CRT 2008 Wednesday February 13 th 2008 A

CRT 2008 Wednesday February 13 th, 2008 A Randomized Study Assessing the Impact of Cilostazol on Platelet Function Profiles in Patients with Diabetes Mellitus and Coronary Artery Disease on Dual Antiplatelet Therapy: Results of the OPTIMUS -2 (Optimizing anti-Platelet Therapy In diabetes Mellit. US) Study Dominick J. Angiolillo, MD, Ph. D Director of Cardiovascular Research Assistant Professor of Medicine University of Florida College of Medicine – Jacksonville, Fl

Presenter Disclosure Information Name: Dominick J Angiolillo Within the past 12 months, the presenter or their spouse/partner have had a financial interest/arrangement or affiliation with the organization listed below. Company Name: Relationship: Bristol Myers Squibb Consultant/Speaker bureau Sanofi-Aventis Consultant/Speaker bureau Eli Lilly Consultant/Speaker bureau Daiichi Sankyo Consultant/Speaker bureau Portola Consultant GSK Educational Grant OPTIMUS 2 was an investigator-initiated study. Dr Angiolillo was recipient of the 2006 GSK International Competitive Educational Grant to perform the study. Otsuka pharmaceuticals provided study medication.
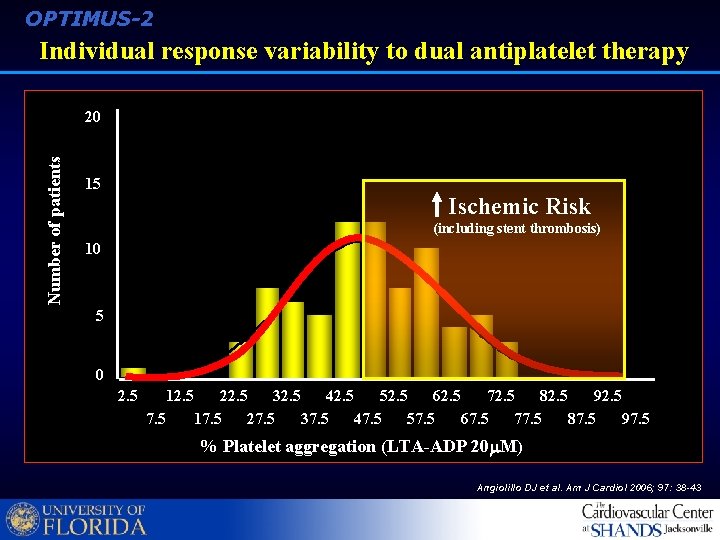
OPTIMUS-2 Individual response variability to dual antiplatelet therapy Number of patients 20 15 Ischemic Risk (including stent thrombosis) 10 5 0 2. 5 12. 5 22. 5 32. 5 42. 5 52. 5 62. 5 72. 5 82. 5 92. 5 7. 5 17. 5 27. 5 37. 5 47. 5 57. 5 67. 5 77. 5 87. 5 97. 5 % Platelet aggregation (LTA-ADP 20 M) Angiolillo DJ et al. Am J Cardiol 2006; 97: 38 -43
