CRT 2011 Impella Innovation Technical Tips Samin Sharma
















- Slides: 41

CRT 2011 Impella Innovation: Technical Tips Samin Sharma, MD, FACC Professor of Medicine (Cardiology) Director Cardiac Cath Lab & Intervention Co-Director Cardiovascular Institute Mount Sinai Hospital, NY

Conflict of Interest Research Grant support & Speaker Bureau for: - BSc - Abbott Vascular - DSI/Lilly - The Medicines Co - Abiomed - Angioscore

Outline • Technology Overview • Clinical Experience and Investigations • Future Directions

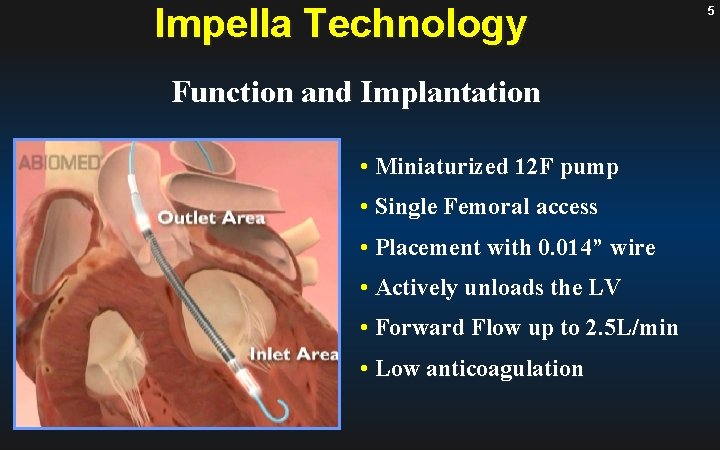
Impella Technology Function and Implantation • Miniaturized 12 F pump • Single Femoral access • Placement with 0. 014” wire • Actively unloads the LV • Forward Flow up to 2. 5 L/min • Low anticoagulation 5

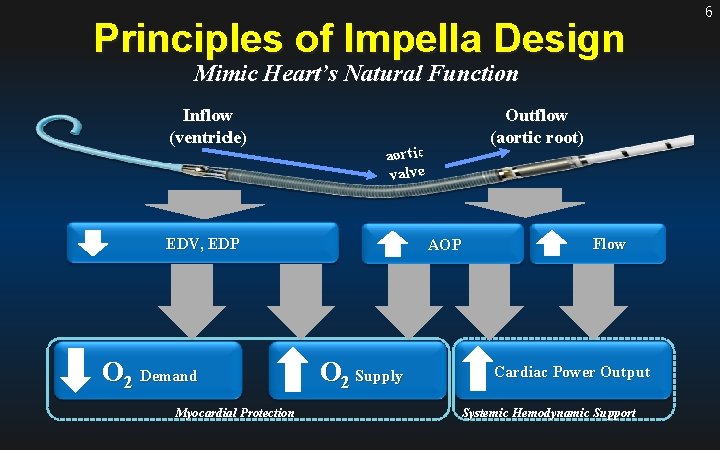
Principles of Impella Design Mimic Heart’s Natural Function Inflow (ventricle) aortic valve EDV, EDP O 2 Demand Myocardial Protection Outflow (aortic root) AOP O 2 Supply Flow Cardiac Power Output Systemic Hemodynamic Support 6

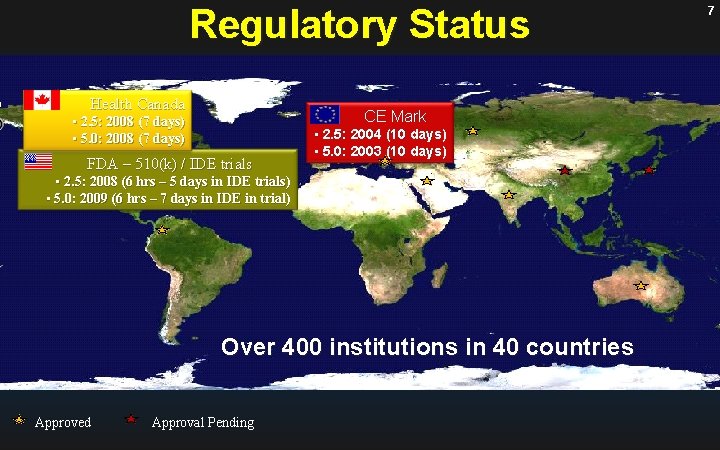
Regulatory Status Health Canada CE Mark • 2. 5: 2008 (7 days) • 5. 0: 2008 (7 days) FDA – 510(k) / IDE trials • 2. 5: 2004 (10 days) • 5. 0: 2003 (10 days) • 2. 5: 2008 (6 hrs – 5 days in IDE trials) • 5. 0: 2009 (6 hrs – 7 days in IDE in trial) Over 400 institutions in 40 countries Approved Approval Pending 7

Outline • Technology Overview • Clinical Experience and Investigations • Future Directions

