CRT 2012 Washington DC February 6 2012 Embolic

CRT 2012 Washington DC, February 6, 2012 Embolic Protection Devices for Stroke Prevention during TAVI Eberhard Grube MD, FACC, FSCAI Universitatsklinikum Bonn, Germany Hospital Alemão Oswaldo Cruz, São Paulo, Brazil Stanford University, Palo Alto, California, USA

Financial Disclosure Within the past 12 months, the presenter or their spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Physician Name Company/Relationship Eberhard Grube, MD Medtronic, Core. Valve: C, SB, AB, OF Sadra Medical: E, C, SB, AB Direct Flow: C, SB, AB Mitralign: AB, SB, E Boston Scientific: C, SB, AB Biosensors: E, SB, C, AB Cordis: AB Abbott Vascular: AB Capella: SB, C, AB Valtech: E, SB, Claret: SB Key G – Grant and or Research Support E – Equity Interests S – Salary, AB – Advisory Board C – Consulting fees, Honoraria R – Royalty Income I – Intellectual Property Rights SB – Speaker’s Bureau O – Ownership OF – Other Financial Benefits‘

Stroke after TAVI Neurologic events are probably widely undereported in registries. A detailed collaboration with the neurology service is a critical part of program building to standardize the evaluation after an event
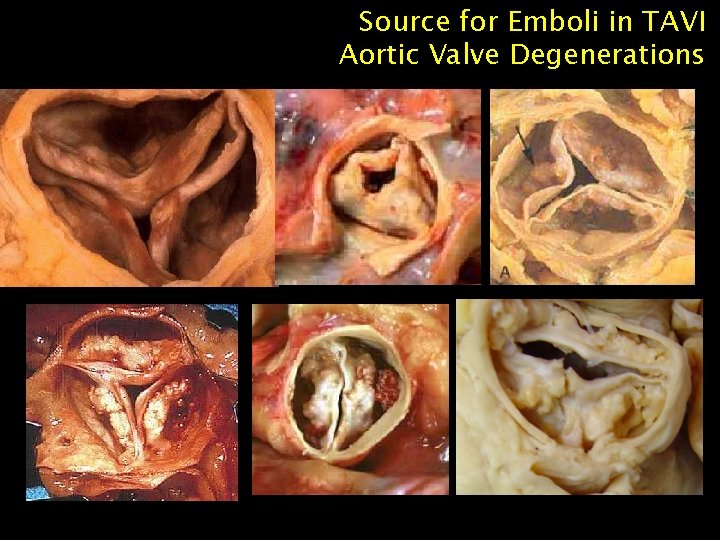
Source for Emboli in TAVI Aortic Valve Degenerations
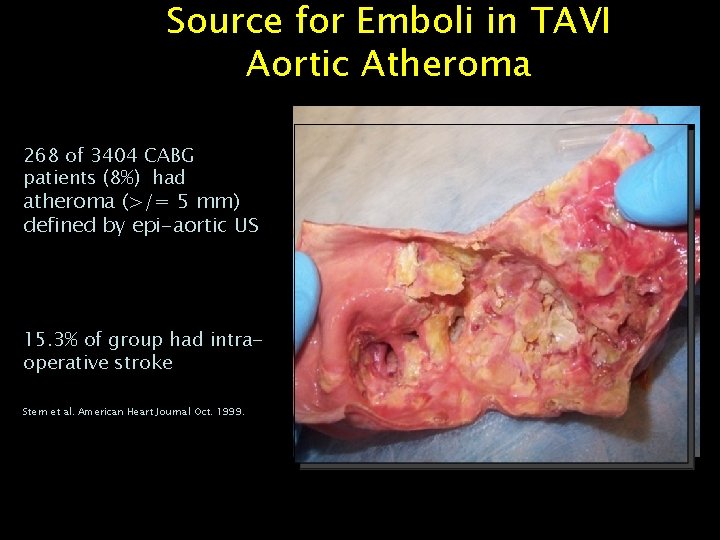
Source for Emboli in TAVI Aortic Atheroma 268 of 3404 CABG patients (8%) had atheroma (>/= 5 mm) defined by epi-aortic US 15. 3% of group had intraoperative stroke Stern et al. American Heart Journal Oct. 1999.
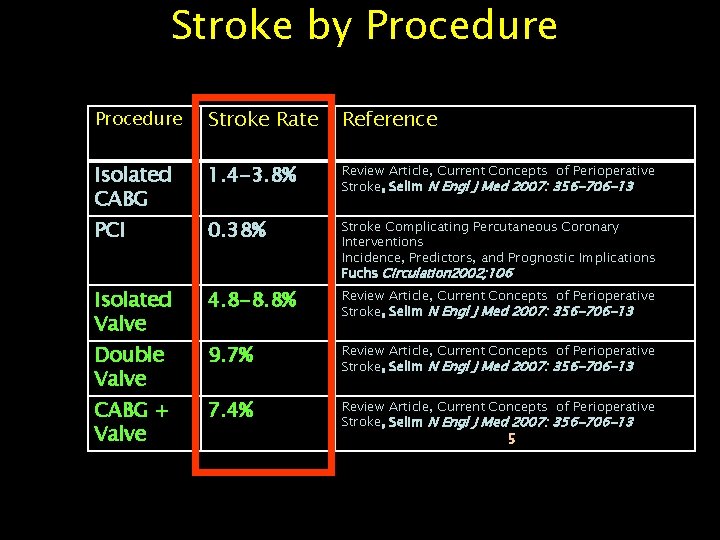
Stroke by Procedure Stroke Rate Reference Isolated CABG 1. 4 -3. 8% Review Article, Current Concepts of Perioperative Stroke, Selim N Engl J Med 2007: 356 -706 -13 PCI 0. 38% Stroke Complicating Percutaneous Coronary Interventions Incidence, Predictors, and Prognostic Implications Fuchs Circulation 2002; 106 Isolated Valve 4. 8 -8. 8% Review Article, Current Concepts of Perioperative Stroke, Selim N Engl J Med 2007: 356 -706 -13 Double Valve 9. 7% Review Article, Current Concepts of Perioperative Stroke, Selim N Engl J Med 2007: 356 -706 -13 CABG + Valve 7. 4% Review Article, Current Concepts of Perioperative Stroke, Selim N Engl J Med 2007: 356 -706 -13 5