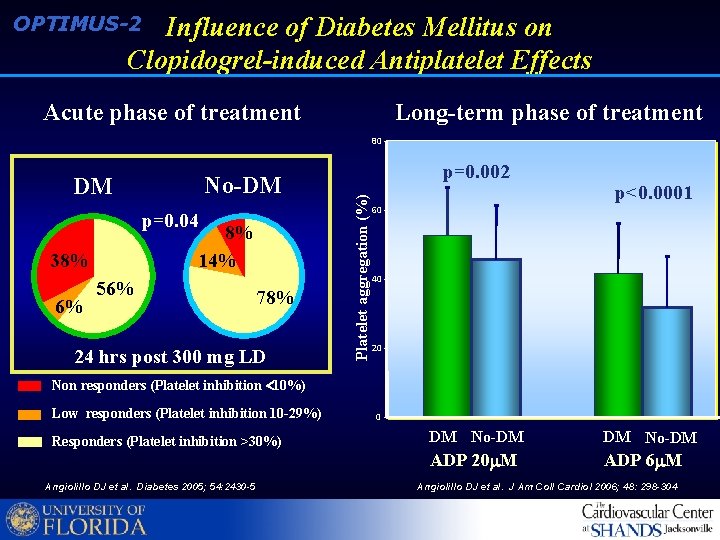
Influence of Diabetes Mellitus on Clopidogrel-induced Antiplatelet Effects OPTIMUS-2 Acute phase of treatment Long-term phase of treatment 80 p=0. 04 8% 14% 38% 6% 56% 78% 24 hrs post 300 mg LD Platelet aggregation (%) No-DM DM p=0. 002 p<0. 0001 60 40 20 Non responders (Platelet inhibition 10%) Low responders (Platelet inhibition 10 -29%) Responders (Platelet inhibition >30%) Angiolillo DJ et al. Diabetes 2005; 54: 2430 -5 0 DM No-DM ADP 20 M ADP 6 M Angiolillo DJ et al. J Am Coll Cardiol 2006; 48: 298 -304
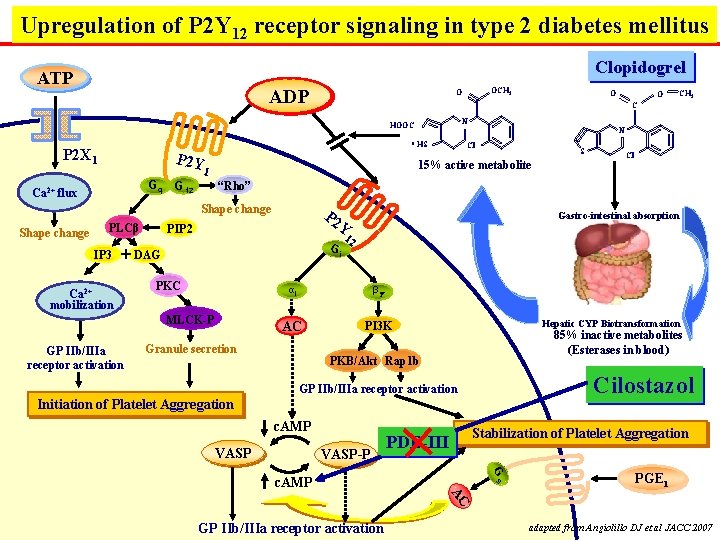
Upregulation of P 2 Y 12 receptor signaling in type 2 diabetes mellitus Clopidogrel ATP ADP P 2 Y G 12 PLCβ IP 3 Cl Gastro-intestinal absorption Y PIP 2 Gi PKC MLCK-P GP IIb/IIIa receptor activation S P 2 + DAG Ca 2+ mobilization Cl “Rho” Shape change CH 3 N 15% active metabolite 1 Gq O N * HS Ca 2+ flux O C HOOC P 2 X 1 OCH 3 O 12 αi βγ AC PI 3 K Granule secretion Hepatic CYP Biotransformation 85% inactive metabolites (Esterases in blood) PKB/Akt Rap 1 b Cilostazol GP IIb/IIIa receptor activation Initiation of Platelet Aggregation c. AMP VASP-P PDE-III Stabilization of Platelet Aggregation Gs c. AMP PGE 1 AC GP IIb/IIIa receptor activation adapted from Angiolillo DJ et al JACC 2007

OPTIMUS-2 Objectives To evaluate platelet function profiles obtained with the adjunct of cilostazol in patients with type 2 diabetes mellitus and coronary artery disease while on standard dual (aspirin and clopidogrel) antiplatelet therapy.
