Creatine Kinase Amy Ward Overview Metabolism Creatine Kinase

Creatine Kinase Amy Ward

Overview § § Metabolism Creatine Kinase Isoforms ATP Recycling Clinical Relevance

Metabolism § ATP is the energy currency in the cell § Cellular respiration occurs in the mitochondria § Muscle and brain are most actively metabolizing tissues

ATP as Energy Source § ATP donates high energy bond in coupled reactions § Substrate ATP Product ADP

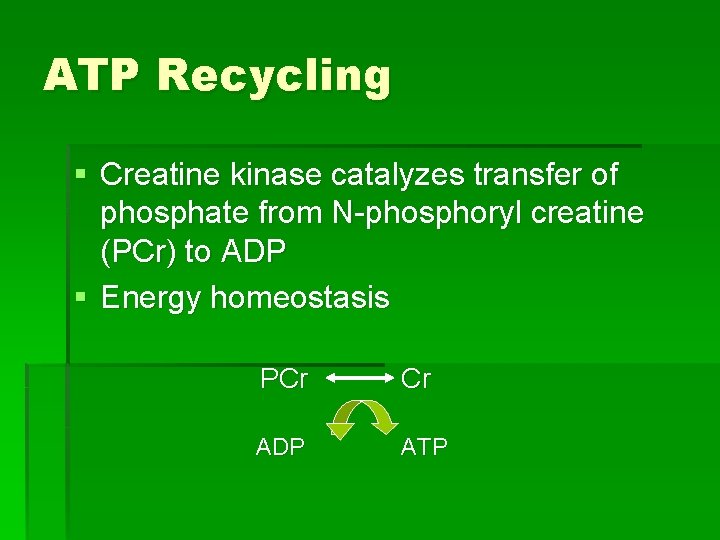
ATP Recycling § Creatine kinase catalyzes transfer of phosphate from N-phosphoryl creatine (PCr) to ADP § Energy homeostasis PCr Cr ADP ATP

Creatine Kinase § Crystallization attempts date back to 1950 s § First successful crystal formed in 1996

Creatine Kinase § Different isoforms depending on location § Coupled to sites of energy production or consumption

CK Isoforms § Cytosolic Isoforms § Muscle-type § Brain-type § Exist as dimers § Temporal energy buffering § Mitochondrial Isoforms § Exist in dimer-octamer equilibrium § Spatial energy buffering

Cytosolic Isoforms § Subunits: M and B § Dimeric isoenzymes in cytosol (85 k. Da): § MM (muscle-type) § BB (brain-type) § MB hybrid