84% SILENT STROKE following TAVI J. Am. Coll. Cardiol. 2010; 55; 1427 -1432; originally published online Feb 23, 2010; Rolf Fimmers, Georg Nickenig, and Daniel Thomas Werner, Christoph Hammerstingl, Hans H. Schild, Jörg O. Schwab, Fritz Mellert, Alexander Ghanem, Andreas Müller, Claas P. Nähle, Justine Kocurek, Nikos Resonance Imaging Implantation: A Prospective Pilot Study With Diffusion-Weighted Magnetic Risk and Fate of Cerebral Embolism After Transfemoral Aortic Valve This information is current as of April 6, 2011 http: //content. onlinejacc. org/cgi/content/full/55/14/1427 located on the World Wide Web at: The online version of this article, along with updated information and services, is Downloaded from content. onlinejacc. org by on April 6, 2011 Bonn
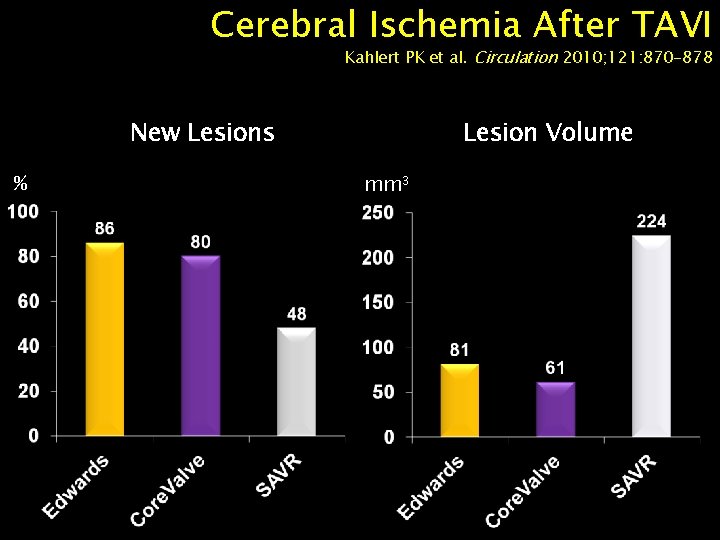
Cerebral Ischemia After TAVI Kahlert PK et al. Circulation 2010; 121: 870 -878 New Lesions % Lesion Volume mm 3
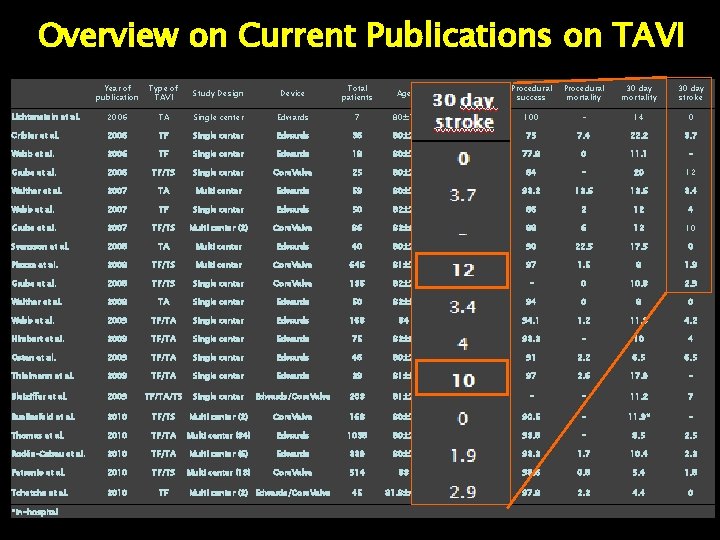
Overview on Current Publications on TAVI Year of publication Type of TAVI Study Design Device Total patients Age Logistic Euro. Score Procedural success Procedural mortality 30 day stroke Lichtenstein et al. 2006 TA Single center Edwards 7 80± 7 35 100 - 14 0 Cribier et al. 2006 TF Single center Edwards 36 80± 7 ~27 75 7. 4 22. 2 3. 7 Webb et al. 2006 TF Single center Edwards 18 80± 7 26. 2± 13. 1 77. 8 0 11. 1 - Grube et al. 2006 TF/TS Single center Core. Valve 25 80± 7 11 84 - 20 12 Walther et al. 2007 TA Multi center Edwards 59 80± 7 27 93. 2 13. 6 3. 4 Webb et al. 2007 TF Single center Edwards 50 82± 7 28 86 2 12 4 Grube et al. 2007 TF/TS Multi center (2) Core. Valve 86 82± 6 21. 7± 12. 