CRT 2012 Washington DC February 5 2012 Core

CRT 2012 Washington DC, February 5, 2012 Core Valve 2012 Current Data and Alternative Access Eberhard Grube MD, FACC, FSCAI University Bonn, School of Medicine, Bonn, Germany Hospital Alemão Oswaldo Cruz - Dante Pazzanese, São Paulo, Brazil Stanford University, Palo Alto, California, USA

Eberhard Grube, MD § Honoraria: § Boston Scientific Corporation § Biosensors International § Consulting Fees: § Medtronic Core. Valve § Boston Scientific Corporation § Cordis Corporation § Johnson & Johnson § Abbott Vascular

Eberhard Grube, MD § Ownership Interest (Stocks, Stock Options, or other Ownership Interest): § Medtronic Core Valve § Biosensors International My presentation will include off-label discussions: Off-label use of Stents and Valve Prosthesis

Medtronic Core. Valve® System Summary of Clinical Experience
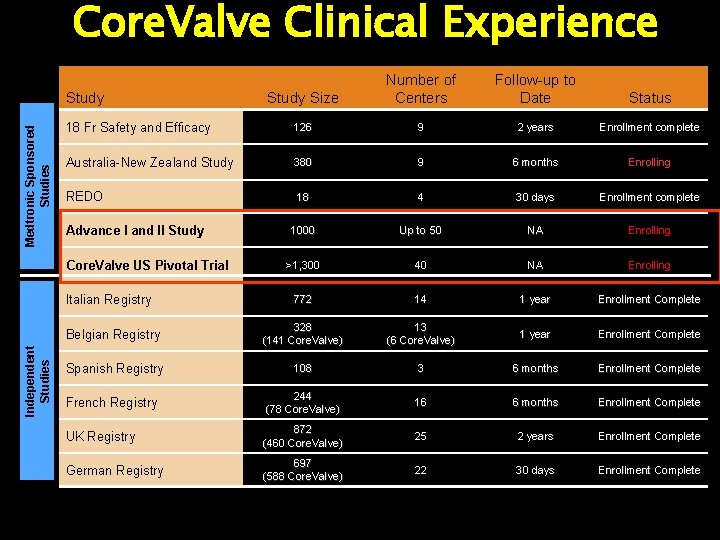
Core. Valve Clinical Experience Study Size Number of Centers Follow-up to Date Status 18 Fr Safety and Efficacy 126 9 2 years Enrollment complete Australia-New Zealand Study 380 9 6 months Enrolling REDO 18 4 30 days Enrollment complete 1000 Up to 50 NA Enrolling >1, 300 40 NA Enrolling 772 14 1 year Enrollment Complete Belgian Registry 328 (141 Core. Valve) 13 (6 Core. Valve) 1 year Enrollment Complete Spanish Registry 108 3 6 months Enrollment Complete French Registry 244 (78 Core. Valve) 16 6 months Enrollment Complete UK Registry 872 (460 Core. Valve) 25 2 years Enrollment Complete German Registry 697 (588 Core. Valve) 22 30 days Enrollment Complete Medtronic Sponsored Studies Study Advance I and II Study Core. Valve US Pivotal Trial Independent Studies Italian Registry