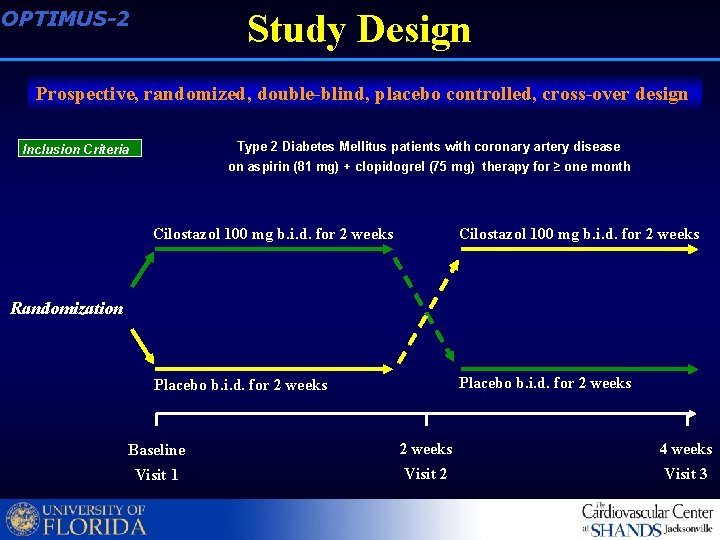
OPTIMUS-2 Study Design Prospective, randomized, double-blind, placebo controlled, cross-over design Type 2 Diabetes Mellitus patients with coronary artery disease Inclusion Criteria on aspirin (81 mg) + clopidogrel (75 mg) therapy for ≥ one month Cilostazol 100 mg b. i. d. for 2 weeks Placebo b. i. d. for 2 weeks Randomization Baseline 2 weeks 4 weeks Visit 1 Visit 2 Visit 3

OPTIMUS-2 Primary: Endpoints P 2 Y 12 reactivity index (PRI) PGE 1 Secondary: ADP - VASP-P - Verify. Now P 2 Y 12 (%inhibition and PRU) - Light Transmittance Aggregometry (LTA) Aggmax and Agglate following 5 and 20 mol/L ADP stimuli w/wo 5 n. M PGE 1 (post. VASP treatment platelet reactivity, IPA, platelet disaggregation) VASP-P Activated Inhibited platelets or resting platelets Power Calculation: Cilostazol will lead to a 15 ± 16% decrease in PRI; a minimum of 17 patients required to achieve a power of 95% ( two-sided = 0. 05)
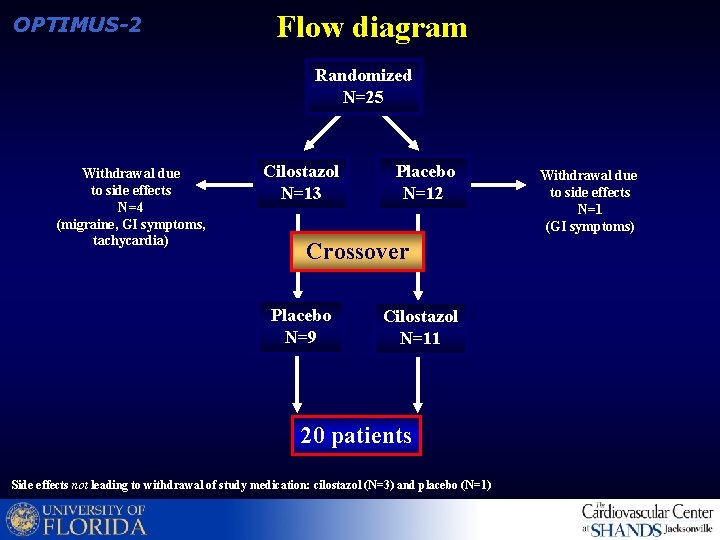