6 88 6 12 10 Svensson et al. 2008 TA Multi center Edwards 40 80± 7 35. 5 90 22. 5 17. 5 0 Piazza et al. 2008 TF/TS Multi center Core. Valve 646 81± 7 23. 1± 13. 8 97 1. 5 8 1. 9 Grube et al. 2008 TF/TS Single center Core. Valve 136 82± 7 23. 1± 13. 8 - 0 10. 8 2. 9 Walther et al. 2008 TA Single center Edwards 50 82± 5 27. 6± 12. 2 94 0 8 0 Webb et al. 2009 TF/TA Single center Edwards 168 84 28. 6 94. 1 1. 2 11. 3 4. 2 Himbert et al. 2009 TF/TA Single center Edwards 75 82± 8 26± 13 93. 3 - 10 4 Osten et al. 2009 TF/TA Single center Edwards 46 80± 7 25. 3 91 2. 2 6. 5 Thielmann et al. 2009 TF/TA Single center Edwards 39 81± 5 44. 2± 12. 6 97 2. 6 17. 9 - Bleiziffer et al. 2009 TF/TA/TS Single center Edwards/Core. Valve 203 81± 7 22± 14 - - 11. 2 7 Buellesfeld et al. 2010 TF/TS Multi center (2) Core. Valve 168 80± 7 23. 8± 15. 4 90. 5 - 11. 9* - Thomas et al. 2010 TF/TA Multi center (34) Edwards 1038 80± 7 25. 7 (TF)/29. 1 (TA) 93. 8 - 8. 5 2. 5 Rodés-Cabau et al. 2010 TF/TA Multi center (6) Edwards 339 80± 7 - 93. 3 1. 7 10. 4 2. 3 Petronio et al. 2010 TF/TS Multi center (13) Core. Valve 514 83 20. 1 98. 6 0. 8 5. 4 1. 8 Tchetche et al. 2010 TF 45 81. 8± 4. 2 25. 2± 8. 4 97. 8 2. 2 4. 4 0 *in-hospital Multi center (2) Edwards/Core. Valve
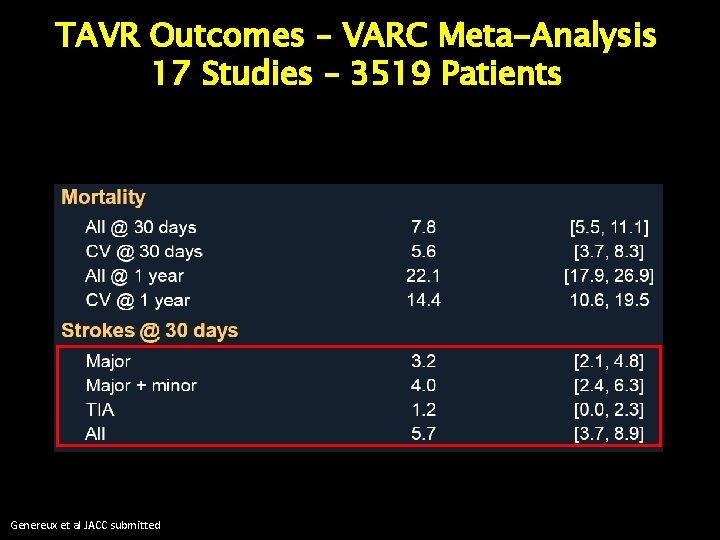
TAVR Outcomes – VARC Meta-Analysis 17 Studies – 3519 Patients Genereux et al JACC submitted
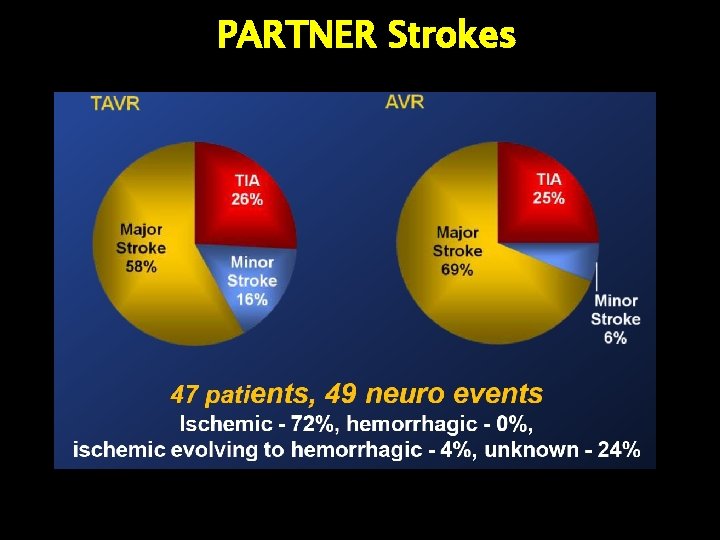
PARTNER Strokes
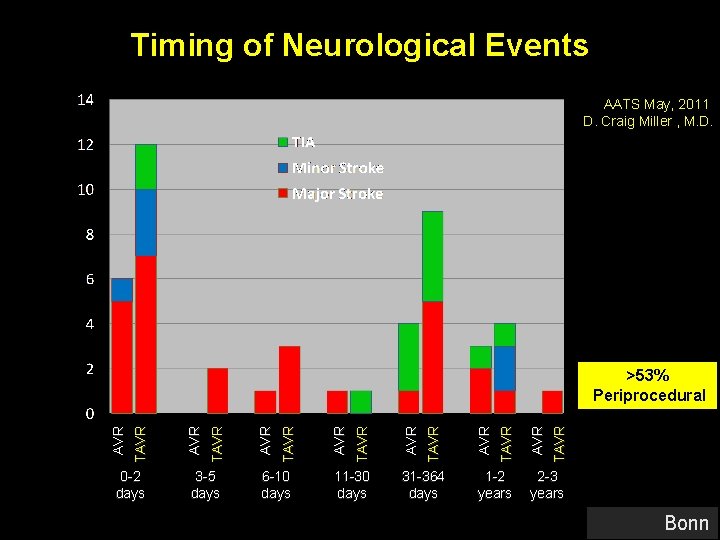
Timing of Neurological Events AATS May, 2011 D. Craig Miller , M. D. 0 -2 days 3 -5 days 6 -10 days 11 -30 days 31 -364 days 1 -2 years TAVR AVR TAVR AVR TAVR >53% Periprocedural 2 -3 years Bonn

