Creating a foldable for the electrons in atoms

Creating a foldable for the electrons in atoms notes � Fold paper into 3 sections
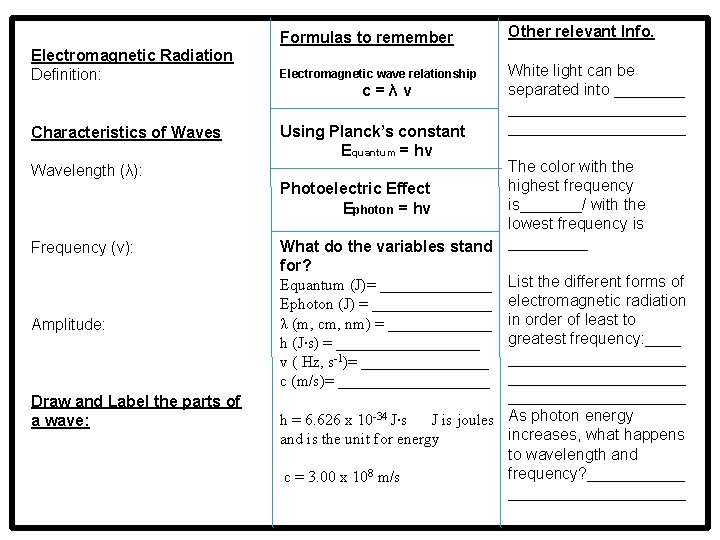
Electromagnetic Radiation Definition: Characteristics of Waves Wavelength (λ): Frequency (v): Amplitude: Draw and Label the parts of a wave: Formulas to remember Other relevant Info. Electromagnetic wave relationship White light can be separated into ______________ c=λv Using Planck’s constant Equantum = hv The color with the highest frequency Photoelectric Effect is_______/ with the Ephoton = hv lowest frequency is What do the variables stand _____ for? Equantum (J)= _______ List the different forms of Ephoton (J) = ________ electromagnetic radiation λ (m, cm, nm) = _______ in order of least to greatest frequency: ____ h (J∙s) = _________ v ( Hz, s-1)= ____________________ c (m/s)= ____________________ h = 6. 626 x 10 -34 J∙s J is joules As photon energy increases, what happens and is the unit for energy to wavelength and frequency? ______ c = 3. 00 x 108 m/s __________

On the back of your foldable… Make sure to show your work!! Practice Problems Name: _____ Date: _____ Period: _______ What is the wavelength of a microwave with a frequency of 5. 22 x 108 Hz? Calculate the frequency of photon that has an energy of 5. 313 x 10 -16 J Formula: Work: Ans with correct units and proper sig figs. Formula: Work: Ans with correct units and proper sig figs. What is the energy of a quantum with a frequency of 1. 05 x 1016 s-1 Formula: Work: Ans with correct units and proper sig figs.

Electrons in Atoms Chapter 5 in your text pp. 117 -141

When did studying light become important? � In the early 1900’s scientist discovered that certain elements gave off visible light when heated in a flame. Analysis of this light revealed that an element’s chemical behavior is related to the arrangement of electrons. � Because of this, it became important to understand nature of light

Wave Nature of Light � Electromagnetic radiation: a form of energy that exhibits wavelike behavior as it travels through space. Examples Visible light from the sun, microwaves, x-rays, radio waves, etc
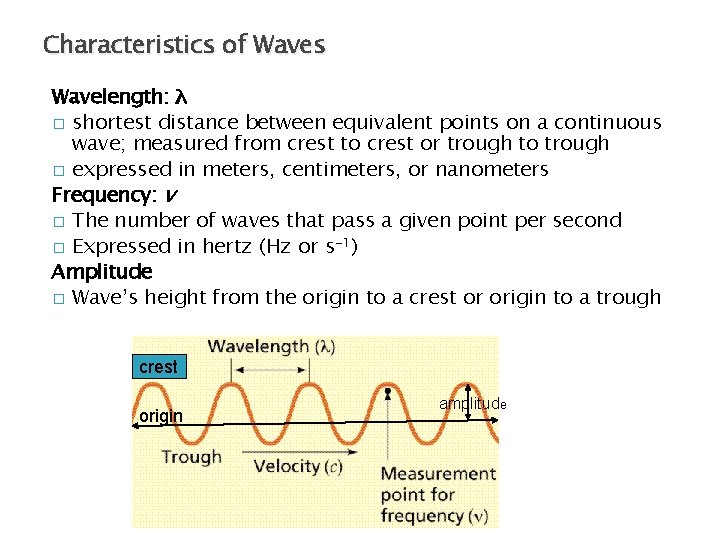
Characteristics of Waves Wavelength: λ � shortest distance between equivalent points on a continuous wave; measured from crest to crest or trough to trough � expressed in meters, centimeters, or nanometers Frequency: v � The number of waves that pass a given point per second � Expressed in hertz (Hz or s-1) Amplitude � Wave’s height from the origin to a crest or origin to a trough crest origin amplitude