OPTIMUS-2 Flow diagram Randomized N=25 Withdrawal due to side effects N=4 (migraine, GI symptoms, tachycardia) Cilostazol N=13 Placebo N=12 Crossover Placebo N=9 Cilostazol N=11 20 patients Side effects not leading to withdrawal of study medication: cilostazol (N=3) and placebo (N=1) Withdrawal due to side effects N=1 (GI symptoms)

Demographics OPTIMUS-2 Age (yrs) Gender (male), n (%) Race, n (% 64 10 15 (60) Caucasian African-American Hispanic Risk factors/past medical history, n (%) 19 (76) 5 (20) 1 (4) Insulin-dependent diabetes Non-insulin-dependent diabetes Hb. A 1 C Smoking Hyperlipidemia Hypertension Body mass index Prior myocardial infarction Prior CABG Multivessel CAD Treatment, n (%) 9 (36) 16 (64) 7. 7 1. 8 6 (24) 23 (92) 24 (96) 31. 5 16 (64) 7 (28) 19 (76) Beta-blockers Nitrates ACE-Inhibitors/ARB 19 (76) 5 (20) 21 (84) CYP 3 A 4 metabolizing statin Non-CYP 3 A 4 metabolizing statin 17 (68) 5 (20)
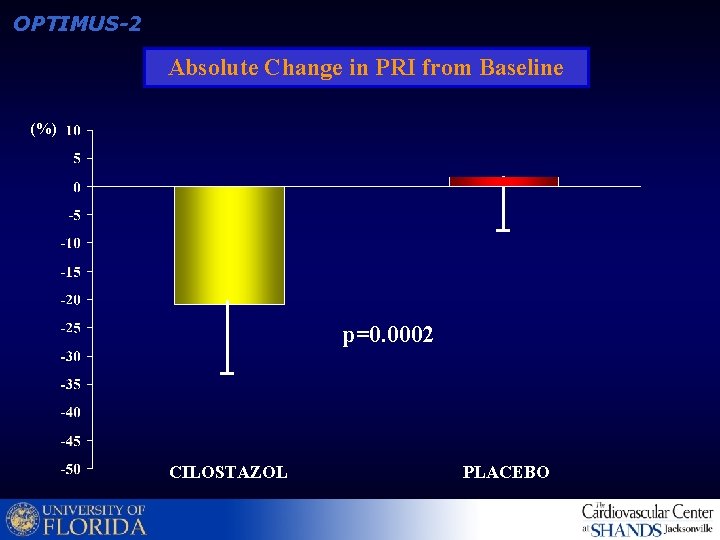
OPTIMUS-2 Absolute Change in PRI from Baseline (%) p=0. 0002 CILOSTAZOL PLACEBO
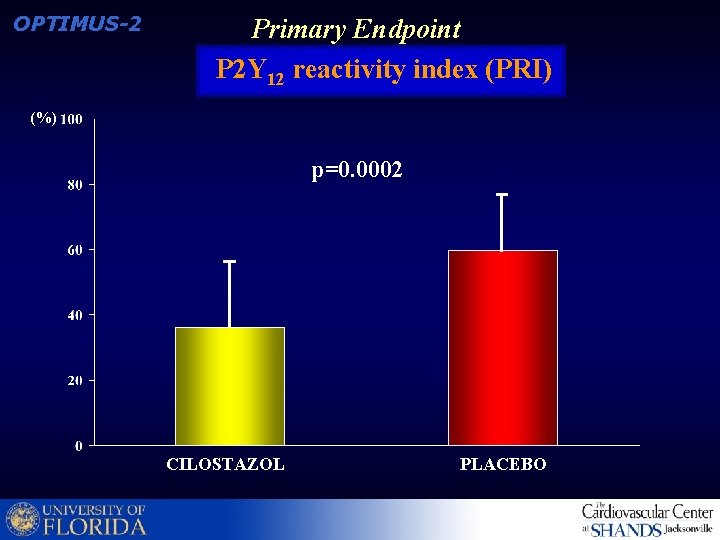
OPTIMUS-2 Primary Endpoint P 2 Y 12 reactivity index (PRI) (%) p=0. 0002 CILOSTAZOL PLACEBO
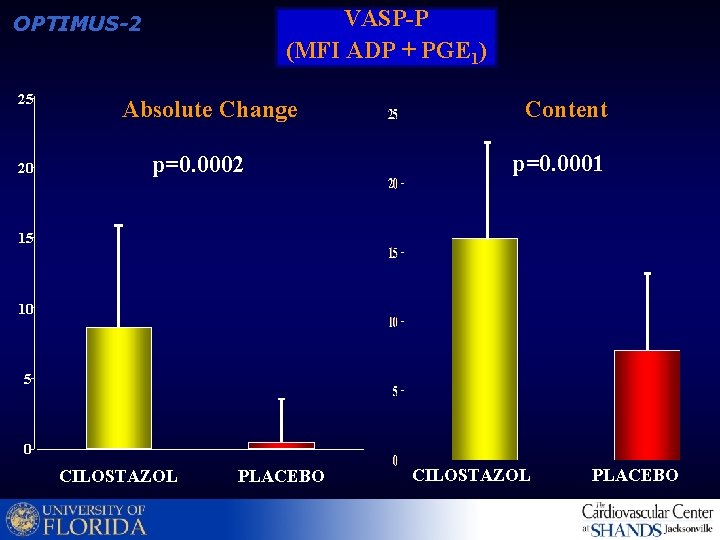
