CRT 2010 Washington USA Feb 21 23 2010

CRT 2010 Washington, USA, Feb 21 -23, 2010 Cardiac Etiologies for Stroke Horst Sievert, Stefan Bertog, Nina Wunderlich Cardio. Vascular Center Frankfurt, Germany

DISCLOSURES Horst Sievert, MD Consulting Fees – Access. Closure, Inc. , AGA Medical Corporation, Angiomed, Ardian, Inc. , Arstasis, Inc. , Avinger, Bridge. Point Medical, Cardio. Kinetix, Inc. , Cardio. MEMS Inc. , Coherex Medical, Inc. , CSI Medical, Endo. Cross, Boston Scientific Corporation, ev 3, Inc. , Flow. Cardia, Inc. , W. L. Gore & Associates, Inc. , Abbott Vascular, Lumen Biomedical, Kensey Nash Corporation, Kyoto Medical, NDC, NMT Medical, Inc. , OAS, Occlutech, Osprey Medical, Inc. , Ovalis, Inc. , Pathway Medical Technologies, Inc. , Pfm Medical, Inc. , Pendra. Care, Percardia Inc. , Remon Medical Technologies, ROX Medical, Sadra Medical, Sorin Group, Spectranetics, Square One Medical, Viacor, Inc. , St. Jude Medical, Medinol Ltd. , Lutonix, Inc. Grants/Contracted Research – Abbott Vascular, Access. Closure, Inc. , AGA Medical Corporation, Angiomed, Boston Scientific Corporation, Cardio. Kinetix, Inc. , Co. Aptus Medical Corporation, Cordis, a Johnson & Johnson company, CSI Medical, Edwards Lifesciences LLC, ev 3, Inc. , W. L. Gore & Associates, Inc. , Kensey Nash Corporation , Mind Guard, NDC, Neovasc, NMT Medical, Inc. , Percardia Inc. , Sorin Group, St. Jude Medical, Terumo Medical Corporation, Top. Spin Medical Ltd. , St. Jude Medical, Lumen Biomedical Ownership Interest (Stocks, Stock Options or Other Ownership Interest) – Cardio-Kinetics Inc. , Access. Closure, Inc. , Co. Aptus Medical Corporation, Lumen Biomedical, Cierra I intend to reference unlabeled/ unapproved uses of drugs or devices in my presentation. I intend to reference PFO, ASD devices, carotid stents, and EPD.

20 million strokes/year Hemorrhagic Ischemic 10% 90% Intracranial Artery to artery Lacunar Cardioembolic Cryptogenic disease stroke embolism stroke 16% 26% 19% Northern Manhatten Stroke study (NOMAS). Circulation 2005; 111, 1327 -1331 36%
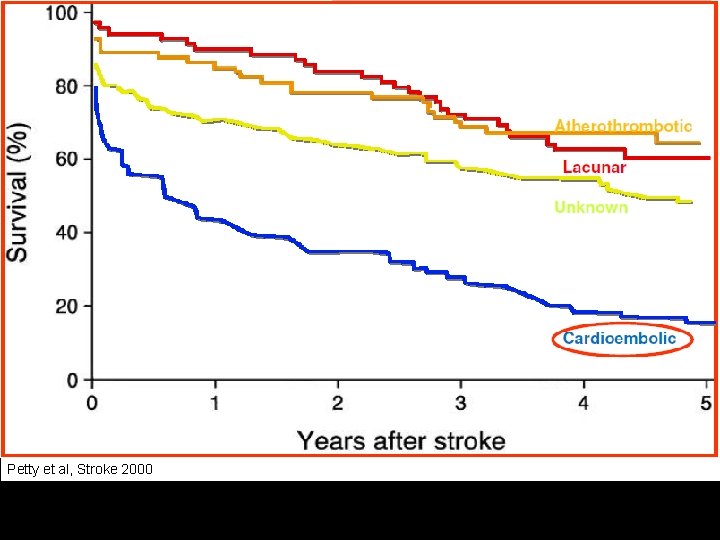
Petty et al, Stroke 2000