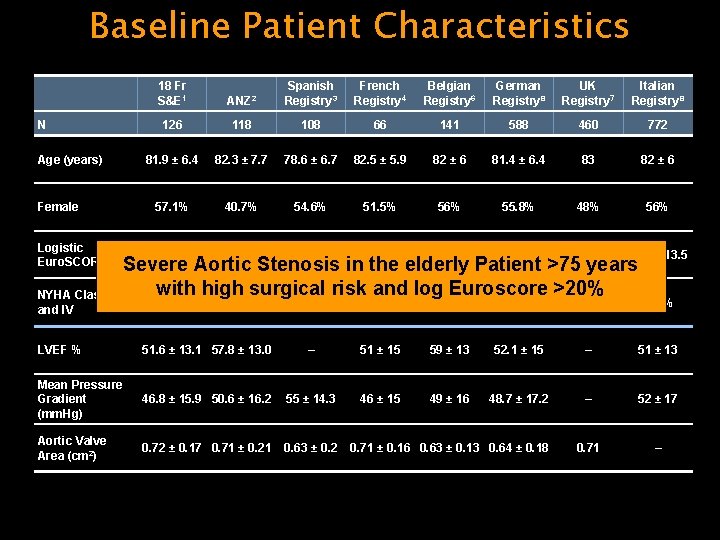
Baseline Patient Characteristics N Age (years) Female Logistic Euro. SCORE (%) 18 Fr S&E 1 ANZ 2 Spanish Registry 3 French Registry 4 Belgian Registry 6 German Registry 8 UK Registry 7 Italian Registry 8 126 118 108 66 141 588 460 772 81. 9 ± 6. 4 82. 3 ± 7. 7 78. 6 ± 6. 7 82. 5 ± 5. 9 82 ± 6 81. 4 ± 6. 4 83 82 ± 6 57. 1% 40. 7% 54. 6% 51. 5% 56% 55. 8% 48% 56% 23. 4 ± 13. 8 18. 4 ± 11. 9 16 ± 13. 9 24. 7 ± 11. 2 25 ± 15 20. 8 ± 13. 3 20. 3 22. 9 ± 13. 5 74. 6% 83. 9% 58. 4% – 78% 88. 2% 74% 70. 6% Severe Aortic Stenosis in the elderly Patient >75 years with high surgical risk and log Euroscore >20% NYHA Class III and IV LVEF % 51. 6 ± 13. 1 57. 8 ± 13. 0 – 51 ± 15 59 ± 13 52. 1 ± 15 – 51 ± 13 Mean Pressure Gradient (mm. Hg) 46. 8 ± 15. 9 50. 6 ± 16. 2 55 ± 14. 3 46 ± 15 49 ± 16 48. 7 ± 17. 2 – 52 ± 17 Aortic Valve Area (cm 2) 0. 72 ± 0. 17 0. 71 ± 0. 21 0. 63 ± 0. 2 0. 71 – 0. 71 ± 0. 16 0. 63 ± 0. 13 0. 64 ± 0. 18 See Reference slide for sources.

Core. Valve Meta-analysis • N=2156 patients • 7 national registries – Australia-New Zealand, Belgium, France, Germany, Italy, Spain, and UK • Mean age of the population: 81. 6 years • Male: 47% • New York Heart Association class III and IV: 77% • Mean Logistic Euro. SCORE: 21. 3% (range, 16. 0 -24. 7) • Mean aortic valve area: 0. 63 cm 2 • Mean gradient: 49. 7 mm Hg 1. Ruiz CE, et al. Weighted meta-analysis of early and late clinical outcomes after Core. Valve® – TAVI in seven national registries. Presented at: Euro. PCR; May 17 -20, 2011; Paris, France. Analysis sponsored by Medtronic, Inc.