What can we do to Reduce Stroke ? • New Embolic Devices • Anti platelet drug regiment, anti coagulant • Less invasive, smaller devices • Less pre-dilatation, careful positioning Bonn
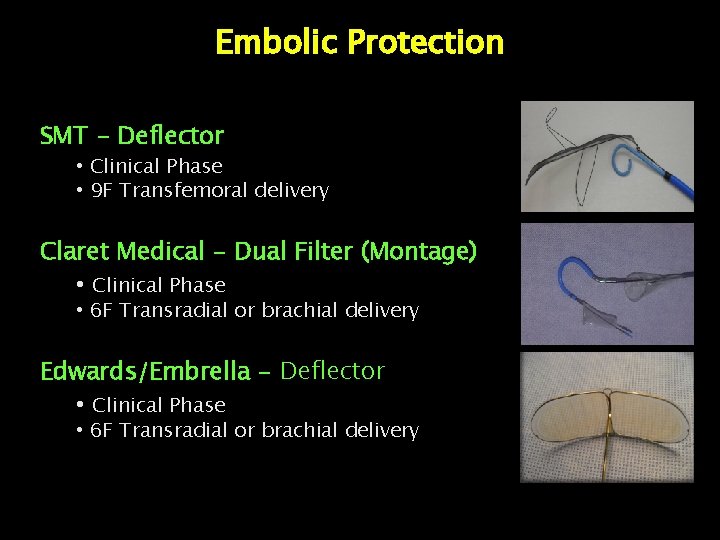
Embolic Protection SMT - Deflector • Clinical Phase • 9 F Transfemoral delivery Claret Medical - Dual Filter (Montage) • Clinical Phase • 6 F Transradial or brachial delivery Edwards/Embrella - Deflector • Clinical Phase • 6 F Transradial or brachial delivery