Electromagnetic Wave Relationship All electromagnetic waves travel at the speed of light (C) 3. 00 x 108 m/s c=λv where c is the speed of light, λ is wavelength, and v is frequency Example: What is the wavelength of a microwave with a frequency of 3. 44 x 109 Hz? λ = c/v λ = 3. 00 x 108 m/s = 8. 72 x 10 -2 m 3. 44 x 10 s-1

Electromagnetic Spectrum Although the speed of Electromagnetic waves is the same, wavelengths and frequencies can be different (ex: higher frequency = shorter wavelength) � � White light, such as sunlight, can be separated into a continuous spectrum of colors if passed through a prism. These are the colors of the rainbow (roy g biv – red, orange, yellow, green, blue, indigo, violet)
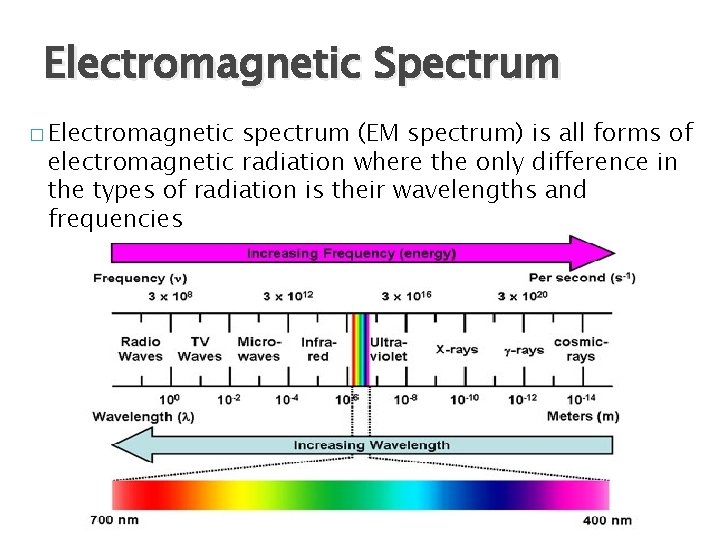
Electromagnetic Spectrum � Electromagnetic spectrum (EM spectrum) is all forms of electromagnetic radiation where the only difference in the types of radiation is their wavelengths and frequencies

Particle Nature of Light � The wave model couldn’t explain the emission of the different wavelengths. In 1900, Max Planck studied the light given off by heated objects. His studies led him to the conclusion that matter can gain or lose energy in small amounts called “quanta. ” � A Quantum is the minimum amount of energy that can be gained/lost by an atom

Planck’s Constant Equantum = hv Equantum is energy, h is Planck’s constant, and v is frequency The value for Planck’s constant is 6. 626 x 10 -34 J∙s J is joules and is the unit for energy Example: What is the energy of a photon from the violet portion of the Sun’s light if it has a frequency of 7. 230 x 1014 s-1 ? Ephoton = (6. 626 x 10 -34 J∙s) (7. 230 x 1014 s-1 ) = 4. 791 x 10 -18 J

Photoelectric effect � Photoelectrons are given off a metal’s surface when light of a certain frequency shines on the surface � Einstein proposed light has a dual nature- A beam of light has wavelike and particlelike properties. A beam of bundles of energy were called photons (massless particle that carries a quantum of energy. � Ephoton = hv As the energy of a photon increases, the frequency increases. What can you conclude about the wavelength.
- Slides: 13