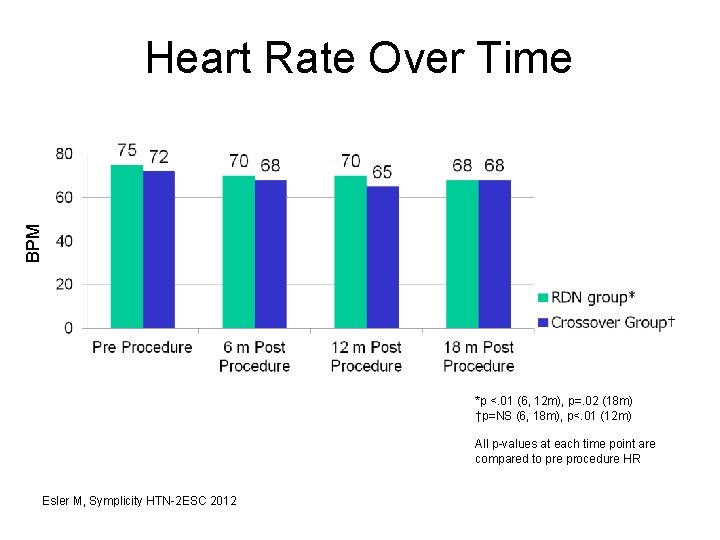
BPM Heart Rate Over Time *p <. 01 (6, 12 m), p=. 02 (18 m) †p=NS (6, 18 m), p<. 01 (12 m) All p-values at each time point are compared to pre procedure HR Esler M, Symplicity HTN-2 ESC 2012

Symplicity HTN-2: Adverse events through 18 mo post procedure • 2 hypotensive events that required hospitalization: 1 in crossover cohort and 1 in RDN cohort • 10 hypertensive events (in 8 patients) requiring hospitalization through 18 months post RDN in combined cohort • 1 mild transient acute renal failure • resolved • 2 unrelated deaths during follow-up *Renal function parameters were not obtained beyond 12 months follow up

Take Home Messages • In patients with resistant hypertension, Trans-catheter Renal Denervation with the Symplicity catheter results in significant reductions in BP • Serious complications are extremely rare • The effect is sustained up to 3 yrs

- Slides: 41