Cardioembolic Stroke • • Left atrial appendage (AF, MS) PFO, ASD Left ventricular thrombi (MI, dilated cardiomyopathy) Atrial myxoma Papillary fibroelastoma Vegetations (IE, marantic endocarditis, Libman Sachs) Prosthetic valve thrombi
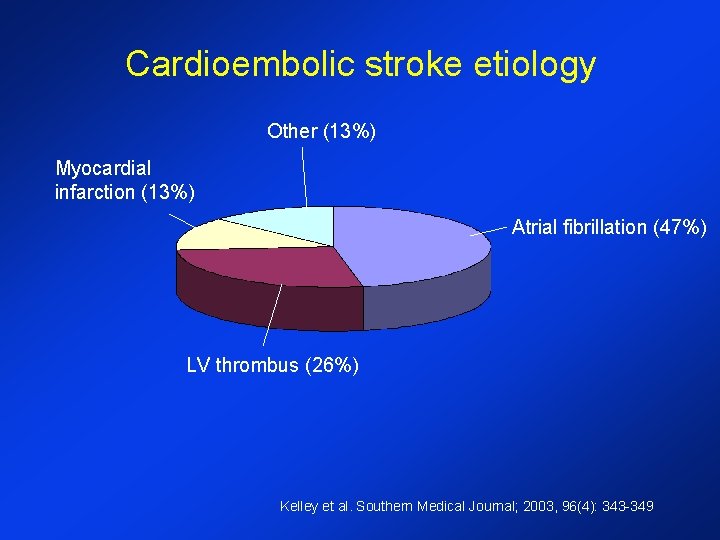
Cardioembolic stroke etiology Other (13%) Myocardial infarction (13%) Atrial fibrillation (47%) LV thrombus (26%) Kelley et al. Southern Medical Journal; 2003, 96(4): 343 -349

Atrial Fibrillation • ~15% of all strokes - Age dependent: at 80 -89 yrs of age, more than 30% of all strokes possibly related to AF* • ~5% overall annual stroke rate (nonrheumatic AF)** • >90% of LA thrombi originate in the LAA *Wolf et al. Arch Intern Med 1987; 147: 1561 -1564 **Lloyd-Jones et al. Circulation 2004; 110: 1042 -1046 †Manning WJ. Clin Cardiol 1995; 18: 547 -554
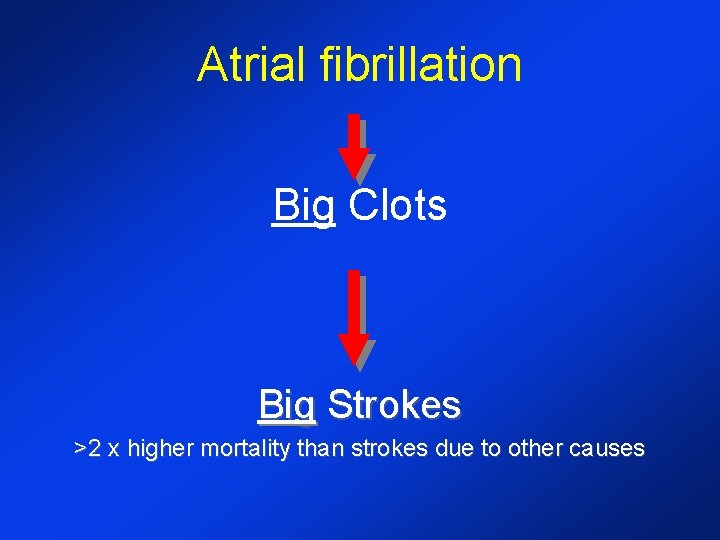
Atrial fibrillation Big Clots Big Strokes >2 x higher mortality than strokes due to other causes
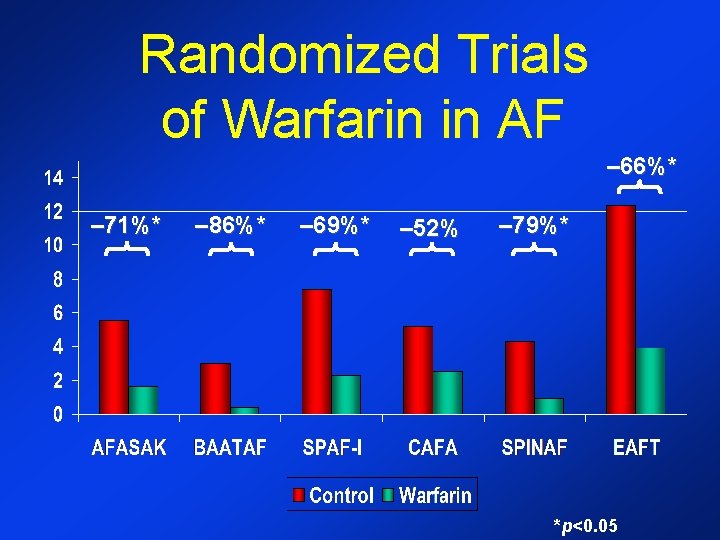
Randomized Trials of Warfarin in AF – 66%* – 71%* – 86%* – 69%* – 52% – 79%* *p<0. 05