SMT – SHEF Device
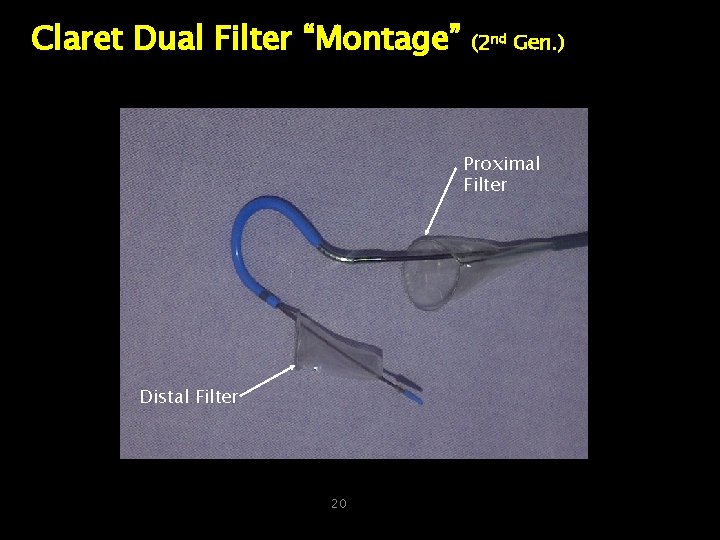
Claret Dual Filter “Montage” (2 nd Gen. ) Proximal Filter Distal Filter 20