Cytosolic Isoforms § § § Function as a temporal energy buffer ADP + PCr ATP + Cr Coupled to: § Glycolysis § Actin-myosin system § Temporal Energy Buffering
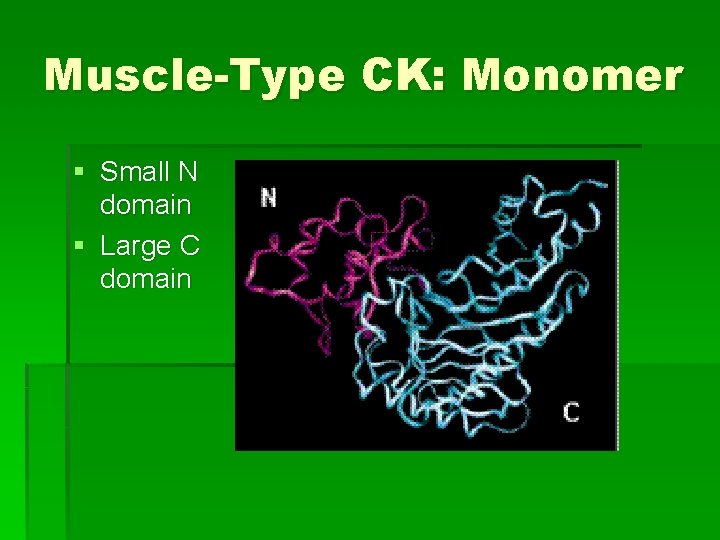
Muscle-Type CK: Monomer § Small N domain § Large C domain
Muscle-Type CK
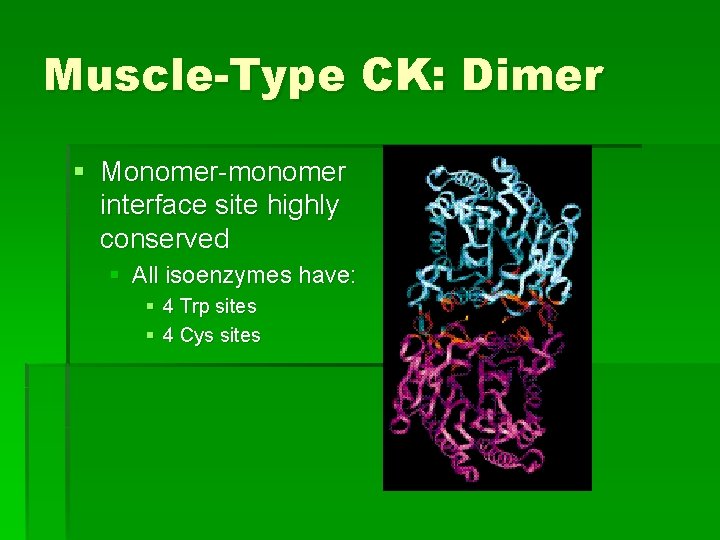
Muscle-Type CK: Dimer § Monomer-monomer interface site highly conserved § All isoenzymes have: § 4 Trp sites § 4 Cys sites
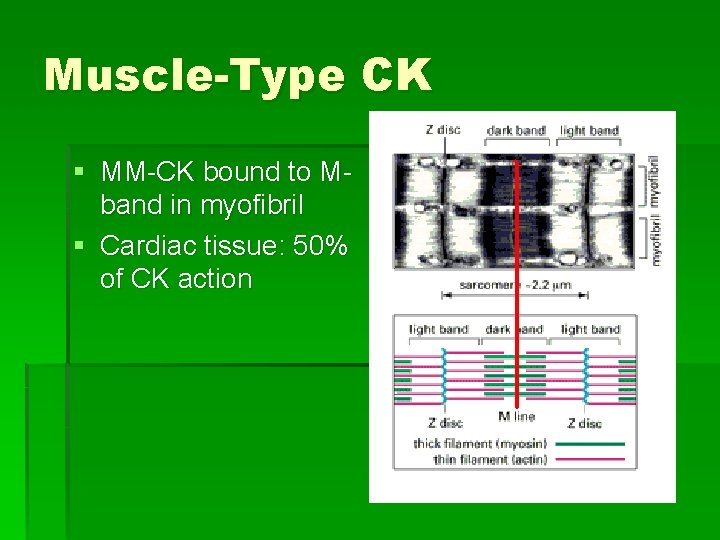
Muscle-Type CK § MM-CK bound to Mband in myofibril § Cardiac tissue: 50% of CK action
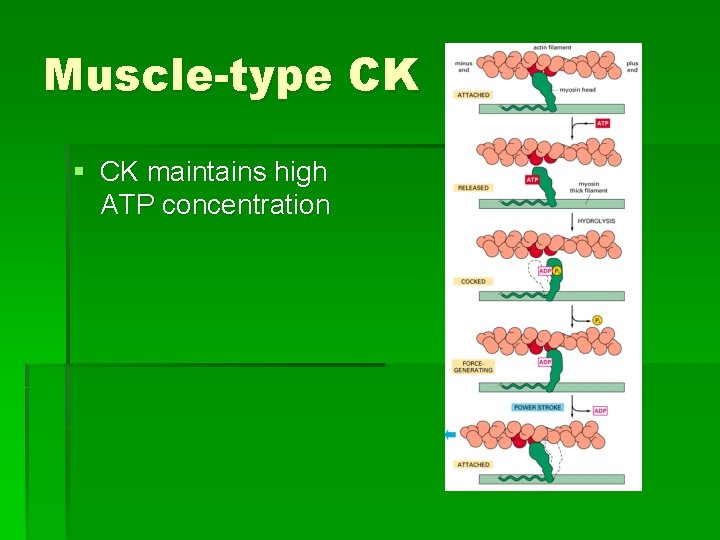
Muscle-type CK § CK maintains high ATP concentration

Muscle-Type CK § Mutation in CK genes linked to myocardial infarction § Heart diseases linked to low levels of CK

Brain-Type CK § § Structure very similar to Muscle-Type CK Most tissues contain MB and BB types High levels in brain, retina, and sperm BB form is the precursor for the other two § BB MM

Brain-Type CK § CK levels associated with learning processes § CK overexpressed in tumours § Decreased CK neurodegeneration

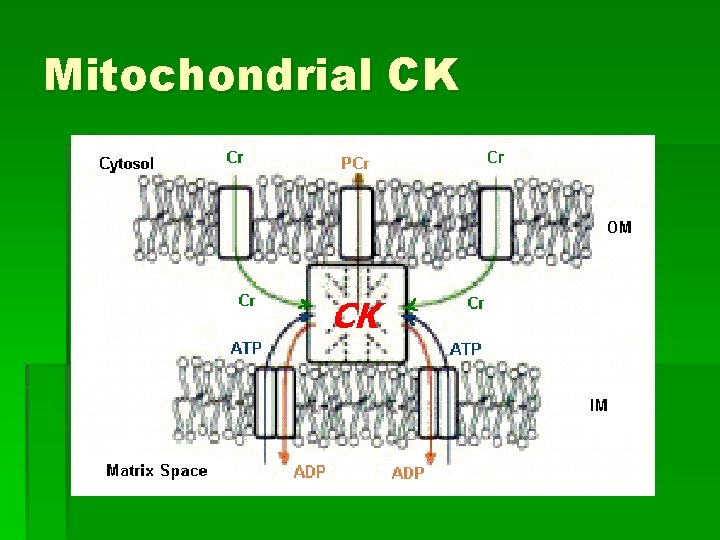
Mitochondrial CK § Bound to outside of inner membrane within cristae § Form microcompartments with porins