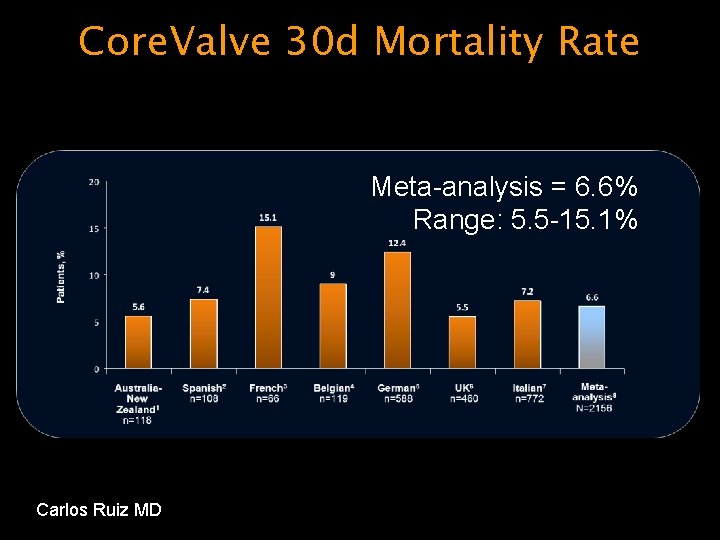
Core. Valve 30 d Mortality Rate Meta-analysis = 6. 6% Range: 5. 5 -15. 1% Carlos Ruiz MD
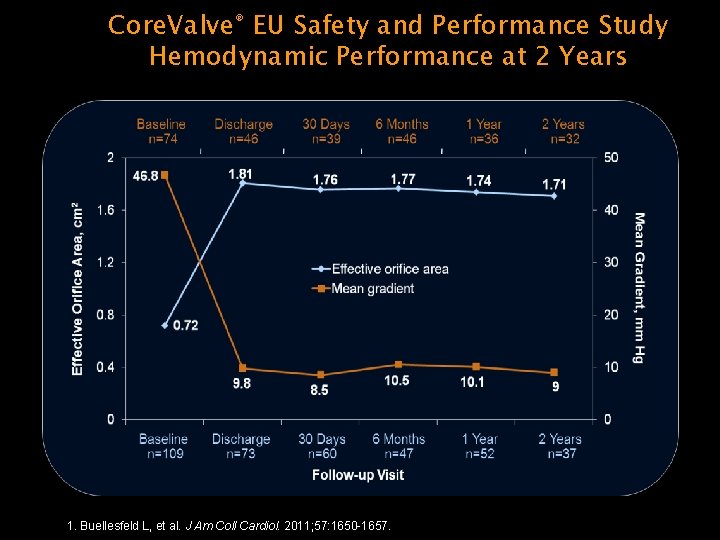
Core. Valve® EU Safety and Performance Study Hemodynamic Performance at 2 Years 1. Buellesfeld L, et al. J Am Coll Cardiol. 2011; 57: 1650 -1657.
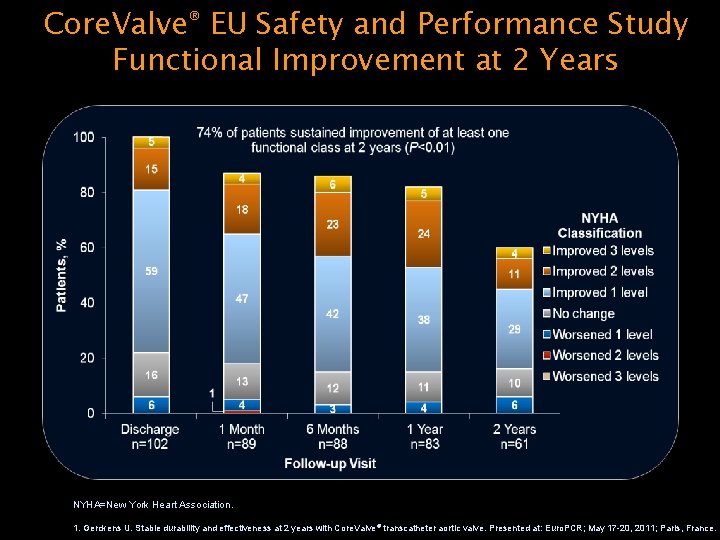
Core. Valve® EU Safety and Performance Study Functional Improvement at 2 Years NYHA=New York Heart Association. 1. Gerckens U. Stable durability and effectiveness at 2 years with Core. Valve® transcatheter aortic valve. Presented at: Euro. PCR; May 17 -20, 2011; Paris, France.