Clinical Data – FIM Study Outline Pilot Study to evaluate safety and effectiveness of the Claret Medical filter Multi-Center, prospective study Sites: Germany (Elisabeth-Krankenhaus Hospital – Essen and Sigmund-Freud University Hospital – Bonn, Dante Pazzanese, São Paulo, Brazil) Drs. Eberhard Grube, Christoph Naber, Georg Nickenig, Alex Abizaid Primary Endpoint: Technical Success (Delivery and Retrieval) Devices Used during TAVI: • Medtronic Core. Valve 26 mm Aortic Valve • Medtronic Core. Valve 29 mm Aortic Valve • Edwards Sapien 23 mm Aortic Valve • Edwards Sapien 26 mm Aortic Valve • Numed Valvuloplasty Balloon (various sizes) TAVI Introduction Sites: Trans-Femoral and Left Subclavian

Clinical Data – Patient Demographics Number of Patients Treated N=40 Access (4) Radial / (36) Brachial Patient Age (avg) 76 -91 (82. 8) Female / Male 21/19 Euro. SCORE 10 -35 Valve Manufacture 38 – Core. Valve (95%) 2– Edwards Sapien (5%) Current Average Deployment Time 4 minutes Brachiocephalic Filter Placement 38/40 (95%) Left Carotid Filter Placement 37/40 (93%) Embolic Material found in 95% of cases
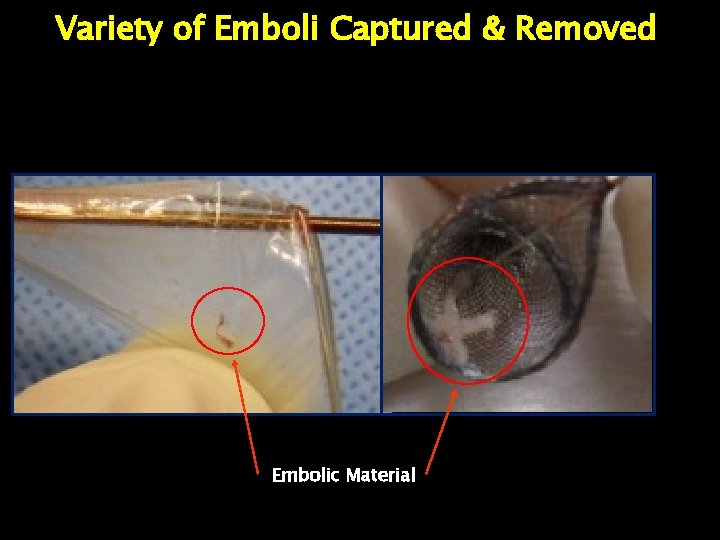
Variety of Emboli Captured & Removed Embolic Material
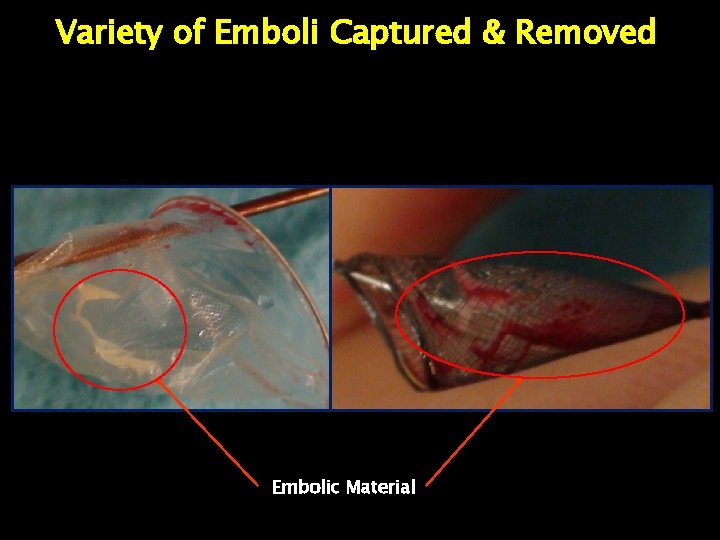
Variety of Emboli Captured & Removed Embolic Material
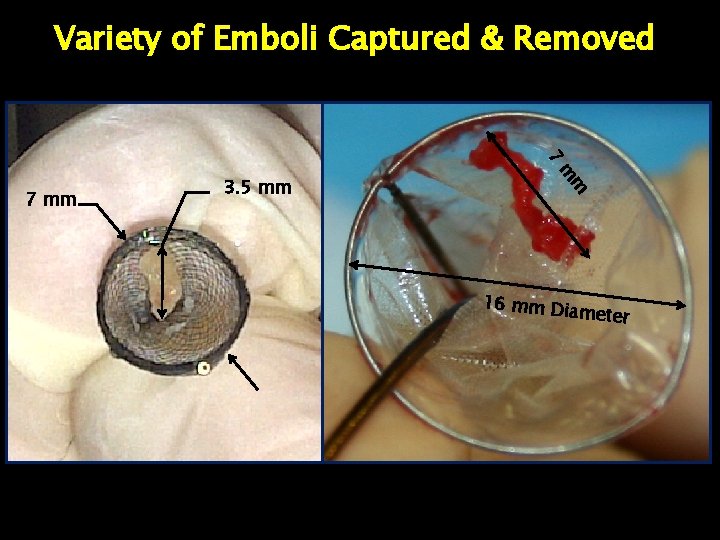
Variety of Emboli Captured & Removed 7 m 3. 5 mm m 7 mm 16 mm Di ameter
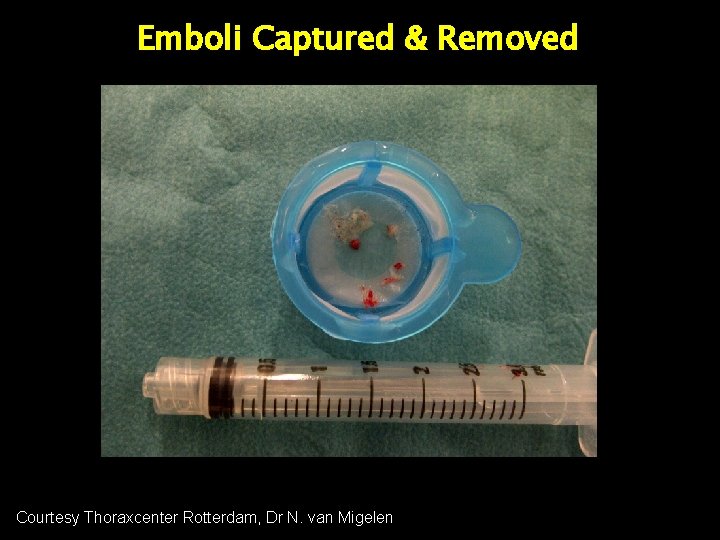
Emboli Captured & Removed 7 m 3. 5 mm m 7 mm 16 mm Di ameter Courtesy Thoraxcenter Rotterdam, Dr N. van Migelen
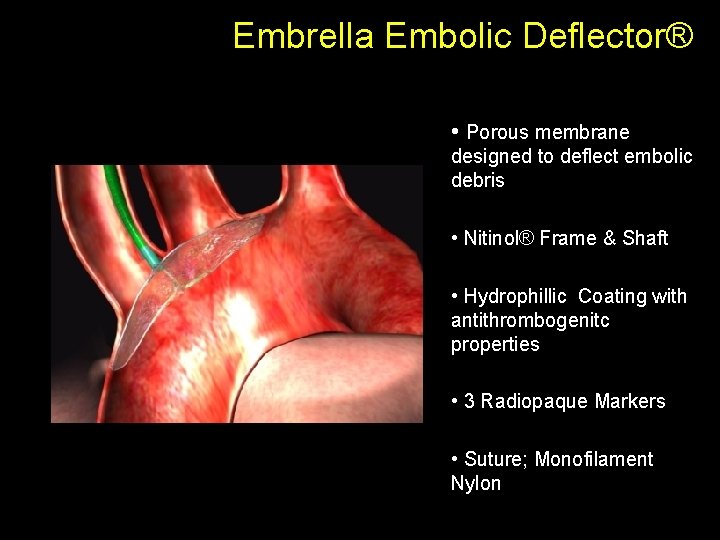
Embrella Embolic Deflector® • Porous membrane designed to deflect embolic debris • Nitinol® Frame & Shaft • Hydrophillic Coating with antithrombogenitc properties • 3 Radiopaque Markers • Suture; Monofilament Nylon