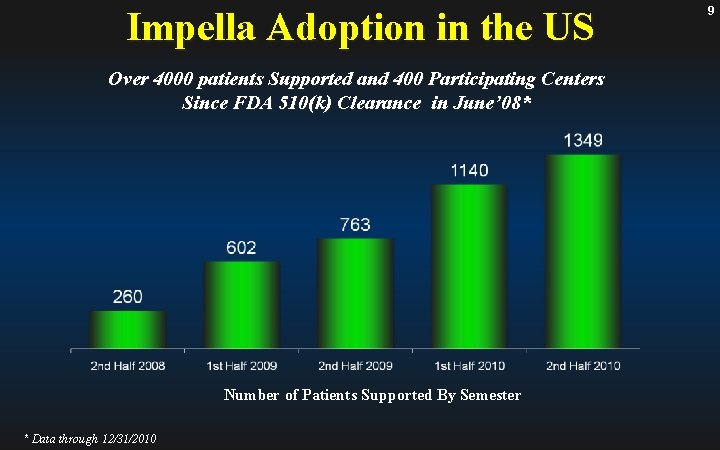
Impella Adoption in the US Over 4000 patients Supported and 400 Participating Centers Since FDA 510(k) Clearance in June’ 08* Number of Patients Supported By Semester * Data through 12/31/2010 9

Impella 2. 5: High Risk PCI Insight from the USpella Registry & PROTECT II Trial

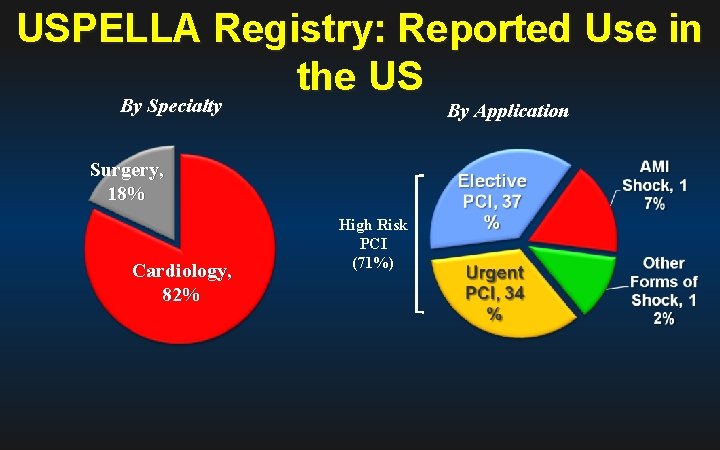
USPELLA Registry: Reported Use in the US By Specialty By Application Surgery, 18% Cardiology, 82% High Risk PCI (71%)

USPELLA Registry: Patients Represent The Most Complex Set of PCI Performed in Contemporary Times Patient Characteristics Age (yrs) Gender (Male in %) Procedure (Elective vs Urgent) LVEF (%) Mean ± SD or % 70 ± 11 74% 45% vs 55% 30 ± 16 LVEF < 35% 71% Unprotected LM 51% Unprotected LM or LPC 52% Multivessel Disease 88% Nb of Segments Treated 2. 5 ± 1. 2 STS Score 5. 6 ± 6. 5 Syntax Score 36 ± 15

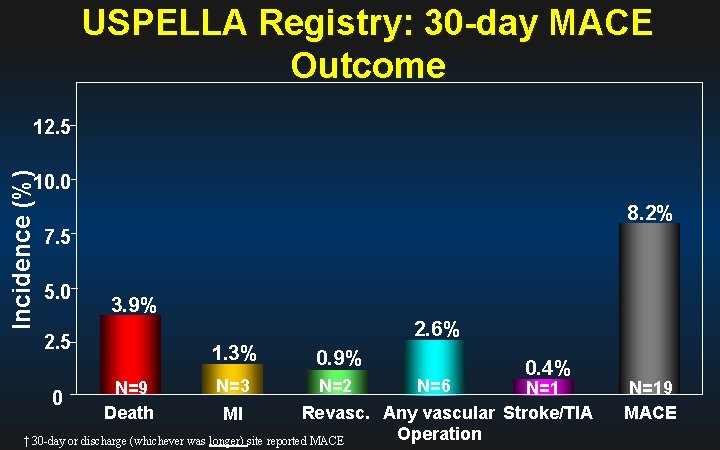
USPELLA Registry: 30 -day MACE Outcome Incidence (%) 12. 5 10. 0 8. 2% 7. 5 5. 0 3. 9% 2. 6% 2. 5 1. 3% 0. 4% N=6 N=2 N=1 0 Revasc. Any vascular Stroke/TIA MI Operation † 30 -day or discharge (whichever was longer) site reported MACE N=9 Death N=3 0. 9% N=19 MACE

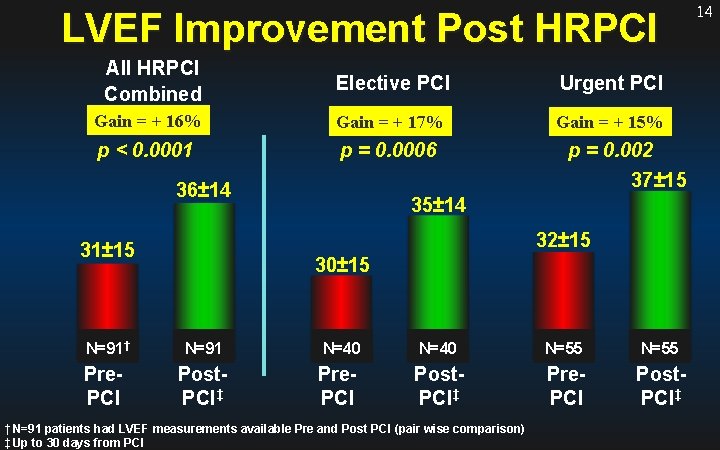
LVEF Improvement Post HRPCI All HRPCI Combined Elective PCI Urgent PCI Gain = + 16% Gain = + 17% Gain = + 15% p < 0. 0001 p = 0. 0006 36± 14 p = 0. 002 37± 15 35± 14 32± 15 31± 15 30± 15 N=91† N=91 N=40 N=55 Pre. PCI Post. PCI‡ † N=91 patients had LVEF measurements available Pre and Post PCI (pair wise comparison) ‡ Up to 30 days from PCI 14