However…….

• Warfarin is not perfect…… - Meta-analysis of 13 randomized trials • Annual risk of intracranial hemorrhage ~0. 5% with warfarin • The risk with warfarin is approximately twice as high as the risk with aspirin • Hart et al. Ann Intern Med 2007; 146, 857 -867

• Warfarin is not perfect…… - Major hemorrhage* in the first year • 7% of consecutive patients >65 yrs of age • 13% in patients >80 yrs - In the first year of treatment 26% of patients >80 yrs stopped treatment • 81% of those due to perceived safety issues - The risk of major hemorrhage is greatest among patients with the highest risk of stroke (CHADs≥ 3) who would need anticoagulation most *Death from hemorrhage, hospitalization with ≥ 2 U PRBC, hemorrhage into critical site (e. g. retropreitoneal, intraocular, pericardial, spinal) Hylek et al. Circulation. 2007; 115, 2689 -2696

• Warfarin is not perfect…… - Therapeutic INR range achieved only 50% of time in clinical practice * - Therapeutic INR achieved only 68% of the time in clinical trials * - Lower INR associated with increased stroke risk - Higher INR associated with increased ICH risk *Gottlieb et al. Arch Intern Med. 1994 Sep 12; 154 (17), 1945 -53.

…. . as a result only ~50% of high risk atrial fibrillation patients are treated with anticoagulation Waldo et al. JACC. 2005; 46: 1729 -36.

Potential medical alternatives to warfarin • Thrombin inhibitors - Ximelagatran - Dabigatran • Other Vit K antagonists - Tecarfarin • Factor Xa inhibitors - Rivaroxaban - Apixaban • Dual antiplatelet therapy

Potential medical alternatives to warfarin • Thrombin inhibitors - Ximelagatran - Dabigatran • Other Vit K antagonists - Tecarfarin • Factor Xa inhibitors - Rivaroxaban - Apixaban • Dual antiplatelet therapy

Thrombin inhibitors • Ximelagatran - Non-inferior to warfarin in SPORTIF V trial (n=6405) Lower overall bleeding risk Safety concerns regarding liver toxicity risk • FDA rejection for approval • Dabigatran - Does not require INR monitoring - Dietary and medication interactions are less important - Compared to warfarin equivalent or superior regarding stroke risk reduction bleeding risk - Needs dose adjustment for renal dysfunction - Trend towards higher MI risk with dabigatran JAMA. 2005; 293: 690 -698 NEJM. 2009 Sep 17; 361 (12), 1139 -1151

Potential medical alternatives to warfarin • Thrombin inhibitors - Ximelagatran - Dabigatran • Other Vit K antagonists - Tecarfarin • Factor Xa inhibitors - Rivaroxaban - Apixaban • Dual antiplatelet therapy

Other Vit K antagonists • Tecarfarin - drug- and food interactions are less important - Small, open label safety and tolerability study (n=66) - INR more likely to be in therapeutic range - Promising but more studies needed Ellis et al. Circulation. Sep 22; 120(12), 1029 -1035