Mitochondrial CK § Transphorylation § Cr enters through pore § Cr + ATP PCr + ADP § PCr exits through pore § PCr mediates between sites of ATP consumption and production § Spatial Energy Buffering
Mitochondrial CK
Mi-CK: Structure

Mi-CK: Monomer § Small (residues 1 -112) N-terminal domain § Large (residues 113 -380) C-terminal domain § ATP binding site located in the cleft between the two domains
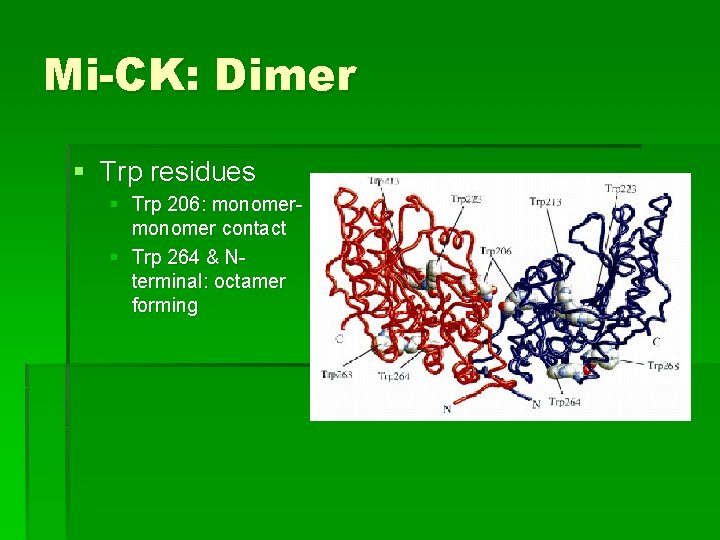
Mi-CK: Dimer § Trp residues § Trp 206: monomer contact § Trp 264 & Nterminal: octamer forming

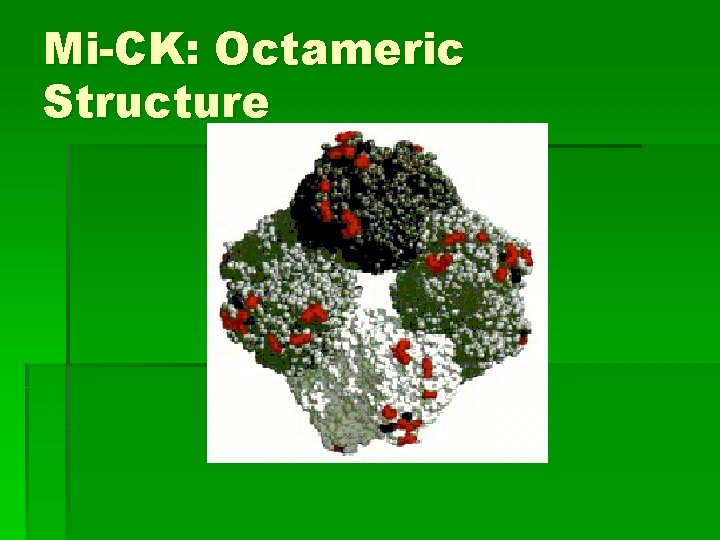
Mi-CK: Octamer § § § stable against denaturation insensitive to proteolysis Dissociation to dimer takes hours to weeks § Accelerated with addition of transition state analogue, TSAC = creatine, Mg. ADP & nitrate

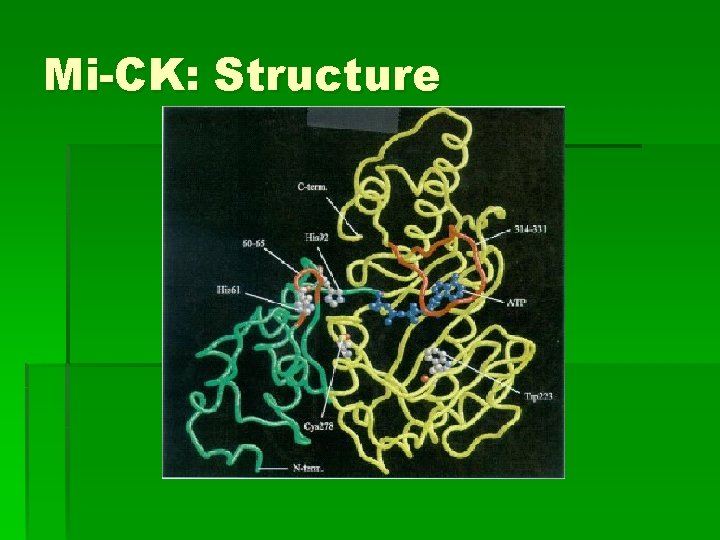
Mi-CK: Structure § Mi-CK fold differs from all other kinases § Structures of Mi-CK-ATP and free enzyme very similar