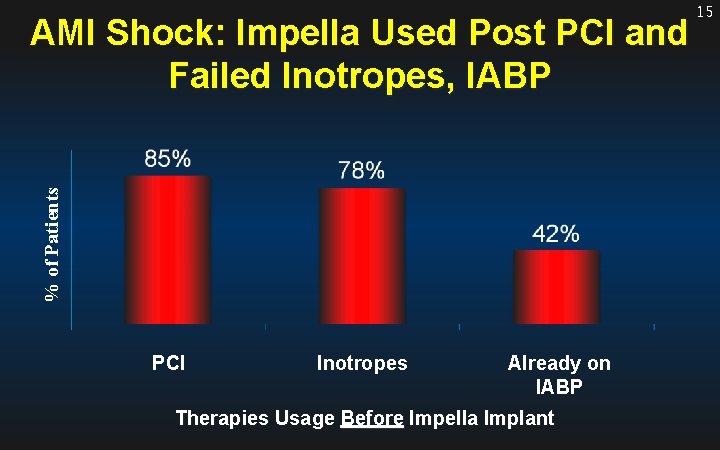
% of Patients AMI Shock: Impella Used Post PCI and Failed Inotropes, IABP PCI Inotropes Already on IABP Therapies Usage Before Impella Implant 15

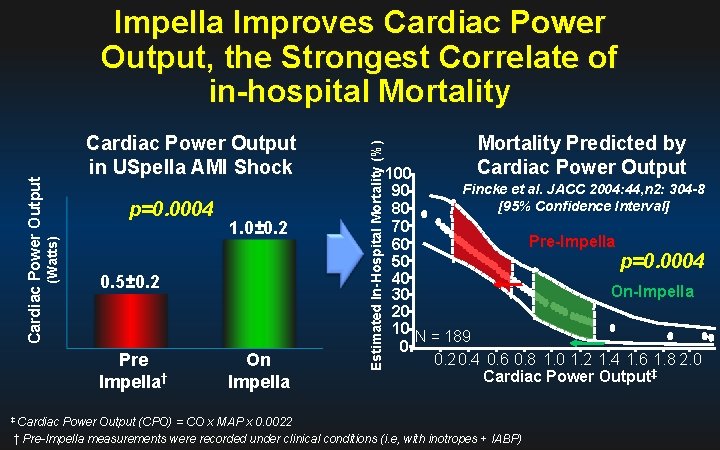
p=0. 0004 0. 5± 0. 2 Pre Impella† ‡ Cardiac 1. 0± 0. 2 On Impella Estimated In-Hospital Mortality (%) Cardiac Power Output in USpella AMI Shock (Watts) Cardiac Power Output Impella Improves Cardiac Power Output, the Strongest Correlate of in-hospital Mortality Predicted by Cardiac Power Output 100 Fincke et al. JACC 2004: 44, n 2: 304 -8 90 [95% Confidence Interval] 80 70 Pre-Impella 60 50 p=0. 0004 40 On-Impella 30 20 10 N = 189 0 0. 2 0. 4 0. 6 0. 8 1. 0 1. 2 1. 4 1. 6 1. 8 2. 0 Cardiac Power Output‡ Power Output (CPO) = CO x MAP x 0. 0022 † Pre-Impella measurements were recorded under clinical conditions (i. e, with inotropes + IABP)

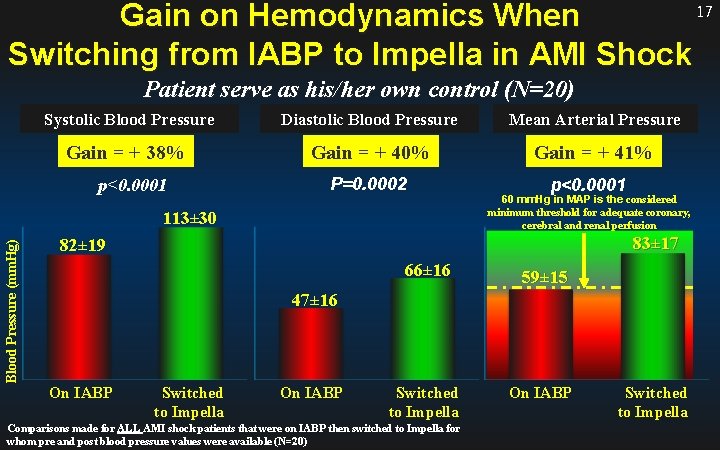
17 Gain on Hemodynamics When Switching from IABP to Impella in AMI Shock Patient serve as his/her own control (N=20) Systolic Blood Pressure Diastolic Blood Pressure Mean Arterial Pressure Gain = + 38% Gain = + 40% Gain = + 41% p<0. 0001 P=0. 0002 Blood Pressure (mm. Hg) 113± 30 p<0. 0001 60 mm. Hg in MAP is the considered minimum threshold for adequate coronary, cerebral and renal perfusion 83± 17 82± 19 66± 16 59± 15 47± 16 On IABP Switched to Impella Comparisons made for ALL AMI shock patients that were on IABP then switched to Impella for whom pre and post blood pressure values were available (N=20) On IABP Switched to Impella