Potential medical alternatives to warfarin • Thrombin inhibitors - Ximelagatran - Dabigatran • Other Vit K antagonists - Tecarfarin • Factor Xa inhibitors - Rivaroxaban - Apixaban • Dual antiplatelet therapy

Factor Xa inhibitors • Rivaroxaban - Shown to be more effective than enoxaparin in the prevention of venous thromboembolism after TKA and total hip replacement (RECORD trial) - Ongoing randomized (vs. warfarin) ROCKET-AF trial (~11 000 pts, estimated study completion date: 2010) • Apixaban - Ongoing randomized (vs. warfarin) ARISTOTLE trial (~18 000 pts, estimated completion date: 4/2011)

Potential medical alternatives to warfarin • Thrombin inhibitors - Ximelagatran - Dabigatran • Other Vit K antagonists - Tecarfarin • Factor Xa inhibitors - Rivaroxaban - Apixaban • Dual antiplatelet therapy

Dual antiplatelet therapy • ACTIVE W* (~6 K patients) randomized controlled trial ASA+Clopidogrel vs warfarin - Terminated prematurely due to a higher risk of strokes in the double antiplatelet group • ACTIVE A** (~7 K patients) unsuitable for warfarin randomized to clopidogrel in addition to ASA versus placebo - Reduction in stroke (2. 4% versus 3. 3%) at the expense of an increased bleeding risk (2. 0% vs. 1. 3%) - Exclusion of patients at high bleeding risk - => patients not a candidate for warfarin due to bleeding risk are also not a candidate for dual antiplatelet therapy *Lancet. 2006 June 10; 367(9526), 1903 -12 **NEJM. 2009 May 14; 360(20), 2066 -2078
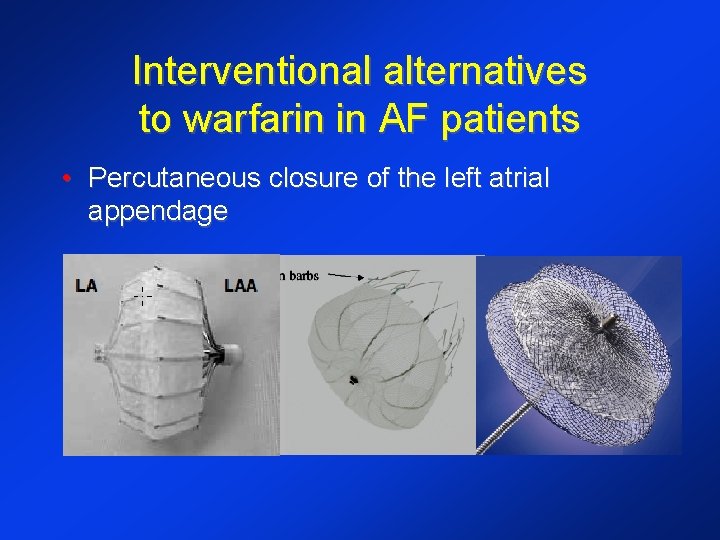
Interventional alternatives to warfarin in AF patients • Percutaneous closure of the left atrial appendage

PROTECT-AF Clinical Trial • Prospective, randomized study of WATCHMAN LAA Device vs Long-Term Warfarin Therapy • 2: 1 allocation ratio device to control • Non-inferiority study -Bayesian sequential design -Analysis at 600 pt-yr and every 150 pt-yr after D. Holmes CRT 2010