Embrella: Study Centers Site Number Center Principal Investigator Number of Subjects Enrolled 01 St. Paul’s Hospital Vancouver, BC John Webb, M. D. 5 02 HELIOS Hospital Center Siegburg, DE Ralf Müller, M. D. Ulrich Gerckens, M. D. 10 03 University Hospital Hamburg, DE Joachim Schofer, M. D. 5

Average Number of Lesions/Subject 6. 0 4. 69 3. 2 * Embrella subjects 1 BAV, 14 TF TAVI **Ghanem et al, J Am Coll Cardiol 2010; 55: 1427– 32 (TF TAVI) 31 ***Astarci et al, Abstract presentation EACTS 2010 (TF TAVI) ***

Summary Stoke due to embolized plaques from the arch or the diseased native valve remains an issue with variable reported incidences (1. 4 -9. 3%) Event range in reported studies is partially due to differences in endpoint definitions which is now taken care of by the joint VARC efforts Various protection tools are currently in development with remarkable demonstration of usefulness and efficacy We have to learn more about embolic risk predictors, dividing patients in low risk and high risk, who are likely to benefit from protective tools. Until then. . .

Summary Given prior surgical and present TAVI experiences with new cerebral lesions occuring in up to 84% of cases… and given the highly embolic environment within the aortic arch and the valve… TAVI procedures should be performed under cerebral protection in order to lower or even eliminate procedurally caused cerebral events! The embolic material that we are liberating during the TAVI procedure cannot be beneficial to our patients.

Thank you!
- Slides: 28