Timing of Impella Support vs Outcome Impella ON “Pre-PCI” Impella ON “Post PCI” (n=22) (n=34) 64 (53 -84) 63 (19 -83) 0. 4 Cardiac Index Pre-Impella (L/m 2) 2. 0± 0. 2 2. 0± 0. 6 0. 9 Heart rate Pre-Impella (beat/min) 73± 24 77± 26 0. 6 MAP Pre-Impella (mm. Hg) 54± 22 50± 17 0. 4 LVEF Pre-Impella (%) 26± 11 26± 12 0. 9 60 80 0. 2 23% 56% 0. 02 p-value Baseline Characteristics Age (Median, range in years) STEMI presentation (%) Outcome 30 -day mortality

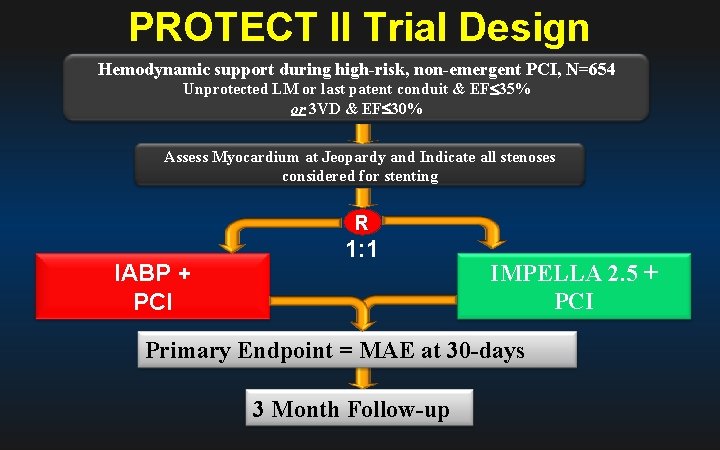
PROTECT II Trial Design Hemodynamic support during high-risk, non-emergent PCI, N=654 Unprotected LM or last patent conduit & EF 35% or 3 VD & EF 30% Assess Myocardium at Jeopardy and Indicate all stenoses considered for stenting R IABP + PCI 1: 1 IMPELLA 2. 5 + PCI Primary Endpoint = MAE at 30 -days 3 Month Follow-up

PROTECT II Interim Results Per Protocol Primary Endpoint: 30 -day Combined Major Adverse Events (MAE) Patients Impella MAE IAB MAE p value All* (N=305, 100%) 38% 43% 0. 40 No atherectomy (N=267, 88%) 32% 43% 0. 06 Atherectomy (N=38, 12%) 72% 46% 0. 12 (Impella N=25, IAB N=13) * For the entire study population, Impella significantly reduced out of hospital MAEs by 52% compared to IAB for the duration of the 90 day monitoring period (p=0. 02, N=302)

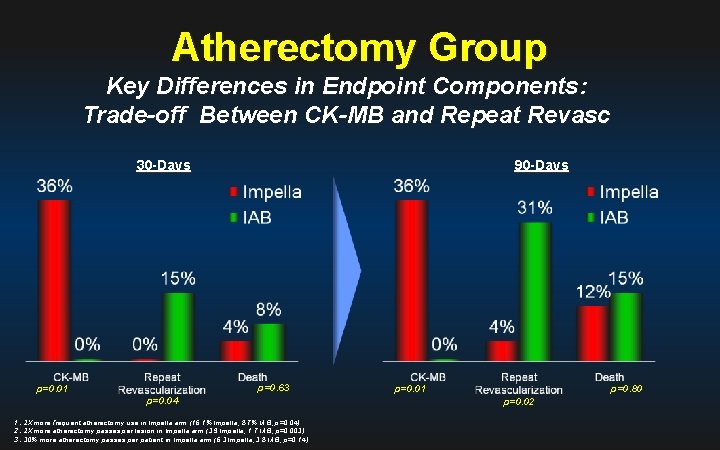
Atherectomy Group Key Differences in Endpoint Components: Trade-off Between CK-MB and Repeat Revasc 30 -Days 90 -Days p=0. 63 p=0. 01 p=0. 04 1. 2 X more frequent atherectomy use in Impella arm (16. 1% Impella, 8. 7% IAB, p=0. 04) 2. 2 X more atherectomy passes per lesion in Impella arm (3. 9 Impella, 1. 7 IAB, p=0. 003) 3. 30% more atherectomy passes per patient in Impella arm (6. 3 Impella, 3. 8 IAB, p=0. 14) p=0. 80 p=0. 01 p=0. 02