PROTECT-AF Trial Endpoints • Primary efficacy endpoint -All stroke: ischemic or hemorrhagic -Cardiovascular or unexplained death -Systemic embolism • Primary safety endpoint D. Holmes CRT 2010
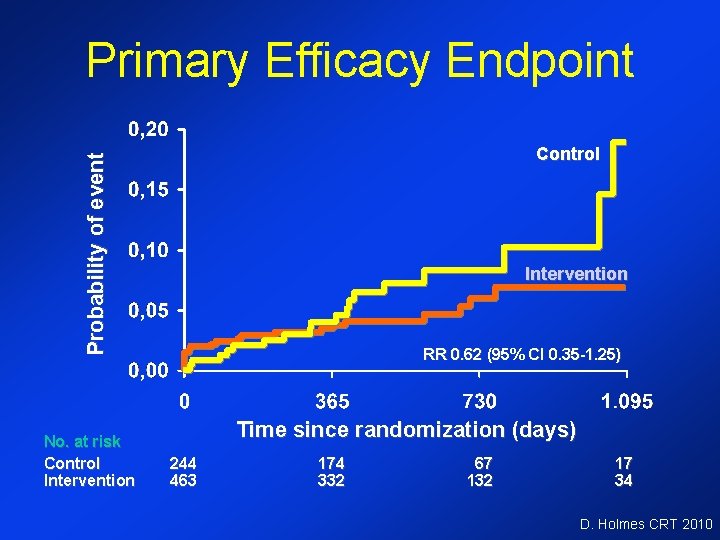
Primary Efficacy Endpoint Probability of event Control No. at risk Control Intervention RR 0. 62 (95% CI 0. 35 -1. 25) Time since randomization (days) 244 463 174 332 67 132 17 34 D. Holmes CRT 2010
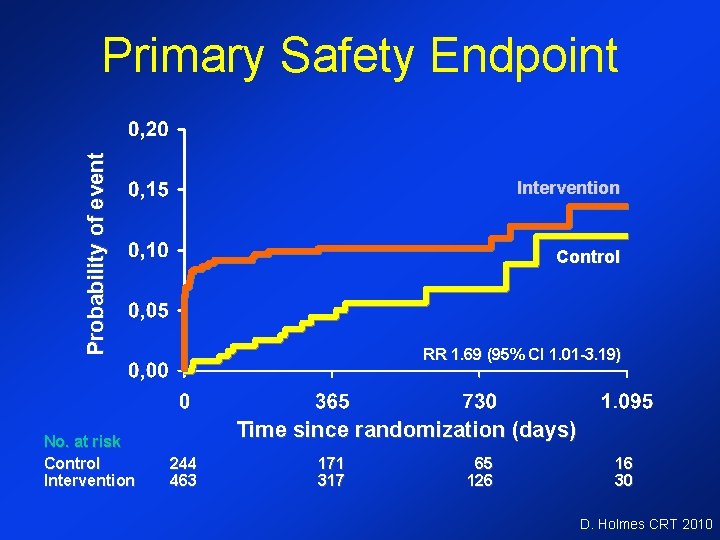
Probability of event Primary Safety Endpoint No. at risk Control Intervention Control RR 1. 69 (95% CI 1. 01 -3. 19) Time since randomization (days) 244 463 171 317 65 126 16 30 D. Holmes CRT 2010
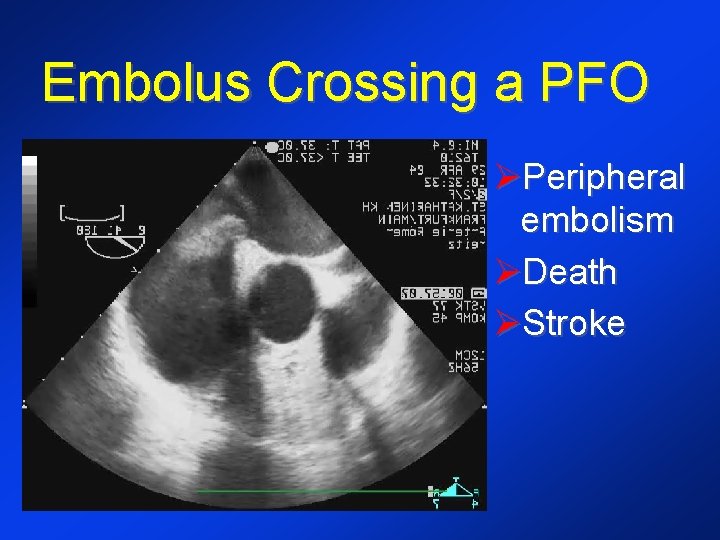
Embolus Crossing a PFO ØPeripheral embolism ØDeath ØStroke