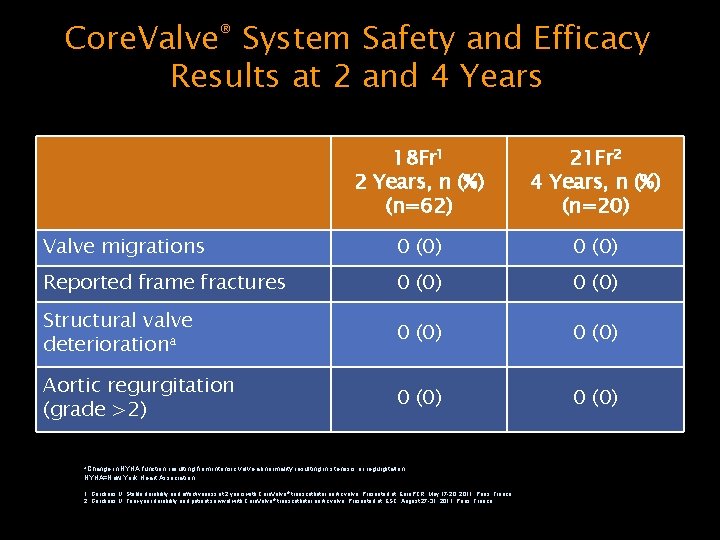
Core. Valve® System Safety and Efficacy Results at 2 and 4 Years 18 Fr 1 2 Years, n (%) (n=62) 21 Fr 2 4 Years, n (%) (n=20) Valve migrations 0 (0) Reported frame fractures 0 (0) Structural valve deteriorationa 0 (0) Aortic regurgitation (grade >2) 0 (0) a. Change in NYHA function resulting from intrinsic valve abnormality resulting in stenosis or regurgitation. NYHA=New York Heart Association. 1. Gerckens U. Stable durability and effectiveness at 2 years with Core. Valve ® transcatheter aortic valve. Presented at: Euro. PCR; May 17 -20, 2011; Paris, France. 2. Gerckens U. Four-year durability and patient survival with Core. Valve ® transcatheter aortic valve. Presented at: ESC; August 27 -31, 2011; Paris, France.
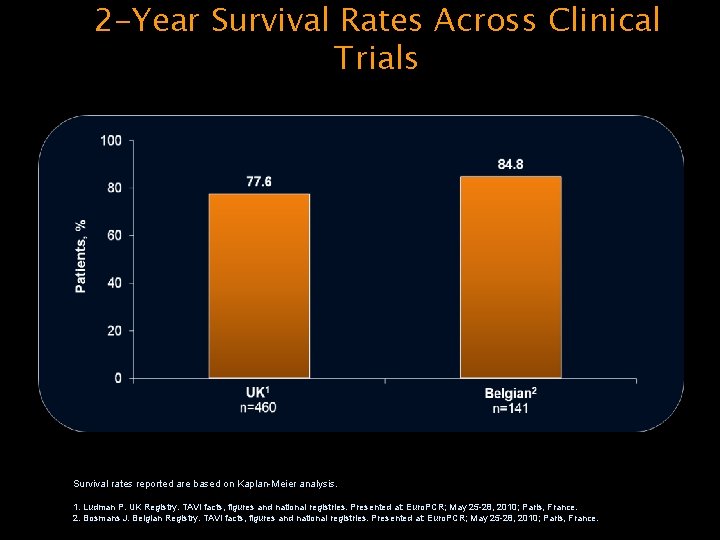
2 -Year Survival Rates Across Clinical Trials Survival rates reported are based on Kaplan-Meier analysis. 1. Ludman P. UK Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France. 2. Bosmans J. Belgian Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France.

Predictors of Adverse Outcome and Complications
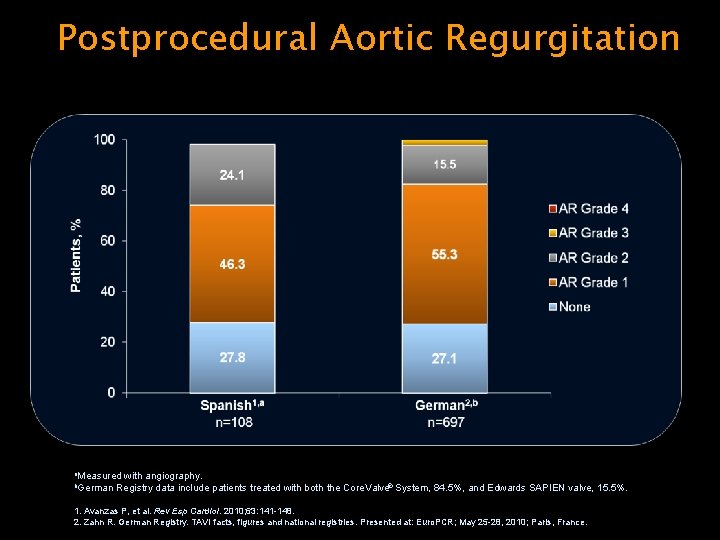
Postprocedural Aortic Regurgitation a. Measured b. German with angiography. Registry data include patients treated with both the Core. Valve® System, 84. 5%, and Edwards SAPIEN valve, 15. 5%. 1. Avanzas P, et al. Rev Esp Cardiol. 2010; 63: 141 -148. 2. Zahn R. German Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France.