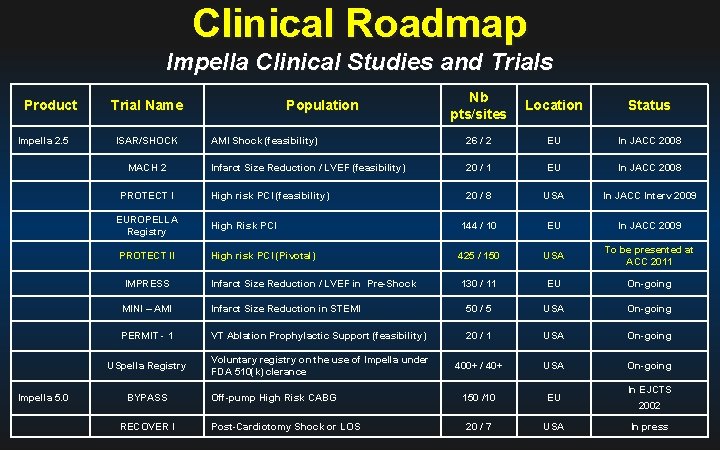
Clinical Roadmap Impella Clinical Studies and Trials Product Impella 2. 5 ISAR/SHOCK MACH 2 PROTECT I Impella 5. 0 Nb pts/sites Location Status AMI Shock (feasibility) 26 / 2 EU In JACC 2008 Infarct Size Reduction / LVEF (feasibility) 20 / 1 EU In JACC 2008 High risk PCI (feasibility) 20 / 8 USA In JACC Interv 2009 Trial Name Population EUROPELLA Registry High Risk PCI 144 / 10 EU In JACC 2009 PROTECT II High risk PCI (Pivotal) 425 / 150 USA To be presented at ACC 2011 IMPRESS Infarct Size Reduction / LVEF in Pre-Shock 130 / 11 EU On-going MINI – AMI Infarct Size Reduction in STEMI 50 / 5 USA On-going PERMIT - 1 VT Ablation Prophylactic Support (feasibility) 20 / 1 USA On-going USpella Registry Voluntary registry on the use of Impella under FDA 510(k) clerance 400+ / 40+ USA On-going 150 /10 EU 20 / 7 USA BYPASS RECOVER I Off-pump High Risk CABG Post-Cardiotomy Shock or LOS In EJCTS 2002 In press

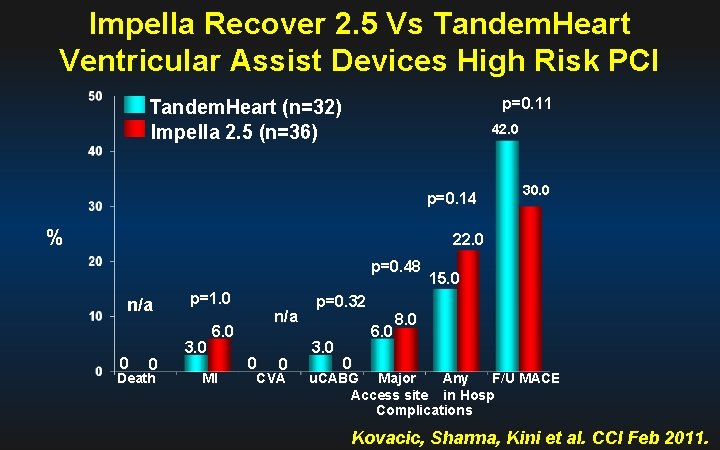
Impella Recover 2. 5 Vs Tandem. Heart Ventricular Assist Devices High Risk PCI p=0. 11 Tandem. Heart (n=32) Impella 2. 5 (n=36) 42. 0 p=0. 14 % 30. 0 22. 0 p=0. 48 n/a 0 0 Death p=1. 0 3. 0 n/a 6. 0 MI 0 0 CVA p=0. 32 3. 0 6. 0 15. 0 8. 0 0 u. CABG Major Any F/U MACE Access site in Hosp Complications Kovacic, Sharma, Kini et al. CCI Feb 2011.

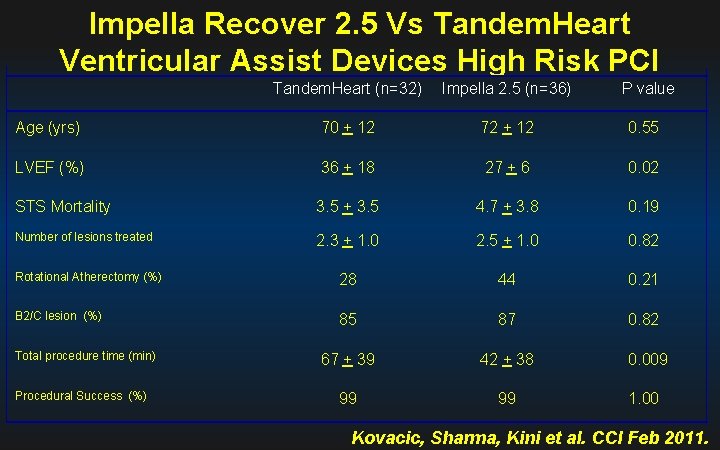
Impella Recover 2. 5 Vs Tandem. Heart Ventricular Assist Devices High Risk PCI Tandem. Heart (n=32) Impella 2. 5 (n=36) P value Age (yrs) 70 + 12 72 + 12 0. 55 LVEF (%) 36 + 18 27 + 6 0. 02 STS Mortality 3. 5 + 3. 5 4. 7 + 3. 8 0. 19 Number of lesions treated 2. 3 + 1. 0 2. 5 + 1. 0 0. 82 Rotational Atherectomy (%) 28 44 0. 21 B 2/C lesion (%) 85 87 0. 82 67 + 39 42 + 38 0. 009 99 99 1. 00 Total procedure time (min) Procedural Success (%) Kovacic, Sharma, Kini et al. CCI Feb 2011.

