Cracking Cracking may be defined as the process

� Cracking : �Cracking may be defined as the process of the decomposition of the higher molecular weight hydrocarbon into lower molecular weight hydrocarbon. �C 10 H 22 C 5 H 12 + C 5 H 10 �(B. P. =174°C) (B. P. =36°C) �Decane n-pentene �Cracking process are of two types : �(a) Thermal Cracking � (b) Catalytic Cracking

THERMAL CRACKING � When cracking is carried out by applying high heat & pressure , it is known as thermal cracking. Liquid phase Thermal cracking Vapour phase thermal (i)Liquid phase Thermal Cracking: - Heavy oil (475 -530°C, high P 7 -10 atm. ) Gasoline(60 -65%) Octane rating of petrol is 70

�(ii) Vapour phase thermal cracking : Kerosene oil ( 670 -720 475 -530°C, high P 7 -10 atm. ) Gasoline 70% (octane No. is 70)

�CATALYTIC CRACKING: In this cracking take place in presence of catalyst usually mixture of silica & alumina or Aluminum silicate. It is known as catalytic cracking. **better than thermal cracking : * not required high pressure * no external fuel is necessary * due to presence of isoparaffin & aromatic compounds quality of gasoline improved. Two type of Catalytic Cracking : (a) Fixed Bed Catalytic Cracking (b) Moving Bed Catalytic Cracking
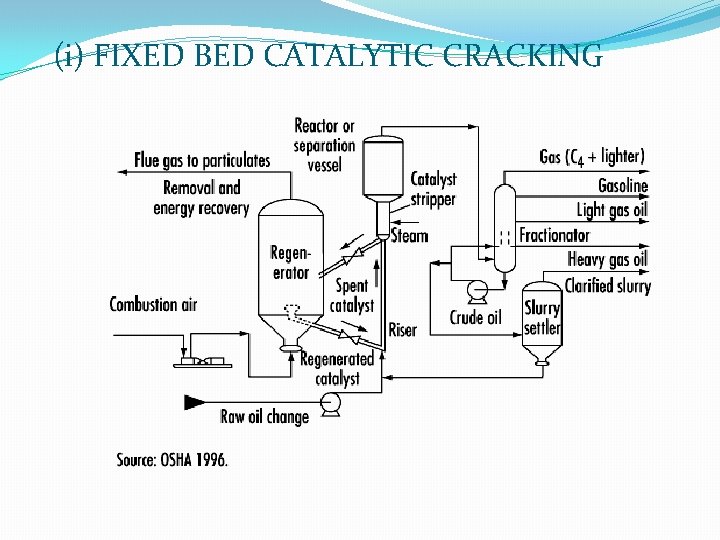
(i) FIXED BED CATALYTIC CRACKING

� Heavy oil vapour Preheater (425 -450°C) Catalyst Chamber (Catalyst Si. O 2 -Al 2 O 3 gel mixed with clay &Zr. O ) (about 40% charge converted into gasoline & 2 -4% C deposited on catalytic bed &deactivated it ) Cracked Vapour passed Fractionating column (some gas get condensed along with gasoline & uncondensed gases move up) Gasoline(send to stabilizing column to remove the dissolved gas) Get Pure Gasoline
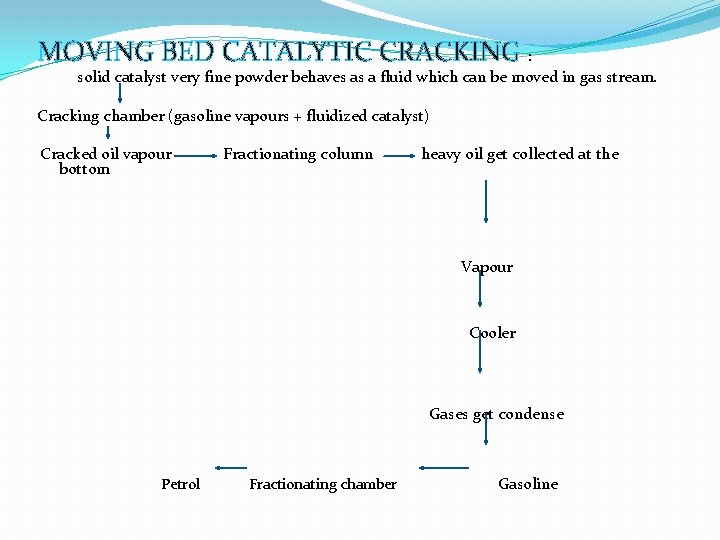
MOVING BED CATALYTIC CRACKING : solid catalyst very fine powder behaves as a fluid which can be moved in gas stream. Cracking chamber (gasoline vapours + fluidized catalyst) Cracked oil vapour bottom Fractionating column heavy oil get collected at the Vapour Cooler Gases get condense Petrol Fractionating chamber Gasoline

Diagram II

REFINING �The straight run gasoline obtained by fractional distillation contains undesirable impurities like compounds of S & some olefins. Therefore , it is essential to refine the gasoline before taking in use. 1. Removal of S Compounds: Gasoline (contain Mercaptane named as Sour their removal in the form of disulphide is called sweetening). Therefore disulpurised petrol (sweet petrol). a. Add Na 2 Pb. O 2 + 2 RSH Pb(SR)2 + 2 Na. OH (Sodium plum bite) (Lead Mercaptide)

b. Pb(SR)2 + add S (Lead Mercaptide) Pb. S + Removal by filteration RS-SR oil Soluble 2. Removal of Olefins & Coloring matter: Gasoline + 80% cold H 2 SO 4 Olefins removed Gasoline + Fuller’s earth Coloring impurities remove 3. Stabilization : Gasoline fractionating column Saturated hydrocarbon Refined gasoline is stored & inhibitors like aromatic amines & phenyl are added to prevent oxidation.

�REFORMING In this process ant knocking gasoline improved. TWO TYPES (a) Thermal Reforming Straight chain HC 500 -600° (b) Catalytic Reforming properties of Brancd Chain & aromatic HC formed carried out in presence of catalyst Pt supported over Alumina. (i) Dehydrogenation (ii) Dehydrocyclization (iii) Hydrocracking

- Slides: 12