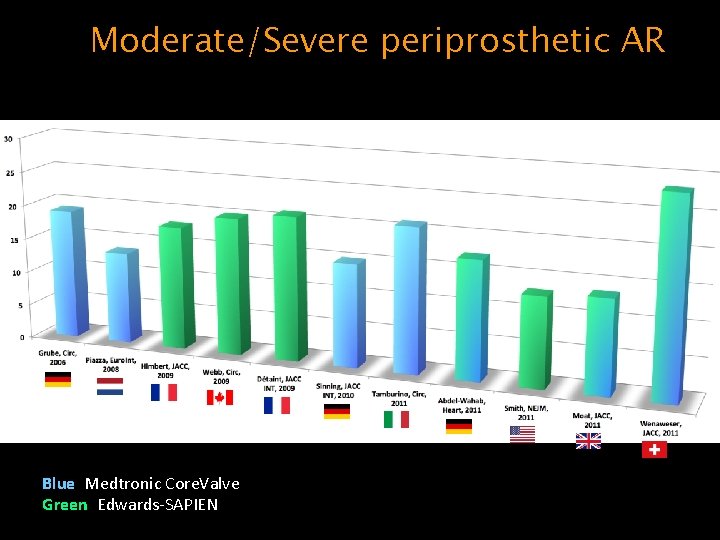
Moderate/Severe periprosthetic AR Blue: Medtronic Core. Valve Green: Edwards-SAPIEN
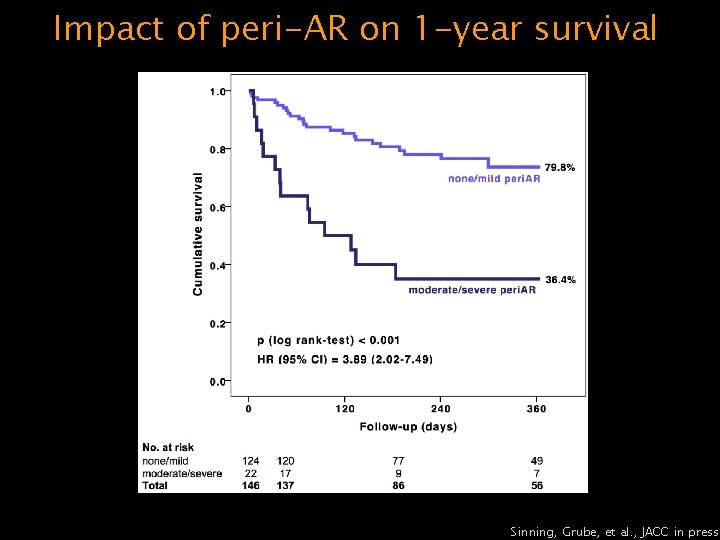
Impact of peri-AR on 1 -year survival Sinning, Grube, et al. , JACC in press

Multivariate analysis: Independent predictors of 30 -day mortality Multivariate Cox Regression with adjustment of several baseline variables Multivariate adjustment included: Age, male gender, diabetes, coronary artery disease, previous myocardial infarction, previous CABG, previous stroke, left-ventricular ejection fraction ≤ 30%, chronic renal failure, COPD, pulmonary hypertension, carotid artery stenosis, porcelain aorta, aortic aneurysm, and peripheral arterial disease. Sinning et al. , AHA 2011, Orlando

Stroke in TAVR Neurologic events are higher with TAVI than with surgical AVR or medical therapy. A detailed collaboration with the neurology service is a critical part of program building to standardize the evaluation after an event