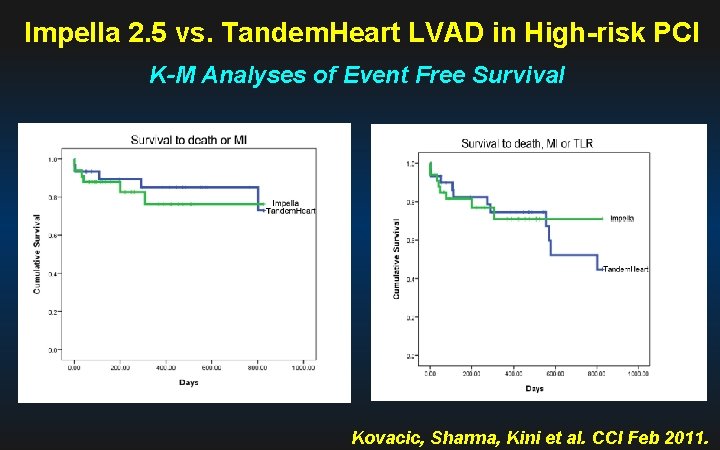
Impella 2. 5 vs. Tandem. Heart LVAD in High-risk PCI K-M Analyses of Event Free Survival Kovacic, Sharma, Kini et al. CCI Feb 2011.

Outline • Technology Overview • Clinical Experience and Investigations • Future Directions

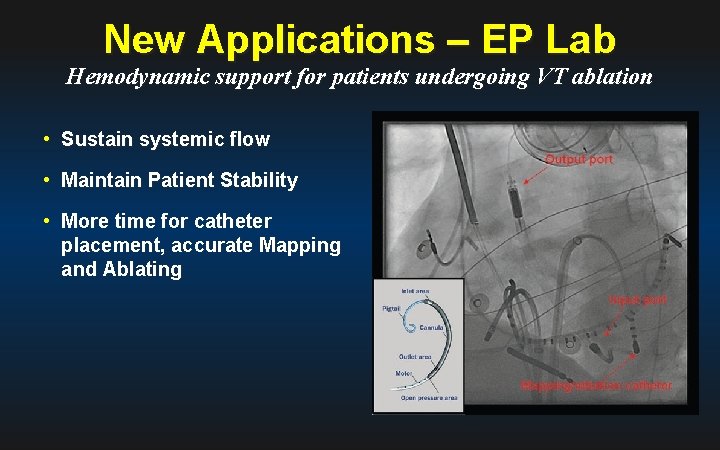
New Applications – EP Lab Hemodynamic support for patients undergoing VT ablation • Sustain systemic flow • Maintain Patient Stability • More time for catheter placement, accurate Mapping and Ablating

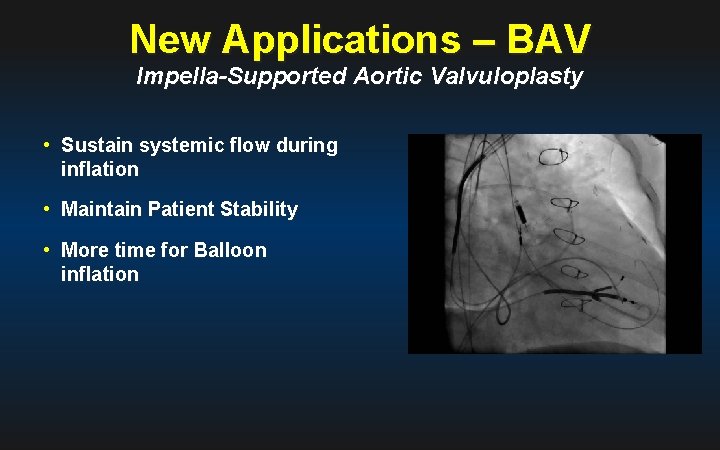
New Applications – BAV Impella-Supported Aortic Valvuloplasty • Sustain systemic flow during inflation • Maintain Patient Stability • More time for Balloon inflation

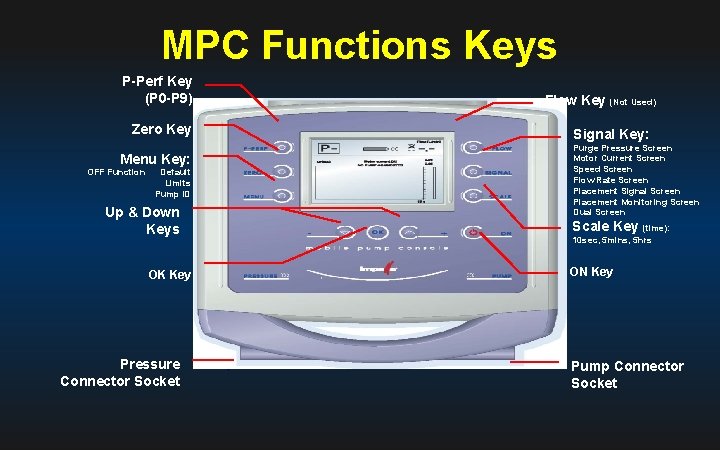
MPC Functions Keys P-Perf Key (P 0 -P 9) Zero Key Menu Key: OFF Function Default Limits Pump ID Up & Down Keys OK Key Pressure Connector Socket Flow Key (Not Used) Signal Key: Purge Pressure Screen Motor Current Screen Speed Screen Flow Rate Screen Placement Signal Screen Placement Monitoring Screen Dual Screen Scale Key (time): 10 sec, 5 mins, 5 hrs ON Key Pump Connector Socket

