CRE and MDRA Notifiable Conditions Neil Pascoe RN

CRE and MDR-A Notifiable Conditions Neil Pascoe RN BSN CIC Epidemiologist Emerging and Infectious Disease Branch Infectious Disease Control Unit Texas HAI Advisory Panel Meeting 1/17/14
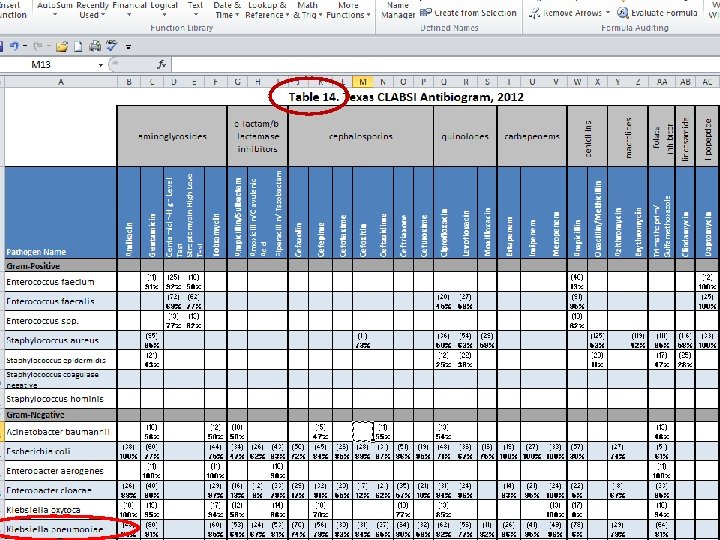
Table 14. Texas CLABSI Antibiogram, 2012
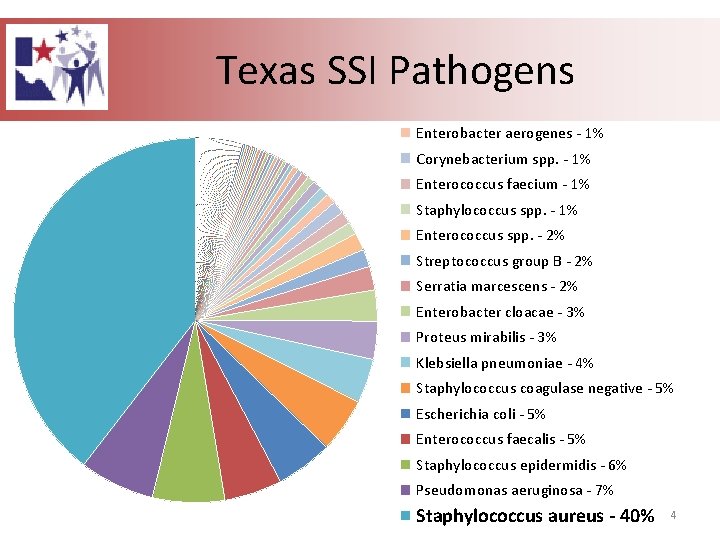
Texas SSI Pathogens Enterobacter aerogenes - 1% Corynebacterium spp. - 1% Enterococcus faecium - 1% Staphylococcus spp. - 1% Enterococcus spp. - 2% Streptococcus group B - 2% Serratia marcescens - 2% Enterobacter cloacae - 3% Proteus mirabilis - 3% Klebsiella pneumoniae - 4% Staphylococcus coagulase negative - 5% Escherichia coli - 5% Enterococcus faecalis - 5% Staphylococcus epidermidis - 6% Pseudomonas aeruginosa - 7% Staphylococcus aureus - 40% 4
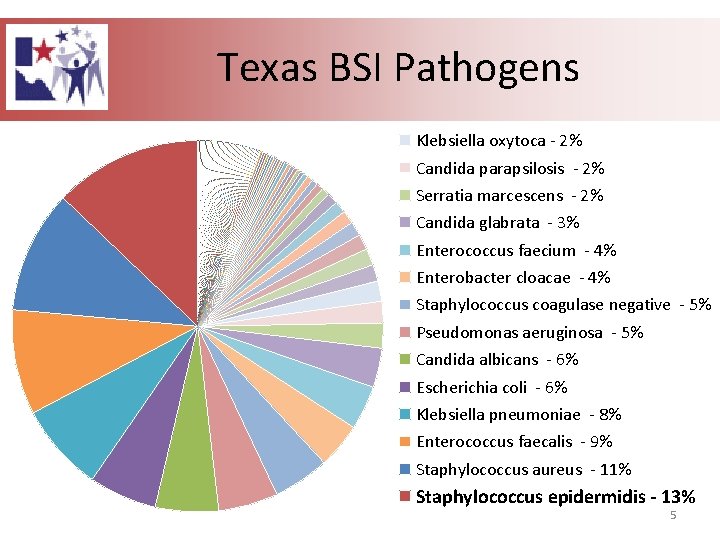
Texas BSI Pathogens Klebsiella oxytoca - 2% Candida parapsilosis - 2% Serratia marcescens - 2% Candida glabrata - 3% Enterococcus faecium - 4% Enterobacter cloacae - 4% Staphylococcus coagulase negative - 5% Pseudomonas aeruginosa - 5% Candida albicans - 6% Escherichia coli - 6% Klebsiella pneumoniae - 8% Enterococcus faecalis - 9% Staphylococcus aureus - 11% Staphylococcus epidermidis - 13% 5