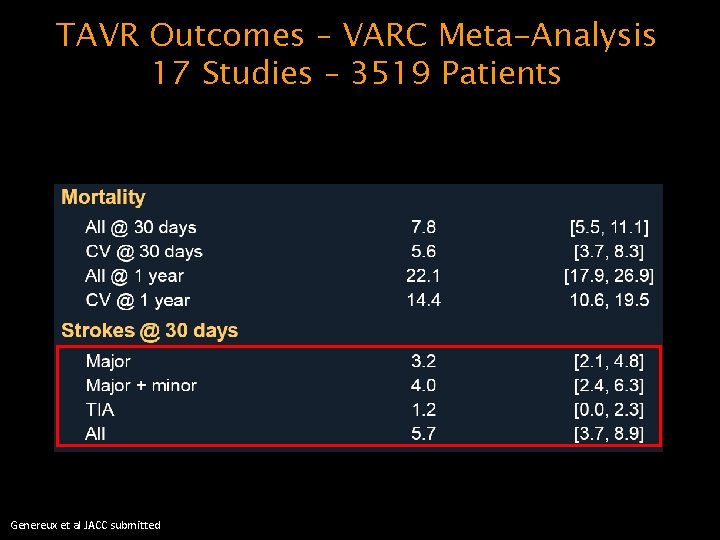
TAVR Outcomes – VARC Meta-Analysis 17 Studies – 3519 Patients Genereux et al JACC submitted
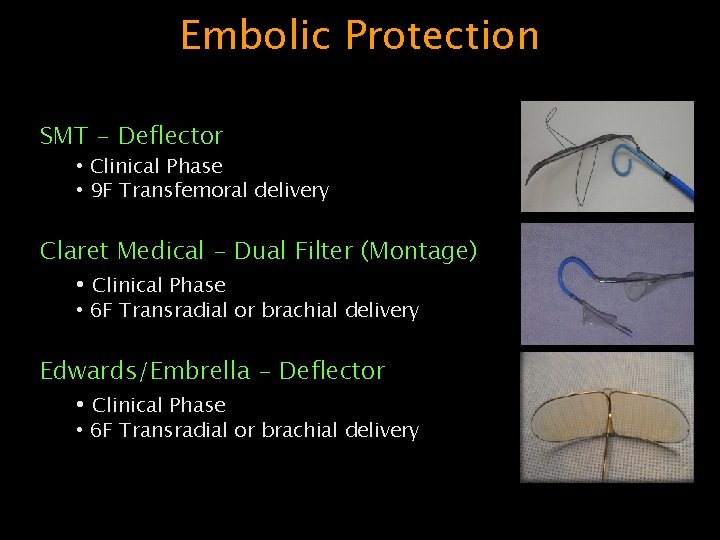
Embolic Protection SMT - Deflector • Clinical Phase • 9 F Transfemoral delivery Claret Medical - Dual Filter (Montage) • Clinical Phase • 6 F Transradial or brachial delivery Edwards/Embrella - Deflector • Clinical Phase • 6 F Transradial or brachial delivery
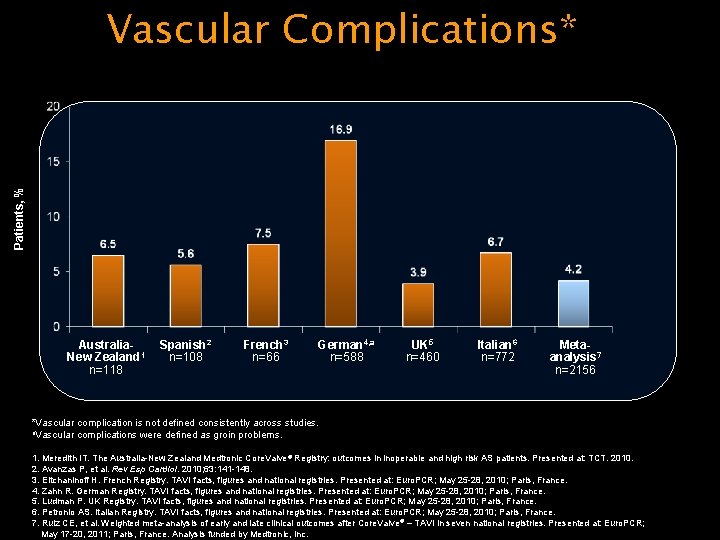
Patients, % Vascular Complications* Australia. New Zealand 1 n=118 Spanish 2 n=108 French 3 n=66 German 4, a n=588 UK 5 n=460 Italian 6 n=772 Metaanalysis 7 n=2156 *Vascular complication is not defined consistently across studies. a. Vascular complications were defined as groin problems. 1. Meredith IT. The Australia-New Zealand Medtronic Core. Valve® Registry: outcomes in inoperable and high risk AS patients. Presented at: TCT. 2010. 2. Avanzas P, et al. Rev Esp Cardiol. 2010; 63: 141 -148. 