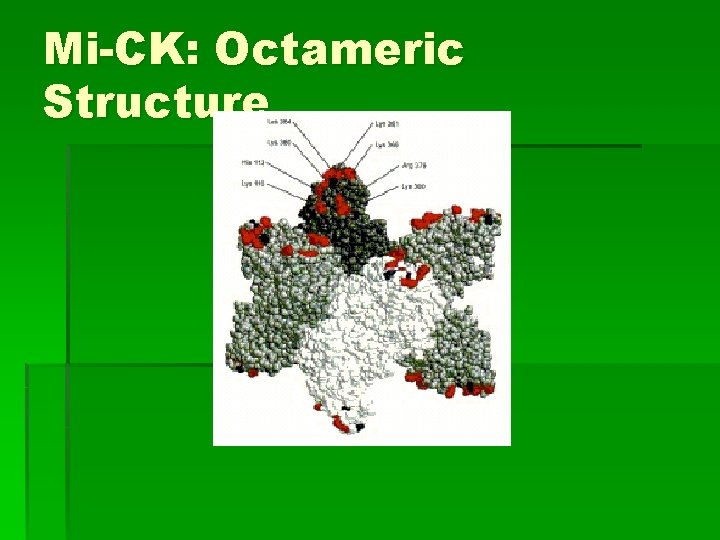
Mi-CK: Structure § Active site residues: § Phosphate groups of ATP interact with Arg residues 125, 127, 287, 315 § Cys 278: substrate binding § His 61: mutation impairs enzyme activity § Loop residues 60 -65 moves toward active site for catalysis § Trp 223: crucial for catalysis
Mi-CK: Octameric Structure
Mi-CK: Octameric Structure
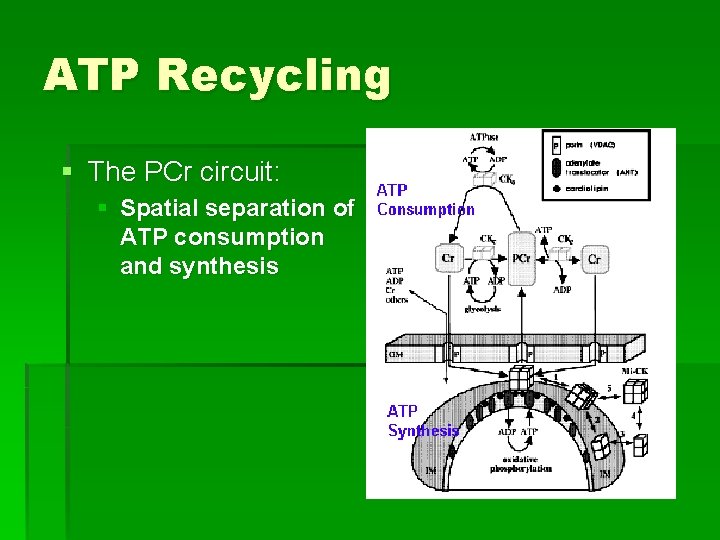
ATP Recycling § The PCr circuit: § Spatial separation of ATP consumption and synthesis

Mitochondrial VS Cytosolic CK § Very similar structures and structural elements § Mi-CK evolved different folding pattern for catalyzing phosphoryl transfer § Allow compartmentalization of function

References 1. Wallimann T et al. 1998. Some new aspects of creatine kinase (CK): compartmentation, structure, function and regulation for cellular and mitochondrial bioenergetics and physiology. Biofactors 8, 229 -234. 2. Schlattner U et al. 1998. Functional aspects of the X-ray structure of mitochondrial creatine kinase: A molecular physiology approach. Molecular and Cellular Biochemistry 184, 125140. 3. Yamamichi H et al. 2001. Creatine kinase gene mutation in a patient with muscle creatine kinase deficiency. Clinical Chemistry 47, 1967 -1973. 4. Alberts B et al. 1994. Molecular Biology of the Cell, 3 rd edition. New York: Garland Publishing. 5. Lipskaya TY. 2000. The physiological role of the creatine kinase system: evolution of views. Biochemistry (Moscow) 66, 115 -129.
- Slides: 33