Stroke due PFO: Medical Therapy • recommended by all scientific societies and guidelines • never been tested in randomized trials • 0. 5 – 5 % severe bleeding per year • not very effective - 1 -7 % annual stroke rate despite medical treatment

Surgical PFO Closure • Probably effective • Only very small series have been published • Never been tested in a randomized trial • From surgical ASD closure in adults we can expect a - mortality of 0. 5% - perioperative stroke rate of 1 -2%
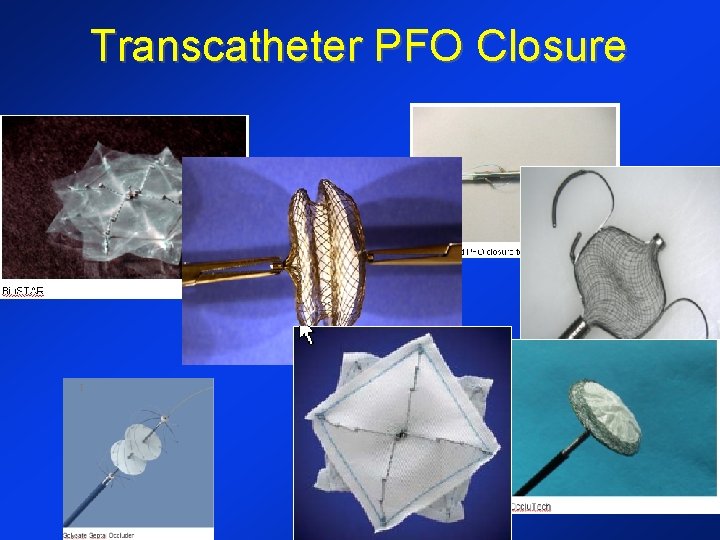
Transcatheter PFO Closure
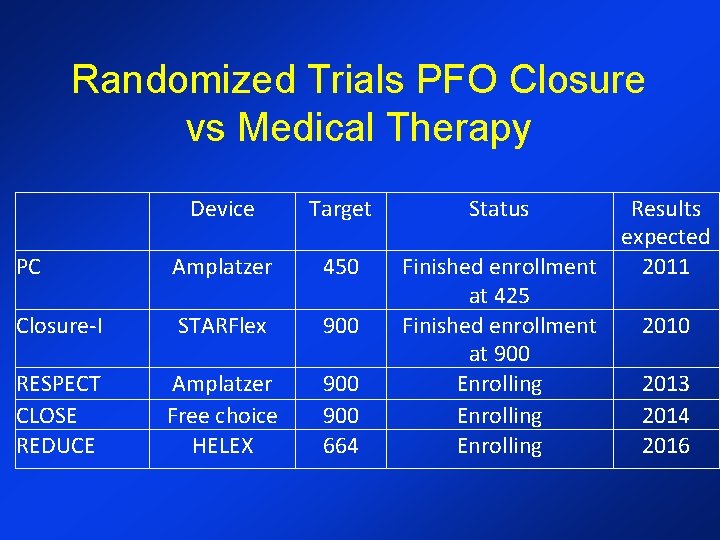
Randomized Trials PFO Closure vs Medical Therapy Device Target PC Amplatzer 450 Closure-I STARFlex 900 RESPECT CLOSE REDUCE Amplatzer Free choice HELEX 900 664 Status Results expected Finished enrollment 2011 at 425 Finished enrollment 2010 at 900 Enrolling 2013 Enrolling 2014 Enrolling 2016