3. Eltchaninoff H. French Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France. 4. Zahn R. German Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France. 5. Ludman P. UK Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France. 6. Petronio AS. Italian Registry. TAVI facts, figures and national registries. Presented at: Euro. PCR; May 25 -28, 2010; Paris, France. 7. Ruiz CE, et al. Weighted meta-analysis of early and late clinical outcomes after Core. Valve® – TAVI in seven national registries. Presented at: Euro. PCR; May 17 -20, 2011; Paris, France. Analysis funded by Medtronic, Inc.
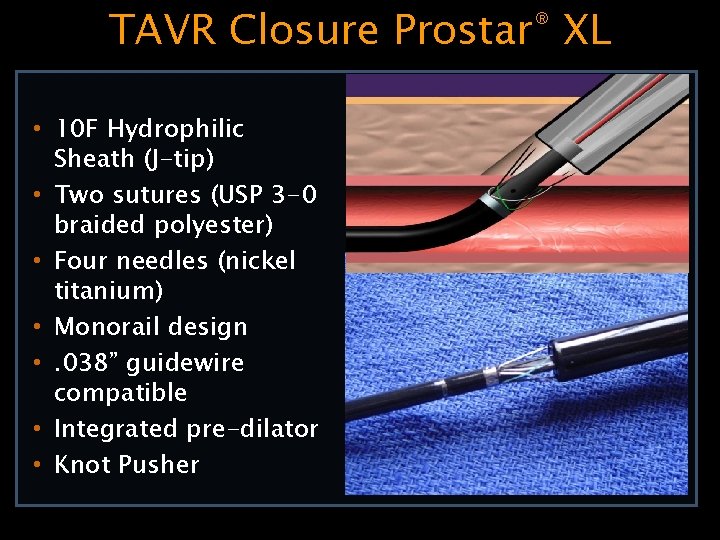
TAVR Closure Prostar® XL • 10 F Hydrophilic Sheath (J-tip) • Two sutures (USP 3 -0 braided polyester) • Four needles (nickel titanium) • Monorail design • . 038” guidewire compatible • Integrated pre-dilator • Knot Pusher
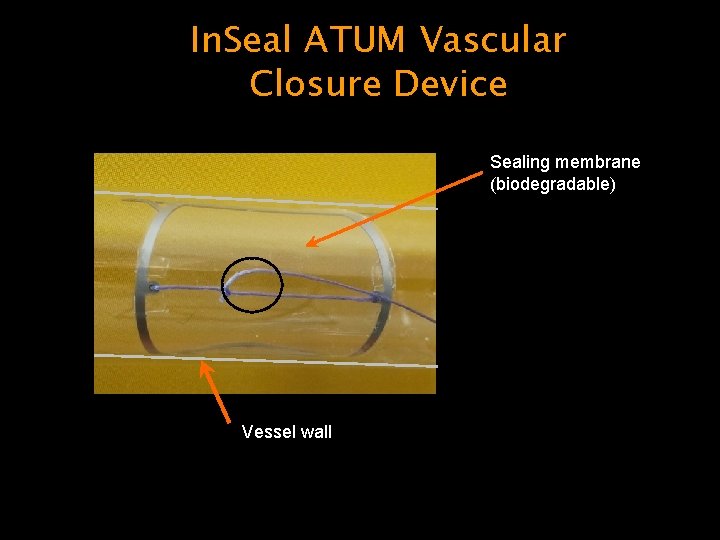
In. Seal ATUM Vascular Closure Device Sealing membrane (biodegradable) Vessel wall