Flagged HAI Pathogen Data • Central Office HAI Staff: Notifiable pathogens • Central Office/Regional HAI Epidemiologist: – VRSA, VISA – GAS in SSI wound – Flagged Facilities – ≥ 3 of same organism in same ICU per month – CRE- any reported – S. aureus: ≥ 10 per facility/6 months – Acinetobacter: ≥ 5 per facility/6 months – Serratia: ≥ 3 per facility/6 months 6
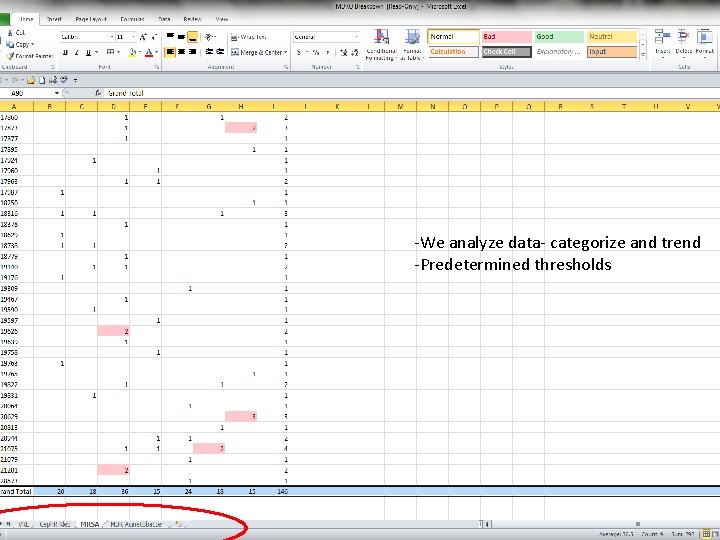
-We analyze data- categorize and trend -Predetermined thresholds
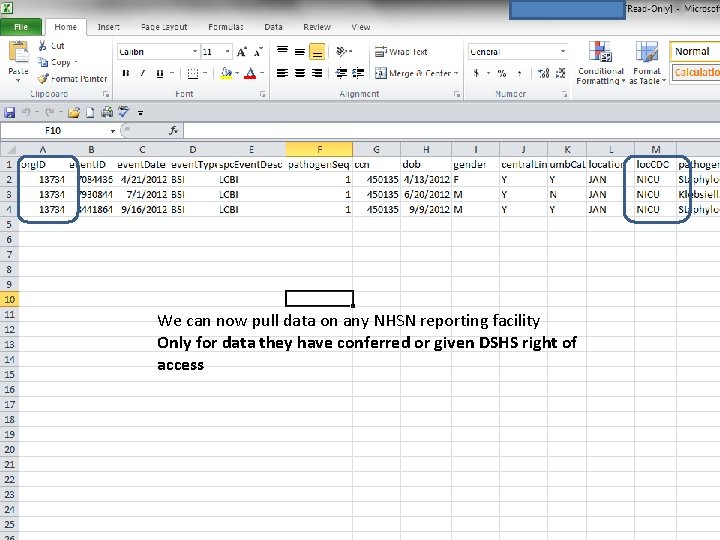
We can now pull data on any NHSN reporting facility Only for data they have conferred or given DSHS right of access