Stroke after Acute MI • Almost no prospective data • Stroke rate post MI - at 30 days 1. 2% - at one year 2. 1% • The most likely source for stroke after myocardial infarction is a left ventricular thrombus Witt et al. Am J Med 2006 April; 119(4): 354 e 1 -9
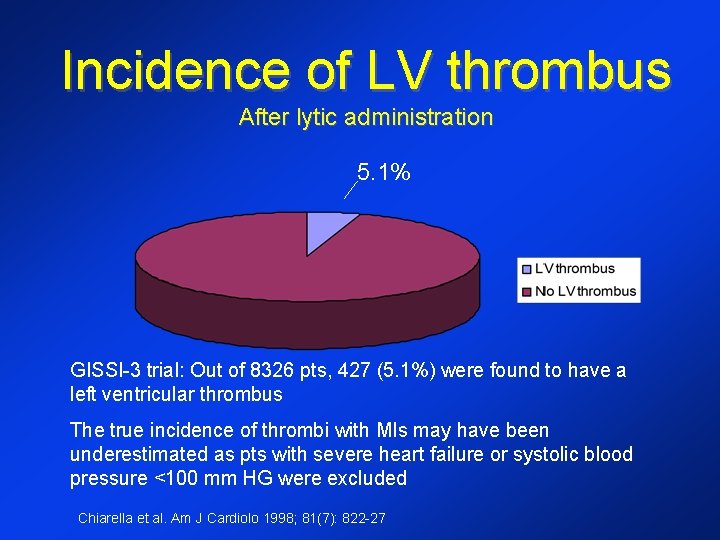
Incidence of LV thrombus After lytic administration 5. 1% GISSI-3 trial: Out of 8326 pts, 427 (5. 1%) were found to have a left ventricular thrombus The true incidence of thrombi with MIs may have been underestimated as pts with severe heart failure or systolic blood pressure <100 mm HG were excluded Chiarella et al. Am J Cardiolo 1998; 81(7): 822 -27

LV thrombus after acute MI is probably much more frequent - Surgical or pathological examination for LV thrombus • Retrospective review of imaging (echo, TEE and MRI including late Gadolinium enhancement) - LV thrombus present in 29% - Sensitivity and specificity with MRI was higher than with echo (88% and 99% for MRI vs. 23% and 96% with TTE and 40% and 96% with TEE) Srichai et al. Am Heart J 2006; 152(1): 75 -84

LV thrombus • Echo risk factors for embolization: - Mobile thrombi - Protruding thrombi Ø Risk for embolisation 7 -10 times Visser et al. JACC 1985; 5(6): 1276 -80

LV thrombus - Most embolic events occur - in patients not receiving warfarin and - within the first 4 months • 43 pts with LV thrombi post acute MI were followed serially for 15 months - None of the 25 pts on anticoagulation had an embolic event - Embolization occurred in 7 of the 18 pts who had not received anticoagulation - all events occurred within four months Weinreich et al. Ann Intern Med 1984; 100(6): 789 -94

Prevention of LV Thrombus • Several small non-randomized trials have shown a benefit of warfarin • 2004 ACC/AHA*: Warfarin reasonable in pts with severe LV dysfunction and extensive regional wall motion abnormality *Antman et al. 2006 ACC/AHA guidelines for the management of patiens with ST-segment elevation myocardial infarction. Available on line at www. americanheart. org

In patients with LV thrombus • 2004 AHA/ACC*: Warfarin for at least 3 -6 months and indefinitely in pts with low bleeding risk • 2006 AHA/ASA**: Warfarin for at least 3 months and up to one year • Concurrent aspirin *Antman et al. 2006 ACC/AHA guidelines for the management of patiens with ST-segment elevation myocardial infarction. Available on line at www. americanheart. org **Sacco et al. ACC/ASA Guidelines for the prevention of stroke. Stroke 2006; 37: 577

Other rare causes of cardiac emboli • • • Atrial myxoma Papillary fibroelastoma Infectious endocarditis Prosthetic valve thrombus Libman-Sachs Prosthetic valve thrombus