Atum VCD FIM Study Patient #001 24 hrs Post Procedure
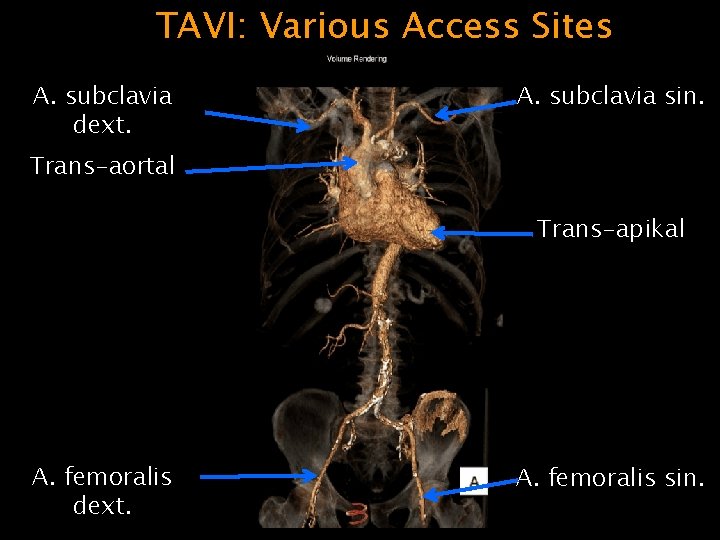
TAVI: Various Access Sites A. subclavia dext. A. subclavia sin. Trans-aortal Trans-apikal A. femoralis dext. A. femoralis sin.
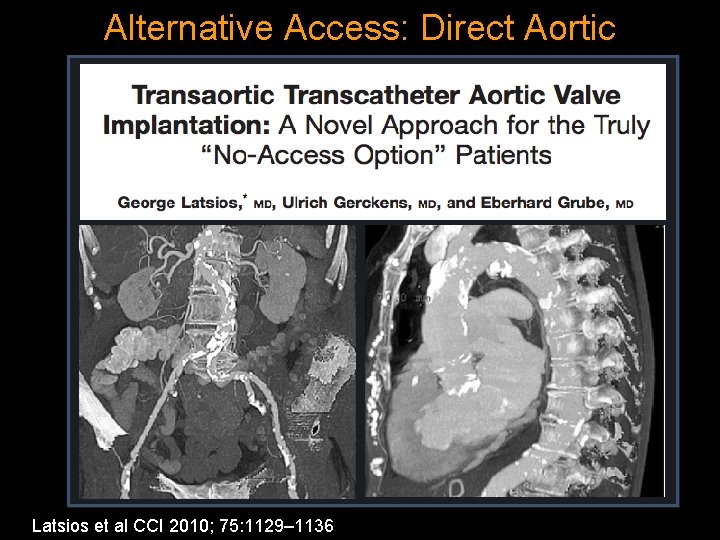
Alternative Access: Direct Aortic Latsios et al CCI 2010; 75: 1129– 1136

Core. Valve® System Ongoing Studies Evaluate procedural best practices • ADVANCE Study 3 --- ACC 2012 – – – Designed to evaluate clinical outcomes in “real-world” patients • ADVANCE-II Study 1 - 1017 patients in up to 44 centers followed for up to 10 years Enrollment and data analysis complete Designed to characterize current Core. Valve® System implantation best practices - >100 patients Evaluate performance in expanded patient population • SURTAVI 1 - Randomized controlled trial of TAVI vs SAVR studying broader patient population (intermediate risk) • REDO Study 2 - Outcomes in patients with a failing aortic bioprosthesis

Thank you very much!
- Slides: 29