Impella Product Enhancements Pre-assembled guiding lumen facilitates loading the placement guidewire Integrated infusion pump, user interface and power supply Bright, highly visible user display Graphical user interface Built in Alarm Help Fast and simple to setup Designed for transport

31 Simplified User Interface • Bright easy to read LCD display • Soft button function change depending on current operation • Combined rotating push button for navigation and selections • Continuous display of key Impella® Controller operating parameters

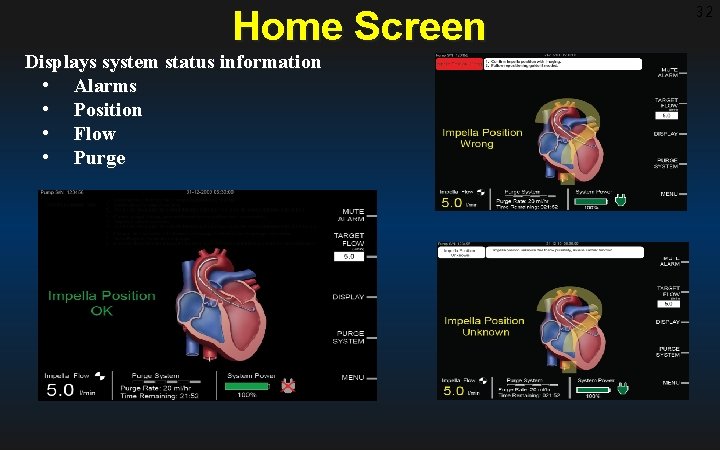
Home Screen Displays system status information • Alarms • Position • Flow • Purge 32

Impella 2. 5 Product Enhancements Simplified Back-loading Pre-assembled guiding lumen facilitates loading the placement guidewire

Impella 2. 5 Product Enhancements Simplified Back-loading Step 1 Thread guidewire Step 2 Remove guiding lumen

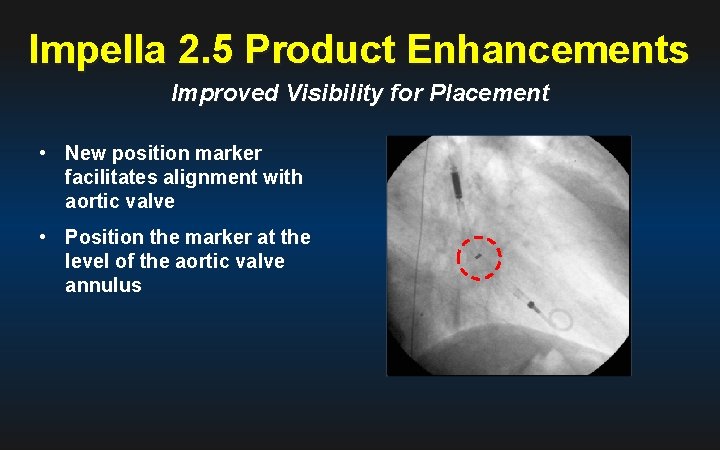
Impella 2. 5 Product Enhancements Improved Visibility for Placement • New position marker facilitates alignment with aortic valve • Position the marker at the level of the aortic valve annulus

Impella 2. 5 Product Enhancements New Repositioning Sheath Design

Wireless Insertion • Eliminates: • Guiding catheter • placement guidewire • back-loading • Reduces the number of steps from 12 to 6 1. The pigtail of the Impella advanced through the sheath 2. Advance the Impella to the aortic valve, until the catheter begins to prolapse 3. Pull the catheter back slightly while turning, the catheter will “pop” across the valve

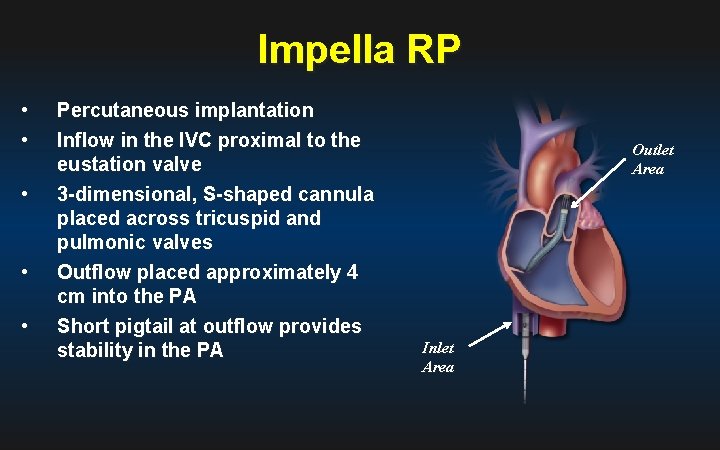
Impella RP • • • Percutaneous implantation Inflow in the IVC proximal to the eustation valve 3 -dimensional, S-shaped cannula placed across tricuspid and pulmonic valves Outflow placed approximately 4 cm into the PA Short pigtail at outflow provides stability in the PA Outlet Area Inlet Area