Interventional techniques to prevent non-LAA cardiac thrombo-embolic events?
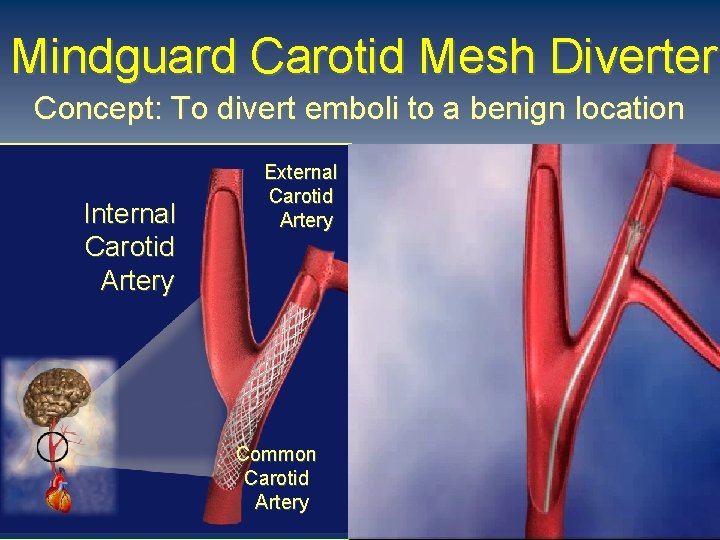
Mindguard Carotid Mesh Diverter Concept: To divert emboli to a benign location Internal Carotid Artery External Carotid Artery Common Carotid Artery

CLARET Aorta Embolic Protection Device • Right radial access • Prox filter for the innominate artery • distal filter for the left carotid • 140 µ filter Reinald Low
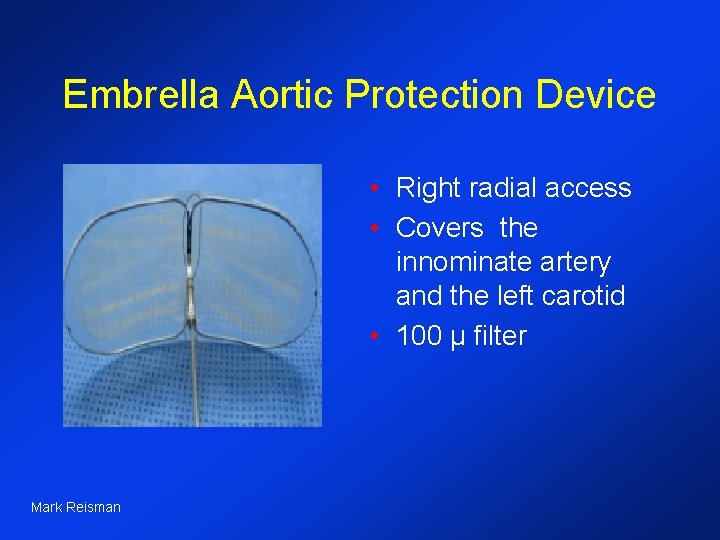
Embrella Aortic Protection Device • Right radial access • Covers the innominate artery and the left carotid • 100 µ filter Mark Reisman
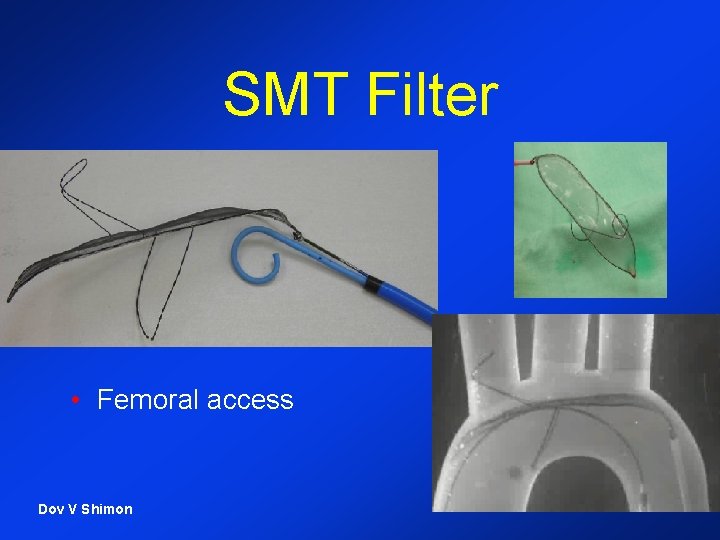
SMT Filter • Femoral access Dov V Shimon

July 9 -11, 2009


Thank You!
- Slides: 49