MDRO Reporting Ø CDC Tool Kit (http: //www. cdc. gov/HAI/organisms/cre/) Ø CRE E. coli, Klebsiella species, MDR Acinetobacter • Voluntary 2014 • Mandatory April 2014 • DSHS lab capacity Ø All source site infections to be reported Ø No isolate submission mandated Ø NHSN case definitions
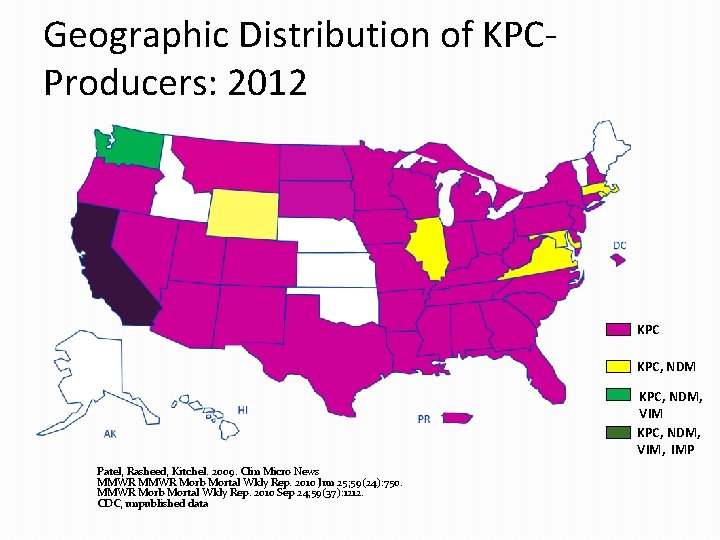
Geographic Distribution of KPCProducers: 2012 KPC, NDM, VIM, IMP Patel, Rasheed, Kitchel. 2009. Clin Micro News MMWR Morb Mortal Wkly Rep. 2010 Jun 25; 59(24): 750. MMWR Morb Mortal Wkly Rep. 2010 Sep 24; 59(37): 1212. CDC, unpublished data


Problems Identifying CRE FDA and CLSI breakpoints differ for some carbapenems Manufacturers of automated susceptibility testing systems must use FDA breakpoints – 2010 breakpoints Many automated systems in use do not have cards that identify 2012 CLSI breakpoints - not validated Automated systems appear to “overcall” resistance compared to reference methods No easy way right now to check for carbapenemases MHT - modified Hodge test PCR – molecular probes for specific genes FDA = Food and Drug Administration CLSI = Clinical and Laboratory Standards Institute

Surveillance and Definitions Facilities/Regions should have an awareness of the prevalence of CRE in their Facility and Community Suggested focus on Klebsiella and E. coli CDC surveillance definition (2012 breakpoints) Nonsusceptible to one of the carbapenems: doripenem, meropenem, or imipenem AND Resistant to all 3 rd generation cephalosporins tested (Some Enterobacteriaceae are intrinsically resistant to imipenem e. g. Morganella, Providencia, Proteus)

CRE Definition q (3) Carbapenem resistant Enterobacteriaceae (CRE)CRE-Ecoli or CRE-Klebsiella as defined in the Centers for Disease Control and Prevention, National Healthcare Safety Network (NHSN) Manual, Patient Safety Component, Protocol for Multidrug-Resistant Organism and Clostridium difficile Infection (MDRO/CDI) Module, or its successor. * * http: //www. sos. state. tx. us/texreg/pdf/backview/1206 prop. pdf

MDR-A Definition q (18) Multi-drug resistant (MDR) Acinetobacter--MDRAcinetobacter as defined by the Centers for Disease Control and Prevention, National Healthcare Safety Network (NHSN) Manual, Patient Safety Component, Protocol for Multidrug. Resistant Organism and Clostridium difficile-Infection (MDRO/CDI) Module, or its successor* * http: //www. sos. state. tx. us/texreg/pdf/backview/1206 prop. pdf




Cluster Investigations Ø South Texas spring 2013 Ø Beaumont spring 2013 Ø Type of patient population, source of infection, same organism? Ø PFGE? What actions do you take when you find a cluster ? For this purpose, how do you define a cluster?
- Slides: 19