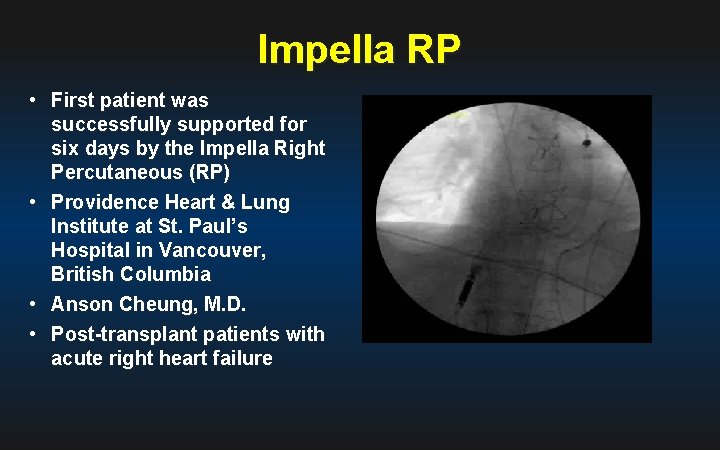
Impella RP • First patient was successfully supported for six days by the Impella Right Percutaneous (RP) • Providence Heart & Lung Institute at St. Paul’s Hospital in Vancouver, British Columbia • Anson Cheung, M. D. • Post-transplant patients with acute right heart failure

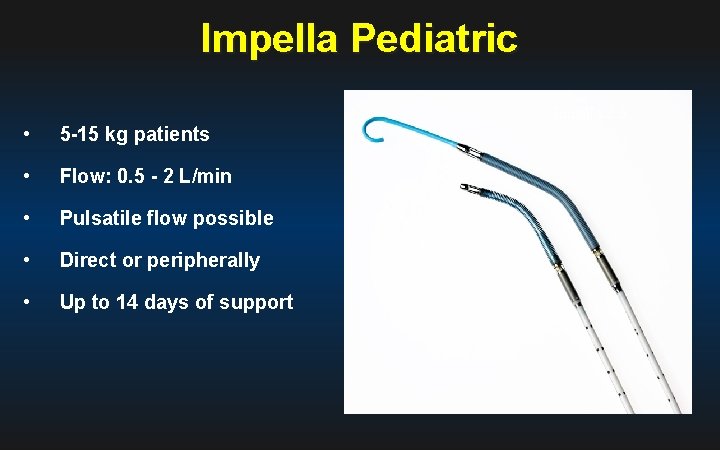
Impella Pediatric Impella 2. 5 • 5 -15 kg patients • Flow: 0. 5 - 2 L/min • Pulsatile flow possible • Direct or peripherally • Up to 14 days of support Impella Pediatric

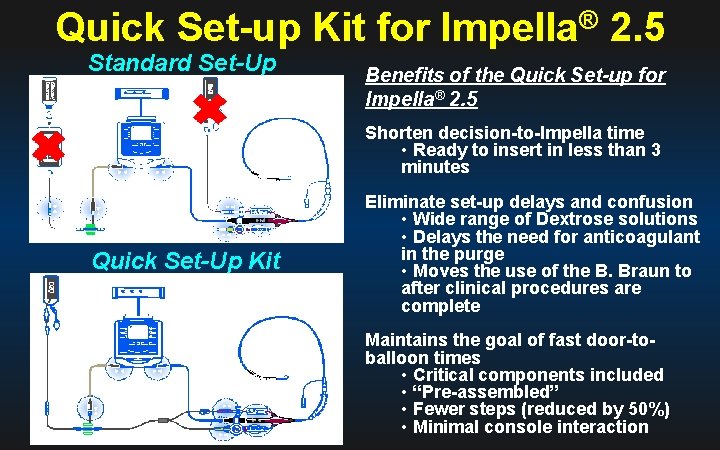
Quick Set-up Kit for Impella® 2. 5 Standard Set-Up Kit Benefits of the Quick Set-up for Impella® 2. 5 Shorten decision-to-Impella time • Ready to insert in less than 3 minutes Quick Set-Up Kit Eliminate set-up delays and confusion • Wide range of Dextrose solutions • Delays the need for anticoagulant in the purge • Moves the use of the B. Braun to after clinical procedures are complete Maintains the goal of fast door-toballoon times • Critical components included • “Pre-assembled” • Fewer steps (reduced by 50%) • Minimal console interaction

Summary 1) Platform for catheter-based cardiac assist pumps designed to directly unload and protect the heart while stabilizing the hemodynamics of the patient 1, 2, 3, 4, 6, 8, 10 2) Designed for Safety and Ease of Use • Minimum Priming time, no sternotomy, no cardiopulmonary bypass 3) Potential to reduce mortality, preserve the cardiac function & improve the patient QOL 1 -6, 12, 13 2, 5, 11, 12, 13 4) Cost-Effective 2, 11, 14, 15 Footnotes/References: 1. Burzotta et al, Journal of Card Vasc Med, 2008 2. Dixon et al, JACC 2009 3. Henriques et al, Journal of American Card, 2006 4. Seyfarth et al, JACC, 2008 5. Sjauw et al, MACH II, JACC 2008 6. Valgimigli et al, Cath Cardiov Interv (2005) 7. Reesink et al, CHEST (2004) 8. Fincke et al, JACC (2004) 9. Sauren et al (2007) 10. Meyns et al, JACC (2003) 11. MACH II 3 -Year Follow up, TCT (2009) 12. USpella, TCT (2009) 13. Europella, Henriques et al, JACC (2009) 14. STEMI Guidelines, ACC/AHA, ICDs 15 AHA Heart Statistics 2009: CABG vs. PCI