VASP-P (MFI ADP + PGE 1) OPTIMUS-2 25 20 Absolute Change p=0. 0002 Content p=0. 0001 15 10 5 0 CILOSTAZOL PLACEBO
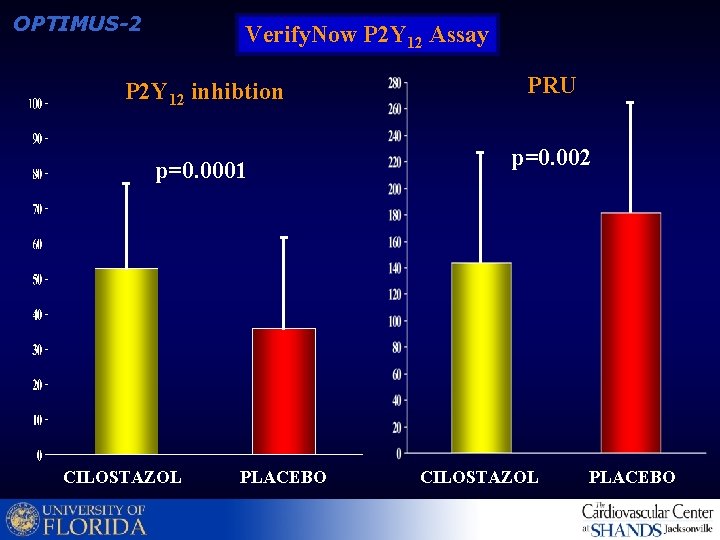
OPTIMUS-2 Verify. Now P 2 Y 12 Assay P 2 Y 12 inhibtion p=0. 0001 CILOSTAZOL PLACEBO PRU p=0. 002 CILOSTAZOL PLACEBO
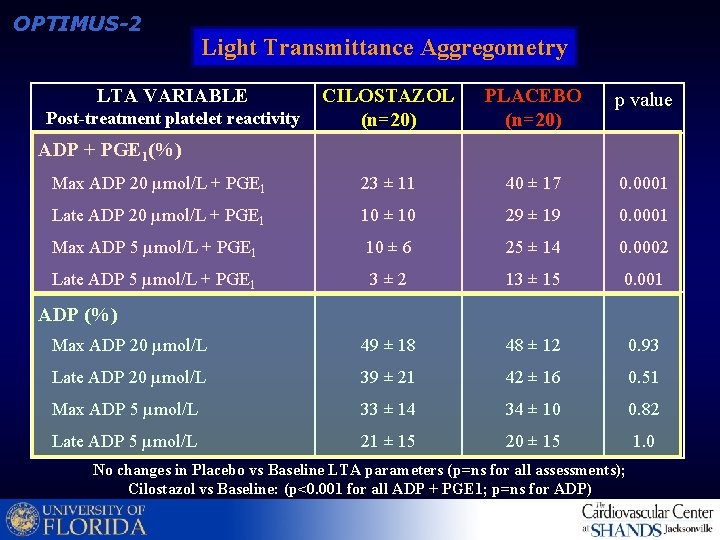
OPTIMUS-2 Light Transmittance Aggregometry LTA VARIABLE CILOSTAZOL (n=20) PLACEBO (n=20) p value Max ADP 20 µmol/L + PGE 1 23 ± 11 40 ± 17 0. 0001 Late ADP 20 µmol/L + PGE 1 10 ± 10 29 ± 19 0. 0001 Max ADP 5 µmol/L + PGE 1 10 ± 6 25 ± 14 0. 0002 Late ADP 5 µmol/L + PGE 1 3± 2 13 ± 15 0. 001 Max ADP 20 µmol/L 49 ± 18 48 ± 12 0. 93 Late ADP 20 µmol/L 39 ± 21 42 ± 16 0. 51 Max ADP 5 µmol/L 33 ± 14 34 ± 10 0. 82 Late ADP 5 µmol/L 21 ± 15 20 ± 15 1. 0 Post-treatment platelet reactivity ADP + PGE 1(%) ADP (%) No changes in Placebo vs Baseline LTA parameters (p=ns for all assessments); Cilostazol vs Baseline: (p<0. 001 for all ADP + PGE 1; p=ns for ADP)
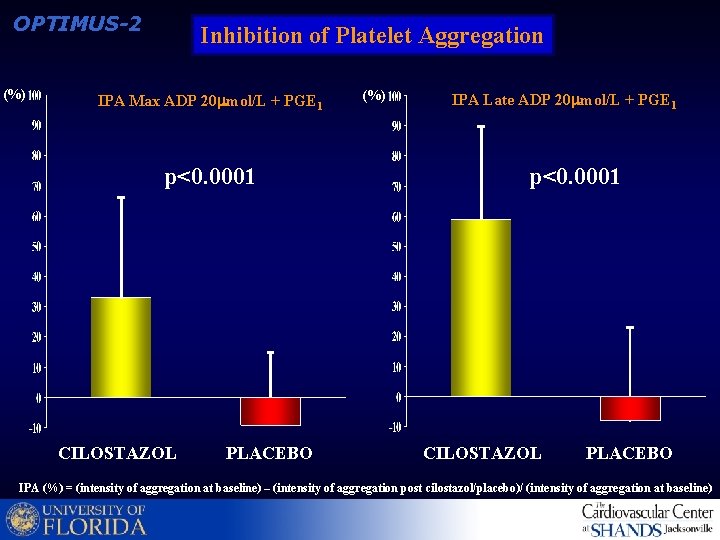
OPTIMUS-2 (%) Inhibition of Platelet Aggregation IPA Max ADP 20 mol/L + PGE 1 p<0. 0001 CILOSTAZOL PLACEBO (%) IPA Late ADP 20 mol/L + PGE 1 p<0. 0001 CILOSTAZOL PLACEBO IPA (%) = (intensity of aggregation at baseline) – (intensity of aggregation post cilostazol/placebo)/ (intensity of aggregation at baseline)
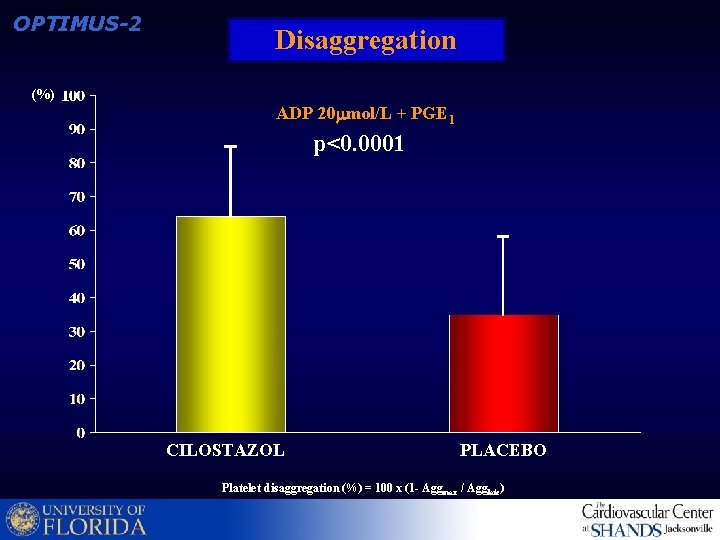
OPTIMUS-2 Disaggregation (%) ADP 20 mol/L + PGE 1 p<0. 0001 CILOSTAZOL PLACEBO Platelet disaggregation (%) = 100 x (1 - Aggmax / Agglate)

OPTIMUS-2 Conclusions • In patients with T 2 DM on chronic standard dual antiplatelet therapy, treatment with cilostazol is associated with enhanced suppression of P 2 Y 12 receptor signaling. • Enhanced P 2 Y 12 inhibition achieved with cilostazol in adjunct to standard antiplatelet treatment regimens may explain why lower ischemic event rates, including stent thombosis, are achieved with triple oral antiplatelet therapy compared to dual therapy. • However, side effects are common with cilostazol therapy which frequently lead to drug withdrawal. • Need for further improvement in antiplatelet strategies…. . awaiting more outcome data with novel and potent drugs currently under advanced clinical investigation.
- Slides: 18